Advanced Pd-Catalyzed Synthesis of Isoquinolones: A Commercial Breakthrough for Pharma Intermediates
Advanced Pd-Catalyzed Synthesis of Isoquinolones: A Commercial Breakthrough for Pharma Intermediates
The landscape of heterocyclic synthesis is undergoing a significant transformation with the disclosure of patent CN113149895A, which details a robust and efficient method for constructing isoquinolone and pyridone scaffolds. These structural motifs are ubiquitous in bioactive natural products and modern drug molecules, serving as critical cores for a wide array of therapeutic agents. Traditionally, accessing these complex cyclic systems has relied on multistep sequences or expensive transition metal catalysts that pose challenges for large-scale manufacturing. This new technology leverages a palladium-catalyzed directed C-H activation strategy, utilizing simple benzamide or acrylamide precursors reacting with widely available phenylpropene derivatives. By shifting the paradigm from costly alkynes to economical alkenes, this innovation offers a compelling value proposition for reliable pharmaceutical intermediate suppliers seeking to optimize their production pipelines. The methodology not only simplifies the synthetic route but also enhances atom utilization, addressing both economic and environmental concerns inherent in fine chemical manufacturing.
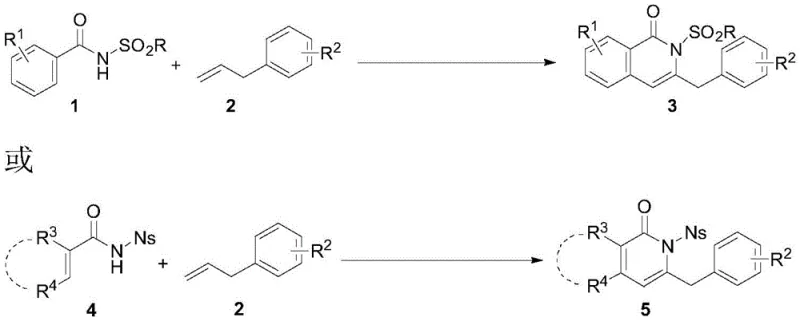
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of isoquinolinone and pyridone rings has been achieved through classical methodologies such as 6 pi-electrocyclic reactions, Larock-type heterocyclizations, or intramolecular aminocarbonyl cyclizations. While chemically valid, these traditional pathways often suffer from significant drawbacks that hinder their commercial viability. A primary limitation is the reliance on complex, pre-functionalized substrates such as o-halobenzamides or internal alkynes, which are frequently expensive and difficult to source in bulk quantities. Furthermore, many of these legacy processes exhibit poor atom economy, generating substantial stoichiometric waste that complicates downstream purification and waste treatment. Transition metal-catalyzed approaches using Rhodium or Cobalt have offered improvements in selectivity but introduce new bottlenecks; these precious metals are not only cost-prohibitive for commodity-scale production but also raise concerns regarding heavy metal residues in final API products. The necessity for stringent metal scavenging steps adds time and cost, eroding the overall efficiency of the manufacturing process.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a tandem reaction scheme involving benzamide or acrylamide compounds and olefin substrates under Palladium catalysis. This method represents a strategic shift towards using olefins, which are naturally abundant, structurally diverse, and significantly cheaper than their alkyne counterparts. The reaction proceeds through a sophisticated cascade of C-H allylation, amine palladation, beta-H elimination, and isomerization, all orchestrated by a Pd(II)/Cu(II) catalytic system. By employing a sulfonyl group (such as Ns, Ts, or Ms) as a directing group, the process achieves exceptional regioselectivity, ensuring that the cyclization occurs precisely at the desired position without forming unwanted isomers. This high level of control eliminates the need for tedious separation of regioisomers, thereby streamlining the workflow. The use of air as a terminal oxidant further underscores the green chemistry credentials of this process, replacing hazardous or expensive stoichiometric oxidants with atmospheric oxygen, which is a critical factor for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Pd-Catalyzed C-H Activation Cyclization
The mechanistic elegance of this transformation lies in the synergistic interaction between the palladium catalyst and the sulfonyl-directed substrate. The reaction initiates with the coordination of the Pd(II) species to the nitrogen atom of the benzamide, facilitated by the electron-withdrawing sulfonyl group which enhances the acidity of the ortho C-H bond. This directs the palladium to activate the specific C-H bond, forming a stable five-membered palladacycle intermediate. Subsequently, the coordinated alkene (phenylpropene) inserts into the Pd-C bond, extending the carbon framework. The presence of the copper co-catalyst, specifically Copper(II) Acetate, plays a dual role: it assists in the re-oxidation of the reduced Pd(0) species back to the active Pd(II) state, closing the catalytic cycle, and may also participate in the activation of the nucleophile. This redox-neutral cycle, driven by aerobic oxidation, ensures that the catalyst loading remains effective throughout the prolonged reaction time of 24 hours at 110 °C.
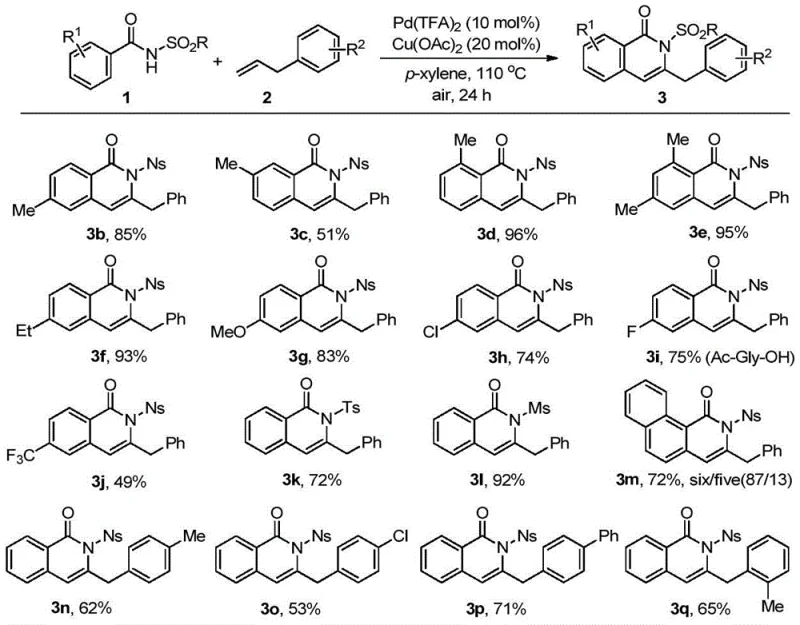
From an impurity control perspective, the choice of the guiding group is paramount. The patent data highlights that groups like Ns (4-nitrophenylsulfonyl) provide superior electronic tuning compared to simpler sulfonamides, leading to higher conversion efficiencies and cleaner reaction profiles. The mechanism inherently suppresses side reactions such as homocoupling of the alkene or non-directed C-H activation, which are common pitfalls in palladium chemistry. The high regioselectivity observed, particularly in the formation of the 3-benzyl substituted isoquinolinones, suggests that the steric and electronic environment created by the catalyst-ligand complex effectively blocks alternative reaction pathways. For R&D teams, understanding this mechanism allows for the rational design of analogs; by modifying the R1, R2, or R3 substituents on the aromatic rings, one can fine-tune the electronic properties of the final molecule without compromising the integrity of the cyclization step, ensuring a robust and predictable synthesis for high-purity pharmaceutical intermediates.
How to Synthesize Isoquinolone Compounds Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it highly attractive for process chemistry teams aiming to transfer technology from the lab to the pilot plant. The standard protocol involves charging a reaction vessel with the benzamide substrate, the phenylpropene coupling partner, palladium trifluoroacetate, and copper acetate in p-xylene. The mixture is then heated to 110 °C under an air atmosphere with open stirring for approximately 24 hours. Upon completion, the workup is straightforward, involving filtration to remove insoluble catalyst residues followed by solvent concentration and purification via standard column chromatography. This simplicity reduces the requirement for specialized equipment such as high-pressure autoclaves or inert gas manifolds, lowering the barrier to entry for adoption. For a detailed breakdown of the specific molar ratios, temperature gradients, and purification parameters, please refer to the standardized synthesis guide below.
- Combine benzamide compound (1) or acrylamide compound (4) with phenylpropene compound (2) in p-xylene solvent.
- Add palladium trifluoroacetate (10 mol%) and copper acetate (20 mol%) as the catalyst system under an air atmosphere.
- Heat the reaction mixture to 110 °C for 24 hours, then purify the resulting isoquinolone or pyridone product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Pd-catalyzed alkene functionalization strategy offers tangible benefits that extend beyond mere chemical novelty. The primary driver for value creation is the substitution of expensive alkyne feedstocks with commodity alkenes. Allylbenzenes and styrene derivatives are produced on a massive industrial scale for the polymer and fragrance industries, ensuring a stable and competitive supply base. This shift drastically reduces the raw material cost basis, insulating the manufacturing process from the volatility often seen in the market for specialized fine chemical building blocks. Furthermore, the use of air as the oxidant eliminates the need to purchase, store, and handle hazardous chemical oxidants like peroxides or hypervalent iodine reagents, which simplifies logistics and reduces safety compliance costs. The overall process intensity is lowered, allowing for longer campaign runs with fewer interruptions for reagent replenishment.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the elimination of precious metal dependencies and expensive substrates. By utilizing a Palladium/Copper system rather than Rhodium or Iridium, the direct catalyst cost is significantly lowered. Moreover, the high atom utilization rate means that a greater proportion of the starting mass ends up in the final product, reducing the cost of goods sold (COGS) associated with waste disposal and raw material consumption. The ability to run the reaction in a common solvent like p-xylene, which is easily recoverable and recyclable, further contributes to substantial cost savings. These factors combine to create a leaner, more cost-effective manufacturing model that improves margin potential for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of generic, widely available reagents. Unlike specialized alkynes which may have single-source suppliers and long lead times, the phenylpropene and benzamide precursors are accessible from multiple global vendors. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Additionally, the robustness of the reaction conditions—tolerating air and moisture better than many sensitive organometallic processes—reduces the likelihood of batch failures due to environmental excursions. This reliability translates to more predictable delivery schedules and the ability to maintain safety stock levels with greater confidence, ensuring continuity of supply for downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this process from gram to multi-ton quantities is facilitated by its thermal and operational stability. The reaction does not require cryogenic temperatures or ultra-high pressures, allowing it to be performed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. From an environmental standpoint, the generation of waste is minimized through high selectivity and the use of aerobic oxidation, aligning with increasingly stringent global regulations on solvent emissions and heavy metal discharge. The simplified workup procedure reduces the volume of aqueous waste generated during quenching and extraction, lowering the burden on wastewater treatment facilities and supporting sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear picture of what partners can expect when integrating this route into their portfolio. Understanding these nuances is essential for making informed decisions about process adoption and vendor selection.
Q: What are the key advantages of this Pd-catalyzed method over traditional Rhodium catalysis?
A: This method utilizes inexpensive palladium catalysts and readily available alkene substrates instead of costly alkynes or allenes required in traditional Rhodium or Cobalt catalyzed routes, significantly reducing raw material costs while maintaining high regioselectivity.
Q: What is the typical yield and purity profile for these isoquinolone derivatives?
A: The patent data demonstrates isolated yields ranging from 44% to 96% across various substrates, with excellent regioselectivity driven by the sulfonyl guiding group, ensuring a clean impurity profile suitable for pharmaceutical applications.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process employs simple operation conditions (open stirring, air atmosphere) and common solvents like p-xylene, avoiding sensitive reagents or extreme pressures, which facilitates straightforward scale-up from gram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Pd-catalyzed C-H activation technology for the next generation of therapeutic agents. As a premier CDMO partner, we possess the technical expertise to rapidly adapt and optimize this proprietary route for your specific project needs. Our facilities are equipped to handle the transition from laboratory discovery to commercial production seamlessly. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your pipeline. Whether you require custom synthesis of novel isoquinolone analogs or reliable supply of established intermediates, our team is ready to assist. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can accelerate your development timelines while optimizing your budget.
