Advanced Valsartan Manufacturing: High-Yield Synthesis for Global Pharmaceutical Supply Chains
Introduction to Next-Generation Valsartan Synthesis
The global demand for antihypertensive medications continues to surge, placing immense pressure on supply chains to deliver high-purity Active Pharmaceutical Ingredients (APIs) efficiently. Patent CN102417486B introduces a transformative synthetic methodology for Valsartan that addresses critical bottlenecks in traditional manufacturing. This innovative approach leverages a bimetallic copper-iron catalytic system for the initial coupling and a zinc-mediated tetrazole formation, effectively bypassing the hazardous and low-yielding tributyltin azide steps common in legacy processes. By optimizing reaction conditions to mild temperatures between 30-35°C and utilizing recyclable solvents, this technology offers a robust pathway for reliable valsartan intermediate supplier operations. The strategic shift away from toxic tin reagents not only aligns with modern green chemistry principles but also significantly streamlines the downstream purification workflow, ensuring a consistent supply of high-quality therapeutic agents for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Valsartan has been plagued by inefficient multi-step sequences and the reliance on hazardous reagents that complicate scale-up. Early literature describes routes exceeding 14 steps, involving complex protection and deprotection strategies that inherently limit overall throughput. More critically, established methods often utilize tributyltin azide for the construction of the essential tetrazole ring, a step that typically suffers from yields around 70% and generates persistent organotin waste. The removal of these tin residues requires rigorous and costly purification protocols to meet safety standards, creating a significant burden on cost reduction in API manufacturing. Furthermore, the use of high-boiling solvents like toluene in acylation steps necessitates energy-intensive distillation for recovery, while side reactions induced by strong bases like diisopropylethylamine can lead to difficult-to-separate impurities that compromise the final drug substance quality.
The Novel Approach
The methodology outlined in CN102417486B presents a streamlined alternative that fundamentally re-engineers the synthesis for industrial viability. By employing a copper and iron salt catalytic system for the N-C bond formation, the process achieves conversion rates exceeding 90%, a substantial improvement over the 80% typical of non-catalyzed or single-metal systems. The replacement of tributyltin azide with a zinc chloride and sodium azide system in ethanol represents a paradigm shift, enabling the tetrazole ring closure to proceed with yields greater than 90% under reflux conditions. This novel approach eliminates the need for toxic tin reagents entirely, thereby removing the associated environmental liabilities and purification complexities. Additionally, the use of dichloroethane and ethanol allows for efficient solvent recovery loops, supporting the commercial scale-up of complex pharmaceutical intermediates with a markedly reduced ecological footprint and enhanced operational safety profile.
Mechanistic Insights into Cu/Fe-Catalyzed Coupling and Zn-Mediated Cyclization
The core innovation of this synthesis lies in the synergistic catalytic effects observed during the initial coupling and the subsequent heterocycle formation. In the first step, the combination of Cu salts (such as CuCl2 or CuCN) and Fe salts (like FeCl2 or Fe3O4) creates a highly active catalytic environment that facilitates the nucleophilic substitution of the bromomethyl group by the valine amine. This bimetallic system likely operates through a mechanism that stabilizes the transition state and prevents the racemization of the chiral center, ensuring the retention of the L-configuration essential for biological activity. The reaction proceeds smoothly at 30-35°C in dichloroethane, avoiding the harsh conditions that often degrade sensitive amino acid derivatives. Following acylation, the tetrazole formation utilizes zinc chloride to activate the nitrile group towards nucleophilic attack by the azide ion. The presence of triethylamine hydrochloride as a phase transfer catalyst is crucial, as it enhances the solubility of the inorganic azide in the organic ethanol phase, driving the equilibrium towards the formation of the five-membered ring with exceptional efficiency.
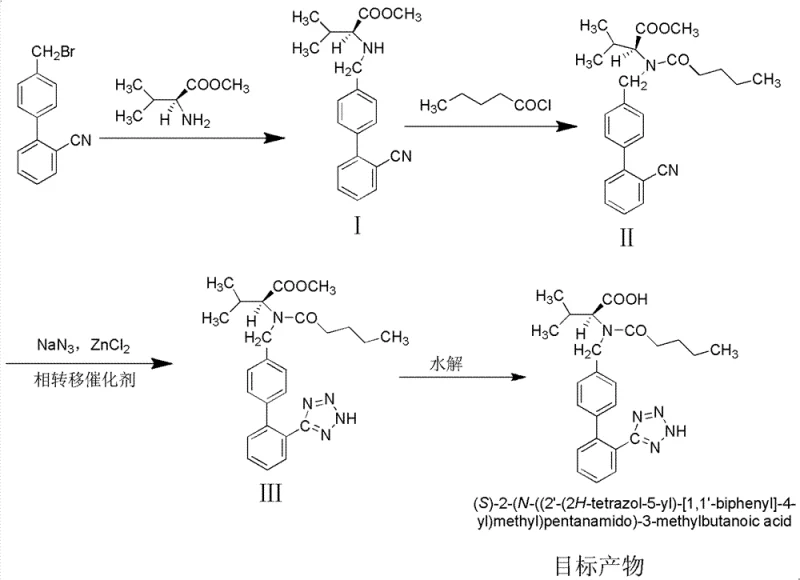
Impurity control is rigorously managed through the specific choice of reagents and workup procedures designed to minimize side products. The use of aluminum trichloride in the acylation step at 0°C ensures selective N-acylation without affecting the ester or nitrile functionalities, preventing the formation of O-acylated byproducts. During the azidation phase, the zinc-mediated pathway avoids the generation of polymeric azide species often seen with tin reagents, resulting in a cleaner reaction profile. The final hydrolysis step is conducted at controlled low temperatures of 0-5°C using sodium hydroxide, which selectively cleaves the methyl ester while preserving the integrity of the newly formed tetrazole ring. Any trace D-isomers generated during the process are effectively removed through a dedicated optical purification stage involving crystallization from isopropyl ether and ethyl acetate mixtures, guaranteeing that the final high-purity antihypertensive intermediates meet the strict enantiomeric excess requirements mandated by regulatory bodies for clinical application.
How to Synthesize Valsartan Efficiently
The execution of this synthetic route requires precise control over stoichiometry and temperature to maximize the benefits of the catalytic systems described. The process begins with the preparation of the key intermediate N-(2'-cyanobiphenyl-4-methylene)-L-valine methyl ester, followed by sequential acylation and cyclization. Operators must adhere strictly to the specified molar ratios, particularly the 1:1 to 1:1.5 ratio of valine ester to bromide, to prevent unreacted starting materials from carrying over. The detailed standardized synthesis steps, including specific workup procedures for solvent recovery and product isolation, are critical for reproducing the high yields reported in the patent documentation. For a comprehensive breakdown of the operational parameters and safety protocols required for each unit operation, please refer to the technical guide below.
- Perform N-C coupling of 4'-bromomethyl-2-cyanobiphenyl with L-valine methyl ester using Cu/Fe catalysts in dichloroethane at 30-35°C.
- Execute acylation of the intermediate with n-pentanoyl chloride using AlCl3 catalyst at 0°C to form the N-pentanoyl derivative.
- Conduct azidation cyclization using sodium azide and zinc chloride in ethanol with a phase transfer catalyst to form the tetrazole ring.
- Complete the synthesis by hydrolyzing the methyl ester under alkaline conditions at 0-5°C to obtain the final Valsartan acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this optimized synthesis route offers compelling advantages that directly address the pain points of modern pharmaceutical procurement. The elimination of tributyltin azide removes a major supply chain vulnerability, as tin reagents are subject to fluctuating availability and stringent handling regulations. By shifting to commodity chemicals like sodium azide and zinc chloride, manufacturers can secure a more stable and predictable supply of raw materials, enhancing supply chain reliability for critical medication production. The simplified purification train, devoid of heavy metal scavenging steps, reduces the dependency on specialized consumables and shortens the overall production cycle time. This operational efficiency translates into substantial cost savings, not merely through raw material pricing but through the drastic reduction in waste disposal costs and energy consumption associated with solvent recycling and distillation processes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the high atom economy and the use of inexpensive, recyclable solvents such as ethanol and dichloroethane. By achieving yields exceeding 75% for the methyl ester intermediate compared to the 40% typical of older technologies, the consumption of starting materials per ton of final product is significantly lowered. The removal of expensive tin catalysts and the associated cleanup resins further decreases the variable cost of goods sold. Additionally, the ability to recycle auxiliary materials like organic bases and solvents directly back into the process minimizes waste treatment expenses, creating a leaner and more cost-effective manufacturing model that improves margin potential for generic producers.
- Enhanced Supply Chain Reliability: The reliance on widely available inorganic salts and common organic solvents mitigates the risk of raw material shortages that can plague specialty reagent supply chains. The robustness of the Cu/Fe catalytic system allows for flexibility in sourcing specific salt grades, reducing the likelihood of production stoppages due to vendor-specific quality issues. Furthermore, the shortened reaction times, particularly in the azidation step where duration is reduced from over 48 hours to under 24 hours, increase the throughput capacity of existing reactor assets. This improved asset utilization enables manufacturers to respond more agilely to market demand spikes, ensuring reducing lead time for high-purity pharmaceutical intermediates and maintaining continuous availability for downstream formulation partners.
- Scalability and Environmental Compliance: Scaling this process to commercial volumes is facilitated by the mild reaction conditions and the absence of pyrophoric or highly toxic reagents. The use of zinc chloride instead of organotin compounds drastically reduces the toxicity profile of the effluent, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. The process generates significantly less hazardous waste, lowering the burden on waste management infrastructure and reducing the carbon footprint of the manufacturing site. This alignment with green chemistry principles not only future-proofs the production facility against regulatory changes but also enhances the brand reputation of the supplier as a responsible partner in the global healthcare ecosystem.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Valsartan synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating technology transfer and procurement officers assessing long-term supply viability. The answers reflect the specific improvements in yield, safety, and purity that distinguish this method from conventional approaches.
Q: How does this synthesis method improve upon traditional tin-based routes?
A: This method replaces toxic tributyltin azide with a safer zinc chloride and sodium azide system. This eliminates the difficult removal of organotin residues, significantly simplifying purification and reducing environmental hazards while maintaining yields above 90% for the key intermediate.
Q: What represents the primary cost driver reduction in this process?
A: The primary cost reduction stems from the use of inexpensive solvents like ethanol and dichloroethane which are easily recycled, alongside the elimination of expensive tin reagents. Furthermore, the high yield of over 75% for the methyl ester intermediate drastically reduces raw material consumption per ton of product.
Q: Is the optical purity of the final product maintained throughout the synthesis?
A: Yes, the process utilizes L-valine methyl ester as the chiral starting material. While minor D-configuration impurities may form, the protocol includes a specific crystallization and optical purification step using NaOH and mixed ethers to ensure the final API meets stringent stereochemical specifications required for pharmaceutical use.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valsartan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102417486B are fully realized in practical manufacturing environments. We are committed to delivering Valsartan and its intermediates with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify identity and potency. Our capability to implement complex catalytic systems allows us to offer a reliable valsartan intermediate supplier service that guarantees consistency and quality batch after batch.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can drive value and security for your antihypertensive drug portfolio.
