Advanced Copper-Catalyzed Synthesis of N-Acetonitrile Bis-Benzenesulfonylimine Derivatives for Pharma
Advanced Copper-Catalyzed Synthesis of N-Acetonitrile Bis-Benzenesulfonylimine Derivatives for Pharma
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for complex intermediates, particularly those containing nitrile functionalities which are pivotal in drug design. A significant breakthrough in this domain is detailed in patent CN113072470A, which discloses a novel method for synthesizing N-acetonitrile bis-benzenesulfonylimine derivatives. These compounds, characterized by their unique structural motif featuring a central nitrogen atom bonded to two sulfonyl groups and an acetonitrile moiety, have demonstrated promising anti-inflammatory activity, making them highly valuable candidates for medicinal chemistry programs. The core innovation lies in the utilization of hydroxyalkenyl azides as efficient cyanating agents, reacting with N-fluorobis(benzenesulfonyl)imide under mild copper catalysis. This approach represents a paradigm shift from traditional, hazardous cyanation methods, offering a cleaner and more operationally simple pathway to access these critical scaffolds. As a leading entity in the chemical sector, understanding the nuances of such patented technologies is essential for maintaining a competitive edge in the supply of high-purity pharmaceutical intermediates.
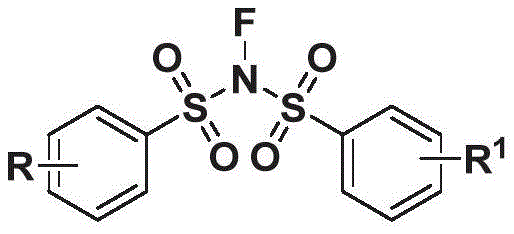
The structural versatility of these derivatives is defined by the substituents R and R1 on the phenyl rings, which can range from simple halogens and alkyl groups to more complex heterocyclic and ester functionalities. This tunability allows medicinal chemists to optimize the pharmacokinetic and pharmacodynamic properties of the final drug candidates. The ability to rapidly synthesize a diverse library of these analogues is crucial for structure-activity relationship (SAR) studies during the early stages of drug discovery. Furthermore, the presence of the sulfonylimine group often imparts specific electronic and steric properties that can enhance binding affinity to biological targets. By mastering the synthesis of this core structure, suppliers can provide research teams with the necessary tools to accelerate the development of next-generation anti-inflammatory therapeutics, addressing a significant unmet medical need in chronic disease management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a cyano group into organic molecules has relied on methodologies that are increasingly viewed as obsolete due to their severe safety and environmental drawbacks. Classic reactions such as the Sandmeyer reaction, developed in the late 19th century, require the generation of unstable diazonium salts and the use of stoichiometric amounts of copper salts, often leading to explosive hazards and difficult waste disposal issues. Similarly, the Rosenmund-von Braun reaction necessitates the use of copper(I) cyanide (CuCN), a highly toxic reagent that poses significant health risks to operators and requires stringent containment measures in a manufacturing setting. More modern transition-metal catalyzed cyanations, while an improvement, often rely on expensive palladium or iridium catalysts and toxic cyanide sources like potassium ferrocyanide or zinc cyanide. These traditional pathways are frequently plagued by harsh reaction conditions, including high temperatures and strong bases, which can lead to the decomposition of sensitive functional groups and the formation of complex impurity profiles that are difficult to purge. For a procurement manager or supply chain head, these factors translate into higher operational costs, increased regulatory burden, and potential disruptions in production continuity due to safety incidents or waste treatment bottlenecks.
The Novel Approach
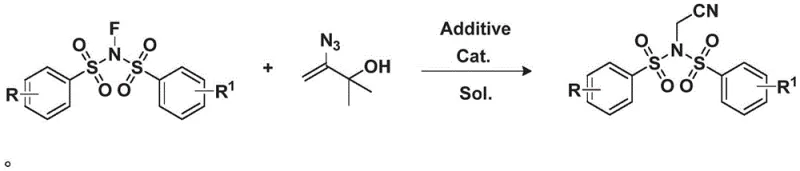
In stark contrast to these legacy methods, the technology described in CN113072470A introduces a remarkably mild and efficient strategy for constructing the nitrile functionality. The process utilizes 2-methyl-3-buten-2-ol derived hydroxyalkenyl azides as a safe and effective substitute for acetonitrile carbon sources. When coupled with N-fluorobis(benzenesulfonyl)imide in the presence of a copper catalyst and a specific additive, the reaction proceeds smoothly to yield the desired N-acetonitrile bis-benzenesulfonylimine derivatives. This novel route eliminates the need for free cyanide ions or toxic heavy metal cyanides, thereby drastically reducing the toxicity profile of the entire manufacturing process. The reaction conditions are exceptionally gentle, typically operating at temperatures around 50°C, which preserves the integrity of sensitive substrates and minimizes energy consumption. From a commercial perspective, this translates to a significantly simplified workflow that reduces the need for specialized equipment and extensive safety protocols, ultimately lowering the barrier to entry for large-scale production. The method's ability to generate high yields with minimal by-products ensures a cleaner crude reaction mixture, simplifying downstream purification and enhancing overall process efficiency.
Mechanistic Insights into Copper-Catalyzed Cyanation
The success of this transformation hinges on the synergistic interaction between the copper catalyst, the ligand additive, and the unique reactivity of the vinyl azide substrate. The proposed mechanism likely involves the activation of the hydroxyalkenyl azide by the copper center, facilitating the extrusion of nitrogen gas and the formation of a reactive copper-carbene or copper-nitrenoid species. This intermediate subsequently undergoes a rearrangement or insertion process that effectively transfers the cyano group to the electrophilic nitrogen of the N-fluorobis(benzenesulfonyl)imide. The use of 1,10-phenanthroline (Phen) as an additive plays a critical role in stabilizing the copper species and modulating its electronic properties, thereby enhancing both the conversion of the starting material and the selectivity towards the desired product. Experimental data from the patent indicates that substituting Phen with other additives like benzoic acid or trifluoroacetic acid results in significantly lower yields, underscoring the specificity of this ligand-catalyst system. For R&D directors, understanding this mechanistic nuance is vital for troubleshooting potential scale-up issues and for designing analogues that might require slight modifications to the catalytic cycle to accommodate different steric or electronic demands.
Furthermore, the choice of solvent and reaction temperature profoundly influences the reaction outcome, acting as key levers for impurity control. The patent highlights dichloromethane (DCM) as the preferred solvent, which provides an optimal balance of solubility for both organic substrates and the inorganic catalyst. Operating at a controlled temperature of 50°C ensures that the reaction kinetics are favorable without triggering thermal decomposition pathways that could lead to polymeric by-products or degradation of the azide functionality. This precise control over reaction parameters is essential for maintaining a consistent impurity profile, which is a critical quality attribute for pharmaceutical intermediates. By avoiding harsh conditions, the process minimizes the formation of genotoxic impurities or heavy metal residues that often plague traditional cyanation methods. This inherent cleanliness of the reaction not only simplifies the analytical burden but also aligns with the increasingly stringent regulatory requirements for drug substance manufacturing, providing a distinct advantage for companies aiming to bring these molecules to clinical trials.
How to Synthesize N-Acetonitrile Bis-Benzenesulfonylimine Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific procedural steps to maximize yield and purity. The general protocol involves dissolving the hydroxyalkenyl azide and the N-fluoro reagent in dry DCM, followed by the sequential addition of the copper catalyst and the phenanthroline ligand. The mixture is then heated to the specified temperature and monitored via TLC or HPLC until completion. While the general concept is straightforward, the devil is in the details regarding stoichiometry, addition rates, and workup procedures. To ensure reproducibility and safety, it is imperative to follow a standardized operating procedure that accounts for the specific handling requirements of azide-containing compounds. The following guide outlines the critical stages of the synthesis based on the optimized conditions disclosed in the patent examples.
- Dissolve hydroxyalkenyl azide and N-fluorobis(benzenesulfonyl)imide in dichloromethane (DCM) solvent under inert atmosphere.
- Add cuprous chloride (CuCl) catalyst and 1,10-phenanthroline (Phen) additive to the reaction mixture.
- Heat the reaction to 50°C for 16 hours, then perform aqueous workup and silica gel chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic methodology offers tangible benefits that extend beyond mere chemical curiosity. The primary advantage lies in the substantial cost reduction in pharmaceutical intermediate manufacturing driven by the elimination of expensive and hazardous reagents. Traditional cyanation processes often incur high costs associated with the purchase of precious metal catalysts, the disposal of toxic cyanide waste, and the implementation of rigorous safety measures. By switching to this copper-catalyzed vinyl azide pathway, manufacturers can bypass these cost centers entirely. The use of earth-abundant copper salts instead of palladium or iridium significantly lowers the raw material bill, while the absence of toxic cyanide sources reduces waste treatment expenses and regulatory compliance costs. Moreover, the mild reaction conditions reduce energy consumption, contributing to a lower carbon footprint and aligning with corporate sustainability goals. These cumulative savings can be passed down the supply chain, offering a more competitive pricing structure for the final active pharmaceutical ingredients (APIs) derived from these intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is bolstered by the use of readily available and inexpensive starting materials. 2-methyl-3-buten-2-ol derivatives and N-fluorobis(benzenesulfonyl)imide are commercially accessible at scale, ensuring a stable supply base that is not subject to the volatility often seen with specialty reagents. The catalyst loading is relatively low, and the catalyst itself is inexpensive, further driving down the cost of goods sold (COGS). Additionally, the simplified workup procedure, which involves standard extraction and chromatography, reduces the labor and time required for isolation. This efficiency translates directly into improved margins for manufacturers and more affordable options for downstream buyers. The avoidance of complex purification steps needed to remove heavy metal residues from precious metal catalysis also saves on consumables like scavengers and specialized filtration media.
- Enhanced Supply Chain Reliability: Supply chain resilience is a top priority for global pharmaceutical companies, and this synthetic route contributes significantly to that stability. The reagents involved are not subject to strict export controls or scarcity issues that often affect cyanide salts or precious metals. This ease of sourcing ensures that production schedules can be maintained without interruption due to raw material shortages. Furthermore, the robustness of the reaction conditions means that the process is less susceptible to variations in utility supply, such as fluctuations in cooling water temperature or steam pressure, which can sometimes derail sensitive chemical transformations. The ability to run the reaction at moderate temperatures also reduces the risk of thermal runaway incidents, enhancing plant safety and minimizing the likelihood of unplanned shutdowns. For supply chain heads, this reliability translates into predictable lead times and the ability to commit to long-term supply agreements with confidence.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the tonne scale is fraught with challenges, but this methodology is inherently designed for scalability. The exothermicity of the reaction is manageable, and the use of common solvents like DCM facilitates easy solvent recovery and recycling, which is crucial for large-scale operations. From an environmental standpoint, the process generates minimal hazardous waste, adhering to the principles of green chemistry. The absence of toxic cyanide by-products simplifies effluent treatment, reducing the load on wastewater treatment plants and ensuring compliance with increasingly strict environmental regulations. This environmental friendliness is not just a regulatory checkbox but a strategic asset, as it future-proofs the manufacturing process against tightening legislation. Companies that adopt such sustainable practices position themselves as preferred partners for major pharmaceutical clients who are under pressure to reduce the environmental impact of their supply chains.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical and commercial aspects of this technology, we have compiled a set of questions based on the patent data and industry standards. These answers provide clarity on the practical implementation of the synthesis and its implications for product quality and availability. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their development pipelines. The following responses are grounded in the specific experimental findings reported in the patent literature.
Q: What are the optimal reaction conditions for this cyanation process?
A: The patent specifies optimal conditions using CuCl (0.2 eq) and Phen (2.0 eq) in DCM at 50°C for 16 hours, achieving yields up to 78%.
Q: Does this method generate toxic by-products compared to traditional cyanation?
A: No, unlike traditional methods using toxic CuCN or heavy metal salts, this vinyl azide pathway generates no toxic side products, enhancing environmental compliance.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the mild temperature (50°C) and use of common solvents like DCM make the process highly scalable and safe for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acetonitrile Bis-Benzenesulfonylimine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methods described in CN113072470A for the production of high-value anti-inflammatory intermediates. Our team of expert chemists has extensively evaluated this copper-catalyzed pathway and confirmed its suitability for commercial scale-up. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global pharmaceutical partners. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of N-acetonitrile bis-benzenesulfonylimine derivatives we produce. We understand that consistency is key in drug development, and our robust quality management systems are designed to deliver materials that meet the highest industry standards, batch after batch.
We invite you to collaborate with us to leverage this advanced technology for your next project. Whether you are in the early stages of lead optimization or preparing for clinical trial material production, our technical procurement team is ready to assist. We encourage you to contact us for a Customized Cost-Saving Analysis that demonstrates how switching to our supply of these intermediates can optimize your overall manufacturing budget. Reach out today to request specific COA data and route feasibility assessments tailored to your specific molecular targets. Let us be your partner in navigating the complexities of modern pharmaceutical synthesis, delivering quality, reliability, and innovation in every shipment.
