Advanced Synthesis of N-Acetonitrile Bis-Benzenesulfonylimine Derivatives for Pharmaceutical Applications
Advanced Synthesis of N-Acetonitrile Bis-Benzenesulfonylimine Derivatives for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign methodologies for constructing complex molecular architectures, particularly those containing nitrile functionalities which are pivotal in drug design. A significant breakthrough in this domain is documented in patent CN113072470A, which discloses a novel preparation method for N-acetonitrile bis-benzenesulfonylimine derivatives. These compounds, characterized by the general structure shown below, represent a valuable class of intermediates with potential anti-inflammatory activities and utility in organic synthesis. The core innovation lies in the strategic use of hydroxyalkenyl azides as acetonitrile carbon substitutes, reacting with N-fluorobis(benzenesulfonyl)imide under mild catalytic conditions. This approach not only expands the chemical space available for medicinal chemists but also addresses critical supply chain concerns regarding safety and scalability. As a leading entity in the chemical sector, understanding the nuances of such patented technologies is essential for maintaining a competitive edge in the global market.
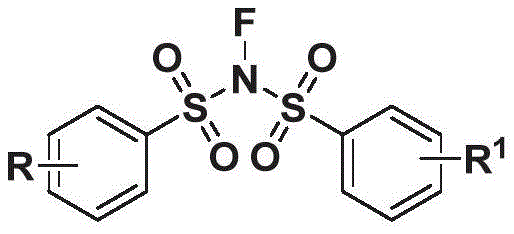
The structural versatility of these derivatives is defined by the substituents R and R1, which can range from simple hydrogen atoms to complex heterocyclic groups, halogens, or trifluoromethyl moieties. This flexibility allows for the fine-tuning of physicochemical properties, making them ideal candidates for structure-activity relationship (SAR) studies in drug discovery programs. For R&D Directors focusing on purity and impurity profiles, the ability to access such diverse analogs through a unified synthetic platform is invaluable. Furthermore, the presence of the sulfonylimine motif suggests potential applications beyond simple intermediates, possibly serving as active pharmaceutical ingredients (APIs) or specialized ligands in catalysis. The patent explicitly highlights the anti-inflammatory potential of these compounds, positioning them as high-value targets for pharmaceutical development. By leveraging this technology, manufacturers can secure a reliable pipeline of high-purity pharmaceutical intermediates that meet the stringent quality standards required by regulatory bodies worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrile compounds has relied heavily on classical methodologies such as the Sandmeyer reaction, the Rosenmund-von Braun reaction, or transition-metal-catalyzed cyanation using toxic sources like copper cyanide (CuCN). While these methods have served the industry for decades, they are increasingly viewed as unsustainable due to their severe environmental and operational drawbacks. The Sandmeyer reaction, for instance, often necessitates the handling of unstable diazonium salts and generates substantial amounts of heavy metal waste, posing significant disposal challenges and safety risks. Similarly, the use of stoichiometric amounts of toxic cyanide sources in Rosenmund-von Braun type reactions creates hazardous working environments and complicates the purification process, often leading to residual metal contamination in the final product. Moreover, many modern C-H bond cyanation strategies, while elegant, frequently require expensive palladium catalysts, harsh oxidants, or specialized ligands that drive up the cost of goods sold (COGS). These factors collectively hinder the commercial viability of traditional routes, especially when scaling up to multi-ton production levels where safety and waste management become paramount concerns for supply chain heads.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN113072470A introduces a paradigm shift by utilizing 2-methyl-3-buten-2-ol derived hydroxyalkenyl azides as a safe and effective cyanating agent. This novel route operates under remarkably mild conditions, typically at temperatures around 50°C, which drastically reduces energy consumption compared to high-temperature traditional processes. The reaction employs a copper-catalyzed system that facilitates the coupling of the vinyl azide with N-fluorobis(benzenesulfonyl)imide, effectively transferring the nitrile group without the need for free cyanide ions. As illustrated in the general reaction scheme below, this transformation is clean, efficient, and produces no toxic by-products, aligning perfectly with green chemistry principles. For procurement managers, this translates to a simplified workflow that eliminates the need for specialized hazardous waste treatment facilities. The use of readily available starting materials further enhances the economic attractiveness of this method, ensuring a stable supply of raw materials and reducing the risk of production bottlenecks caused by scarce reagents.
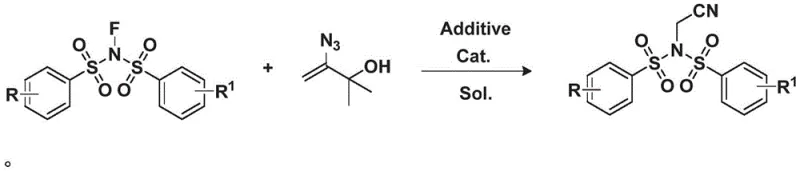
Mechanistic Insights into CuCl-Catalyzed Cyanation
The success of this synthetic methodology hinges on the precise interplay between the copper catalyst and the nitrogen-containing ligand, which together orchestrate the activation of the vinyl azide precursor. Mechanistically, the reaction likely proceeds through a copper-mediated activation of the azide group, followed by a rearrangement or fragmentation that releases nitrogen gas and generates a reactive nitrile equivalent in situ. The presence of 1,10-phenanthroline (Phen) as an additive plays a crucial role in stabilizing the copper species and enhancing the electron density at the metal center, thereby facilitating the nucleophilic attack or insertion steps required for bond formation. Experimental optimization detailed in the patent reveals that cuprous chloride (CuCl) is the preferred catalyst, outperforming other copper salts like CuI or Cu(OAc)2 in terms of conversion and yield. This specificity suggests a delicate balance of Lewis acidity and redox potential that is unique to the CuCl/Phen system. For technical teams, understanding this mechanistic nuance is vital for troubleshooting and process optimization, ensuring that the reaction remains robust even when scaling from gram to kilogram quantities. The mild thermal profile (20-60°C) further indicates a low activation energy barrier, which minimizes the formation of thermal degradation products and ensures a cleaner impurity profile.
Impurity control is another critical aspect where this mechanism offers distinct advantages. Traditional cyanation methods often suffer from side reactions such as homocoupling or over-cyanation, which can be difficult to separate from the desired product. In this novel protocol, the use of N-fluorobis(benzenesulfonyl)imide acts as both a reactant and a protecting group strategy, guiding the reaction towards the specific formation of the N-acetonitrile bis-benzenesulfonylimine scaffold. The by-product of the reaction is essentially benign, avoiding the generation of heavy metal sludge or toxic gases that plague older technologies. This inherent selectivity simplifies the downstream processing, allowing for straightforward extraction and crystallization steps to achieve high purity levels. For R&D directors concerned with regulatory compliance, the absence of genotoxic impurities associated with free cyanide or heavy metal residues is a significant benefit. The ability to tune the electronic nature of the sulfonyl groups via substituents R and R1 also provides a handle to modulate the reactivity and solubility of the intermediates, facilitating easier purification and handling during the manufacturing process.
How to Synthesize N-Acetonitrile Bis-Benzenesulfonylimine Efficiently
Implementing this synthesis on an industrial scale requires strict adherence to the optimized parameters identified in the patent to ensure consistent quality and yield. The process begins with the dissolution of the hydroxyalkenyl azide and the N-fluoro reagent in a suitable organic solvent, with dichloromethane (DCM) being the preferred medium due to its ability to solubilize both reactants and facilitate the catalytic cycle. The addition of the catalyst and additive must be controlled to maintain the correct stoichiometric ratio, typically using a slight excess of the N-fluoro reagent to drive the reaction to completion. Following the reaction period, standard workup procedures involving aqueous quenching and organic extraction are employed to isolate the crude product. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and purification techniques, are outlined in the guide below to assist technical teams in replicating this high-efficiency process.
- Dissolve hydroxyalkenyl azide and N-fluorobis(benzenesulfonyl)imide in dichloromethane (DCM) solvent.
- Add cuprous chloride (CuCl) catalyst and 1,10-phenanthroline (Phen) additive to the reaction mixture.
- Heat the reaction to 50°C for 16 hours, then extract and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits that extend beyond mere chemical efficiency, directly impacting the bottom line and operational resilience of chemical manufacturing enterprises. The primary advantage lies in the drastic simplification of the supply chain for raw materials; 2-methyl-3-buten-2-ol and N-fluorobis(benzenesulfonyl)imide are commercially available and do not suffer from the geopolitical or logistical constraints often associated with specialized cyanating agents. This accessibility ensures a continuous flow of production, mitigating the risk of delays that can disrupt downstream API synthesis. Furthermore, the elimination of toxic cyanide sources removes the need for expensive safety infrastructure and specialized waste disposal contracts, leading to substantial cost savings in operational expenditures. For procurement managers, this translates to a lower total cost of ownership for the intermediate, allowing for more competitive pricing in the final drug product. The mild reaction conditions also reduce energy consumption, contributing to a smaller carbon footprint and aligning with corporate sustainability goals which are increasingly important for multinational clients.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the use of earth-abundant copper catalysts instead of precious metals like palladium or rhodium, which are subject to volatile market prices. By replacing expensive noble metals with inexpensive copper salts, the direct material cost of the catalyst system is reduced by orders of magnitude. Additionally, the high atom economy of the reaction, coupled with the absence of toxic by-products, minimizes the loss of valuable starting materials and reduces the burden on waste treatment facilities. This efficiency means that less raw material is required to produce the same amount of product, directly lowering the variable cost per kilogram. For large-scale operations, these savings accumulate rapidly, providing a strong margin buffer that can be reinvested into R&D or passed on to customers to secure long-term contracts.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on hazardous reagents that require special permits for transport and storage. This novel method circumvents those regulatory hurdles by utilizing stable, non-hazardous precursors that can be shipped and stored using standard logistics protocols. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent qualities, further ensures that production can proceed without interruption even under less-than-ideal circumstances. This reliability is crucial for meeting tight delivery schedules demanded by pharmaceutical clients, who cannot afford delays in their own clinical or commercial timelines. By securing a source of intermediates produced via this resilient pathway, companies can guarantee consistent availability, strengthening their reputation as a dependable partner in the global pharmaceutical supply network.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden challenges related to heat transfer and mixing, but the mild exothermic nature of this reaction makes it inherently safer and easier to scale. The low operating temperature (50°C) reduces the risk of thermal runaway, a critical safety consideration for batch reactors of any size. Moreover, the environmental profile of the process is exemplary, as it avoids the generation of persistent organic pollutants or heavy metal waste streams that are difficult to remediate. This compliance with stringent environmental regulations facilitates faster permitting and approval for new production lines, accelerating time-to-market for new products. For supply chain heads, this means a future-proof manufacturing asset that can operate sustainably for years without requiring costly retrofits to meet evolving environmental standards.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical and commercial aspects of this technology, we have compiled a set of answers based on the detailed data provided in the patent documentation. These questions cover the critical parameters that influence reaction success, the scope of applicable substrates, and the safety profile of the resulting compounds. Understanding these details is essential for technical teams evaluating the feasibility of integrating this synthesis into their existing portfolios. The following responses provide clarity on how this method compares to industry standards and what specific benefits it offers for the production of high-value pharmaceutical intermediates.
Q: What are the key advantages of this cyanation method over traditional Sandmeyer reactions?
A: Unlike traditional Sandmeyer reactions which require harsh conditions and generate toxic waste, this novel method utilizes mild temperatures (50°C) and avoids toxic by-products, ensuring a safer and more environmentally friendly process suitable for large-scale production.
Q: What catalyst system provides the highest yield for this synthesis?
A: Experimental data indicates that the combination of cuprous chloride (CuCl) as the catalyst and 1,10-phenanthroline (Phen) as the additive in dichloromethane (DCM) solvent yields the best results, achieving up to 78% isolated yield.
Q: Can this method be applied to substrates with diverse substituents?
A: Yes, the method demonstrates excellent substrate scope, accommodating various substituents such as halogens, nitro groups, alkyl chains, and trifluoromethyl groups on the benzene rings, making it highly versatile for generating diverse pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acetonitrile Bis-Benzenesulfonylimine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN113072470A for the next generation of anti-inflammatory therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet rigorous quality specifications, supported by our state-of-the-art analytical capabilities and stringent QC labs. By partnering with us, you gain access to a supply chain that is not only robust and compliant but also optimized for cost and speed, allowing you to focus on your core competencies in drug development while we handle the complexities of chemical synthesis.
We invite you to engage with our technical procurement team to discuss how this specific technology can be tailored to your project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of this route for a specific analog, we are ready to provide the data-driven insights you need. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in Cu-catalyzed cyanation can accelerate your path to market with reliable, high-quality intermediates.
