Revolutionizing Isoquinoline Production: A Green Rhodium-Catalyzed Route for Commercial Scale Manufacturing
Revolutionizing Isoquinoline Production: A Green Rhodium-Catalyzed Route for Commercial Scale Manufacturing
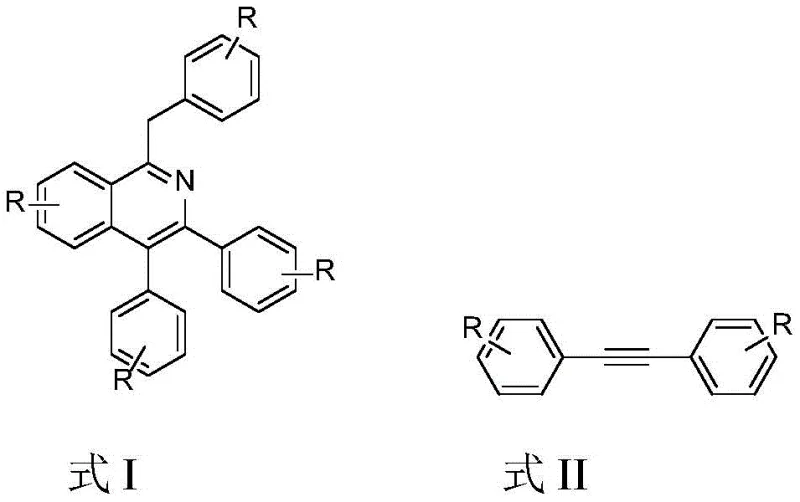
The pharmaceutical and fine chemical industries are constantly seeking more efficient and environmentally benign pathways to construct complex heterocyclic scaffolds, particularly the isoquinoline core which is ubiquitous in bioactive alkaloids and functional materials. Patent CN108484499B introduces a groundbreaking methodology for preparing polysubstituted isoquinoline derivatives directly from hydroxylamine and diaryl alkyne compounds. This innovation represents a significant leap forward from classical condensation reactions, utilizing a trivalent rhodium catalyst to drive the cyclization under relatively mild conditions in ethanol. For R&D directors and procurement specialists, this technology offers a compelling alternative to traditional routes that often suffer from harsh acidic conditions and poor atom economy. By leveraging common raw materials like hydroxylamine and alkynes, this process not only simplifies the synthetic sequence but also aligns perfectly with modern green chemistry principles, reducing the environmental footprint associated with large-scale manufacturing of high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isoquinoline skeleton has relied heavily on named reactions such as the Bischler-Napieralski or Pictet-Spengler reactions, which, while effective, present substantial drawbacks for modern industrial application. These classical methods typically necessitate the use of strong acids and aggressive dehydrating agents like phosphorus pentoxide, creating severe corrosion issues for reactor equipment and generating hazardous waste streams that are costly to treat. Furthermore, many contemporary transition-metal catalyzed approaches, such as those developed by Larock or Fagnou, often require pre-functionalized substrates containing halogens or complex nitrogen-containing precursors that involve multi-step syntheses. A critical bottleneck in these existing technologies is the frequent requirement for stoichiometric amounts of oxidants, such as copper acetate, which not only increases raw material costs but also generates significant quantities of heavy metal waste. This reliance on toxic reagents and complex substrate preparation limits the scalability and economic viability of producing diverse isoquinoline libraries needed for rapid drug development and material science applications.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN108484499B utilizes a direct oxidative annulation strategy that bypasses the need for pre-oxidized substrates or external oxidants. The core of this innovation lies in the use of readily available diaryl alkyne compounds and hydroxylamine as the nitrogen source, reacting in a simple alcohol solvent like ethanol. This one-pot procedure eliminates the need for intermediate isolation and purification steps, drastically reducing processing time and solvent consumption. The reaction proceeds via a rhodium-catalyzed C-H activation mechanism that is highly selective, tolerating a wide range of functional groups including alkyl, alkoxy, and halogen substituents without the need for protecting groups. By completing the cyclization through simple dehydration, the process achieves high atom utilization, with water and catalytic amounts of potassium chloride being the only byproducts. This streamlined approach not only enhances the overall yield but also significantly lowers the barrier to entry for manufacturing complex polysubstituted isoquinolines on a commercial scale.
Mechanistic Insights into Rhodium-Catalyzed Cyclization
The success of this synthetic route hinges on the unique ability of the trivalent rhodium catalyst, specifically [Cp*RhCl2]2, to activate inert carbon-hydrogen bonds and facilitate the formation of new carbon-nitrogen bonds efficiently. In this catalytic cycle, the rhodium species coordinates with the alkyne and the in situ generated nitrone or similar nitrogenous intermediate derived from hydroxylamine. This coordination lowers the activation energy for the subsequent migratory insertion and reductive elimination steps that form the isoquinoline ring. The use of potassium acetate as a base is crucial, as it assists in the deprotonation steps required to regenerate the active catalytic species without introducing harsh conditions that could degrade sensitive functional groups. The mechanism avoids the formation of radical species that often lead to polymerization or side reactions in free-radical processes, ensuring a clean reaction profile. This precise control over the reaction pathway is what allows the system to accommodate diverse substrates, from electron-rich methoxy-substituted alkynes to electron-deficient halogenated variants, maintaining high selectivity and minimizing the formation of regioisomers that would complicate downstream purification efforts.
From an impurity control perspective, the absence of external oxidants like copper salts is a major advantage for ensuring the purity of the final pharmaceutical intermediate. Traditional oxidative couplings often leave trace amounts of heavy metals in the product, requiring expensive and time-consuming scavenging processes to meet strict regulatory limits for drug substances. In this rhodium-catalyzed system, the catalyst loading is kept low, typically around 1.0 mol%, and the byproducts are non-toxic salts and water. The high chemical selectivity of the reaction means that side products arising from over-oxidation or substrate decomposition are minimal. This inherent cleanliness of the reaction mixture simplifies the work-up procedure, often allowing for straightforward crystallization or basic chromatography to achieve high-purity standards. For quality control teams, this translates to more consistent batch-to-batch reproducibility and a reduced risk of genotoxic impurities, which is a critical consideration when supplying intermediates for oncology or antiviral drug candidates where isoquinoline scaffolds are frequently employed.
How to Synthesize Polysubstituted Isoquinoline Efficiently
To implement this robust synthetic methodology in a laboratory or pilot plant setting, operators must adhere to specific parameters regarding temperature, pressure, and reagent ratios to maximize yield and safety. The process is designed to be operationally simple, requiring only a single charging of reagents followed by heating in a sealed vessel, which minimizes human error and exposure to hazardous chemicals. Detailed standard operating procedures should emphasize the importance of maintaining an inert nitrogen atmosphere to prevent any potential interference from oxygen, although the system shows some tolerance. The following guide outlines the critical steps derived from the patent examples to ensure successful replication of the high yields reported, serving as a foundational protocol for process chemists looking to adapt this technology for specific target molecules.
- Mix diaryl alkyne compound, aqueous hydroxylamine solution, potassium acetate base, and [Cp*RhCl2]2 catalyst in ethanol solvent within a sealed tube.
- Heat the reaction mixture to 140°C under a nitrogen atmosphere and stir continuously for 12 to 24 hours to ensure complete cyclization.
- Purify the crude product via silica gel column chromatography using petroleum ether and ethyl acetate as eluents to isolate the high-purity isoquinoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed technology offers tangible benefits that extend beyond mere chemical elegance, directly impacting the bottom line and operational resilience. The shift from multi-step, oxidant-heavy processes to a one-pot dehydration cyclization fundamentally alters the cost structure of isoquinoline manufacturing. By eliminating the need for stoichiometric oxidants and complex substrate pre-functionalization, the raw material bill of materials is significantly reduced. Furthermore, the use of ethanol as a solvent, a commodity chemical with a stable global supply chain, replaces more exotic or toxic solvents often required in traditional heterocycle synthesis. This simplification of the supply chain reduces the risk of disruptions caused by the scarcity of specialized reagents and ensures a more predictable procurement cycle for long-term production contracts.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the drastic simplification of the reaction workflow and the elimination of expensive waste treatment protocols. Since the reaction does not require stoichiometric amounts of copper acetate or other heavy metal oxidants, the cost associated with purchasing these reagents and subsequently disposing of the hazardous heavy metal waste is completely removed. Additionally, the high atom economy means that a larger proportion of the starting mass ends up in the final product, reducing the effective cost per kilogram of the active ingredient. The low catalyst loading further contributes to cost efficiency, as the expensive rhodium catalyst is used in minute quantities and can potentially be recovered or recycled in a continuous flow setup. These factors combined result in a substantially lower cost of goods sold, making the final isoquinoline derivatives more competitive in the global market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as hydroxylamine, diaryl alkynes, and ethanol ensures a robust and resilient supply chain that is less susceptible to geopolitical or logistical shocks. Unlike processes that depend on bespoke, multi-step synthesized intermediates which may have limited suppliers, the starting materials for this reaction are widely available from multiple global vendors. This diversity in sourcing options empowers procurement teams to negotiate better pricing and secure backup supplies easily. Moreover, the simplicity of the one-pot reaction reduces the dependency on highly specialized contract manufacturing organizations that possess complex multi-step capabilities, allowing for more flexible production scheduling and faster response times to fluctuating market demands for pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the use of standard stainless steel reactors capable of handling moderate temperatures and pressures, without the need for specialized lining to resist strong acids. The green nature of the process, generating only water and salt as byproducts, aligns perfectly with increasingly stringent environmental regulations regarding industrial effluent. This compliance reduces the regulatory burden and the capital expenditure required for waste treatment facilities. The ability to run the reaction in alcohol solvents also improves safety profiles by avoiding chlorinated solvents, thereby lowering insurance premiums and operational risks. Consequently, manufacturers can scale up production volumes rapidly to meet commercial demand without facing the typical bottlenecks associated with environmental permitting and hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing clarity for technical teams evaluating the feasibility of this method for their specific projects. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: Is an external oxidant required for this isoquinoline synthesis?
A: No, unlike traditional methods that require stoichiometric copper acetate or other oxidants, this patented process completes the cyclization through dehydration alone, generating only water and catalytic potassium chloride as byproducts.
Q: What is the optimal catalyst loading for commercial production?
A: The patent specifies a molar dosage of the trivalent rhodium catalyst between 0.5% and 2.5% relative to the alkyne substrate, with 1.0% being the preferred loading for balancing cost and reaction efficiency.
Q: Can this method tolerate halogenated substrates?
A: Yes, the reaction system demonstrates excellent universality and tolerates various substituents including fluorine, chlorine, and bromine, allowing for the synthesis of diverse isoquinoline libraries for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Isoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the rhodium-catalyzed isoquinoline synthesis described in CN108484499B and are fully equipped to translate this laboratory innovation into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity observed in the patent can be consistently replicated on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of isoquinoline intermediate meets the exacting standards required by the global pharmaceutical industry. Our commitment to green chemistry aligns with this technology, allowing us to offer sustainable manufacturing solutions that reduce environmental impact while maintaining cost competitiveness.
We invite R&D directors and procurement leaders to collaborate with us to evaluate the feasibility of this route for your specific pipeline candidates. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener methodology for your supply chain. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your project benefits from the latest advancements in heterocyclic chemistry and secure, reliable supply chain partnerships.
