Revolutionizing Valsartan Production: A Tin-Free Synthetic Route for Commercial Scalability
Revolutionizing Valsartan Production: A Tin-Free Synthetic Route for Commercial Scalability
The global pharmaceutical landscape is continuously evolving, driven by the urgent need for safer, more efficient, and environmentally sustainable manufacturing processes for critical antihypertensive medications. Patent CN101270096B introduces a groundbreaking methodology for the synthesis of Valsartan, a widely prescribed angiotensin II receptor antagonist, by fundamentally reengineering the traditional catalytic approach. This technical disclosure addresses one of the most persistent challenges in large-scale API manufacturing: the reliance on toxic organotin compounds that complicate purification and pose significant environmental hazards. By leveraging a novel condensation and valerylation strategy, this invention provides a robust pathway that eliminates the need for hazardous tin catalysts entirely, thereby streamlining the production workflow and enhancing the safety profile of the final active pharmaceutical ingredient. For industry stakeholders, this represents not merely a chemical optimization but a strategic advantage in regulatory compliance and supply chain resilience, ensuring that high-purity Valsartan can be delivered consistently to meet global healthcare demands without the burden of heavy metal residue management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Valsartan has heavily relied on the use of halogenated alkyl tin compounds as catalysts during the crucial tetrazole ring formation step, a dependency that introduces severe operational and regulatory bottlenecks. These organotin reagents are notoriously difficult to remove from the reaction mixture, often requiring complex and costly downstream purification processes to meet the stringent International Council for Harmonisation (ICH) guidelines, which mandate that residual organic tin compounds must be maintained below 1 ppm in the finished drug product. Failure to achieve this threshold can result in batch rejection, leading to substantial financial losses and supply disruptions for pharmaceutical manufacturers. Furthermore, the toxicity associated with tin compounds necessitates specialized handling procedures, increased waste treatment costs, and poses significant occupational health risks to plant personnel, making the conventional route increasingly untenable in the context of modern green chemistry initiatives and tightening environmental regulations across major markets.
The Novel Approach
In stark contrast to the legacy methodologies, the innovative process described in patent CN101270096B circumvents the use of any tin-based reagents by utilizing a direct valerylation of a specific biphenyl condensate followed by an azide-mediated cyclization. This approach utilizes readily available starting materials, specifically N-[(2'-cyano-1,1'-biphenyl-4-yl)alkyl]-L-valine ester hydrochloride, which undergoes a controlled acylation reaction to form a stable pentanoyl intermediate. The subsequent cyclization is achieved using metal azides in the presence of amine salts or Lewis acids, effectively constructing the tetrazole ring without generating toxic heavy metal byproducts. This fundamental shift in reaction chemistry not only simplifies the workup procedure by eliminating the need for aggressive tin-scavenging steps but also significantly reduces the environmental footprint of the manufacturing process, aligning perfectly with the sustainability goals of modern pharmaceutical enterprises while maintaining high reaction efficiency and product integrity.
Mechanistic Insights into Tin-Free Tetrazole Cyclization
The core of this technological advancement lies in the precise manipulation of reaction conditions to facilitate the conversion of the cyano group into the tetrazole moiety without metallic catalysis. The process initiates with the valerylation step, where the amino group of the condensate is acylated using valeryl chloride in an aromatic solvent system at controlled low temperatures ranging from 0°C to 30°C. This mild condition prevents side reactions and ensures the formation of the N-(1-pentanoyl) intermediate with high selectivity. Following this, the reaction mixture is subjected to elevated temperatures between 70°C and 150°C in the presence of a metal azide, such as sodium azide or potassium azide, alongside a promoter like triethylamine hydrochloride or zinc chloride. This combination facilitates the [2+3] cycloaddition reaction, effectively transforming the nitrile functionality into the desired tetrazole ring structure while preserving the stereochemical integrity of the L-valine backbone, which is critical for the biological activity of the final antihypertensive agent.
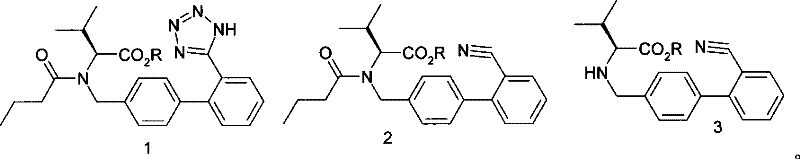
Impurity control is inherently superior in this tin-free regime due to the absence of organometallic residues that typically co-elute with the product during chromatography or crystallization. In traditional tin-catalyzed routes, trace amounts of tin can coordinate with the tetrazole nitrogen atoms, forming stable complexes that are resistant to standard washing protocols, thereby necessitating additional recrystallization cycles that erode overall yield. By removing the tin variable, the impurity profile of the crude reaction mixture becomes significantly cleaner, dominated primarily by unreacted starting materials or simple hydrolysis byproducts that are easily removed through aqueous workups. The protocol specifies a rigorous alkaline hydrolysis step followed by acidification to pH 1-2, which effectively precipitates the free acid form of Valsartan while leaving inorganic salts and organic impurities in the aqueous or organic phases, respectively. This streamlined purification logic ensures that the final crystalline product meets stringent purity specifications with minimal processing effort, directly translating to higher throughput and reduced manufacturing costs.
How to Synthesize Valsartan Efficiently
Implementing this synthesis route requires careful attention to solvent selection and temperature gradients to maximize the conversion efficiency while maintaining operational safety, particularly when handling azide reagents. The patent outlines a standardized procedure where the condensate hydrochloride is first neutralized and acylated in a biphasic system or aromatic solvent, followed by the direct addition of cyclization reagents to the same pot or a transferred solution, minimizing material handling losses. Detailed standard operating procedures regarding the stoichiometric ratios of valeryl chloride, the specific choice of Lewis acid promoters, and the precise cooling rates for crystallization are essential for reproducing the reported yields of over 75% on an industrial scale. Operators must adhere to strict safety protocols during the heating phase to manage the exothermic nature of the azide reaction, ensuring that the reflux conditions are maintained steadily for the prescribed duration of 10 to 50 hours depending on the solvent system employed.
- Perform valerylation by reacting N-[(2'-cyano-1,1'-biphenyl-4-yl)alkyl]-L-valine ester with valeryl chloride in an aromatic solvent at 0-30°C.
- Execute cyclization by treating the pentanoyl intermediate with a metal azide and Lewis acid or amine salt at 70-150°C.
- Isolate the final product through alkaline hydrolysis, acidification, and crystallization from ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this tin-free synthesis technology offers profound advantages that extend beyond mere chemical elegance, directly impacting the bottom line through reduced raw material costs and simplified logistics. The elimination of expensive and hazardous organotin catalysts removes a significant cost center associated with both the purchase of high-purity reagents and the disposal of toxic waste streams, which are subject to increasingly stringent environmental levies. Furthermore, the use of common aromatic solvents like toluene or p-chlorotoluene, which are readily available in bulk quantities from multiple global suppliers, mitigates the risk of supply chain bottlenecks that often plague specialized reagent markets. This accessibility ensures that production schedules remain robust against market volatility, allowing procurement managers to negotiate more favorable terms and secure long-term supply agreements with greater confidence in continuity.
- Cost Reduction in Manufacturing: The removal of organotin catalysts fundamentally alters the cost structure of Valsartan production by eliminating the need for specialized scavenging resins and extensive purification steps required to meet heavy metal limits. Without the burden of tin removal, the consumption of auxiliary materials such as silica gel or activated carbon is drastically reduced, and the number of recrystallization cycles can be minimized, leading to substantial savings in solvent usage and energy consumption. Additionally, the higher overall yield reported in the patent, exceeding 75% from the condensate, means that less raw material is required to produce the same amount of active ingredient, directly lowering the cost of goods sold and improving margin potential for generic manufacturers seeking to optimize their production economics in a competitive marketplace.
- Enhanced Supply Chain Reliability: Relying on a synthesis route that utilizes commodity chemicals rather than specialized organometallic catalysts significantly de-risks the supply chain against geopolitical or logistical disruptions. Since the key reagents such as sodium azide, valeryl chloride, and aromatic solvents are produced by a wide network of chemical manufacturers globally, there is no single point of failure that could halt production. This diversification of the supply base allows for greater flexibility in sourcing and inventory management, ensuring that manufacturing operations can continue uninterrupted even if one supplier faces difficulties. Moreover, the simplified regulatory profile of the final product, free from tin residues, accelerates the quality release process, reducing the time inventory sits in quarantine and improving cash flow velocity for the supply chain organization.
- Scalability and Environmental Compliance: The process is explicitly designed for large-scale industrial production, featuring straightforward unit operations such as liquid-liquid extraction and crystallization that are easily scalable from pilot plants to multi-ton reactors without complex engineering modifications. The absence of toxic tin waste simplifies the environmental compliance landscape, reducing the administrative burden of waste tracking and reporting while lowering the fees associated with hazardous waste disposal. This alignment with green chemistry principles not only enhances the corporate social responsibility profile of the manufacturer but also future-proofs the production facility against tightening environmental regulations, ensuring long-term operational viability and reducing the risk of regulatory shutdowns or fines that could disrupt supply to global customers.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and validation of this tin-free synthetic route, providing clarity for R&D and quality assurance teams evaluating the technology for adoption. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation, focusing on the practical implications of switching from traditional tin-catalyzed methods to this improved protocol. Understanding these nuances is critical for assessing the feasibility of technology transfer and ensuring that the expected benefits in purity and yield are realized in a commercial manufacturing setting.
Q: Why is eliminating organotin catalysts critical for Valsartan production?
A: Organotin compounds are highly toxic and difficult to remove to the required ICH limit of less than 1 ppm. Eliminating them simplifies purification and ensures regulatory compliance.
Q: What solvents are preferred for this tin-free synthesis route?
A: Aromatic solvents such as toluene, p-chlorotoluene, or xylene are preferred. P-chlorotoluene can significantly reduce reaction time to under 10 hours.
Q: How does this method impact the overall yield compared to traditional methods?
A: This improved method achieves a total yield greater than 75% calculated from the condensate, offering competitive efficiency while avoiding heavy metal contamination.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valsartan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the one described in CN101270096B requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this tin-free chemistry are fully actualized in large-volume batches. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every batch of Valsartan meets stringent purity specifications and complies with international pharmacopoeia standards. We understand the critical nature of antihypertensive supply chains and are committed to delivering consistent quality that supports your regulatory filings and market commitments.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply strategy to achieve significant operational efficiencies. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits specific to your volume requirements and regional market dynamics. We encourage you to contact us today to obtain specific COA data from our recent pilot runs and to schedule a comprehensive route feasibility assessment, ensuring that your partnership with NINGBO INNO PHARMCHEM drives both innovation and profitability in your Valsartan procurement portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
