Revolutionizing DMF Manufacturing with Stable N-Heterocyclic Carbene Catalysts for Global Supply Chains
Revolutionizing DMF Manufacturing with Stable N-Heterocyclic Carbene Catalysts for Global Supply Chains
The global demand for N,N-dimethylformamide (DMF) as a critical polar aprotic solvent continues to surge across the pharmaceutical, agrochemical, and polymer industries, driving an urgent need for more sustainable and efficient manufacturing technologies. Patent CN102190592B introduces a groundbreaking synthetic methodology that fundamentally shifts the paradigm from traditional metal-based catalysis to advanced organocatalysis using N-heterocyclic carbenes (NHC) and imidazolium salts. This technological leap addresses the chronic pain points of equipment fouling and environmental contamination that have plagued the industry for decades, offering a robust pathway for the production of high-purity formamide compounds. By leveraging these metal-free catalytic systems, manufacturers can achieve superior process stability and significantly reduce the operational complexities associated with inorganic salt byproducts. For R&D directors and procurement leaders, this innovation represents not just a chemical improvement, but a strategic asset for securing a resilient and cost-effective supply chain in the competitive fine chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The incumbent industrial standard for DMF synthesis relies heavily on the carbonylation of dimethylamine using carbon monoxide in the presence of a sodium methoxide catalyst dissolved in methanol. While this method historically offered low raw material costs, it suffers from a fatal flaw inherent to the chemistry of alkali metal bases in the presence of inevitable feedstock impurities. Trace amounts of carbon dioxide and water, which are unavoidable in industrial-grade carbon monoxide streams, react aggressively with the sodium methoxide to generate sodium formate, sodium bicarbonate, and sodium carbonate. These inorganic salts possess extremely low solubility in the organic DMF product matrix, leading to rapid crystallization and precipitation within the reaction system. Over time, these solid deposits accumulate on the inner walls of reactors and, more critically, clog the narrow channels of heat exchangers and connecting pipelines. This physical obstruction necessitates frequent and disruptive maintenance cycles, often requiring production shutdowns every forty days and intensive cleaning of heat exchange units every few days, resulting in substantial losses in productivity and increased labor intensity.

The Novel Approach
In stark contrast to the problematic legacy processes, the novel approach detailed in the patent utilizes stable nitrogen-heterocyclic carbenes or imidazolium salts as the primary catalytic drivers for the carbonylation reaction. These organic catalysts operate through a fundamentally different mechanistic pathway that completely bypasses the formation of inorganic metal salts, thereby eliminating the root cause of equipment blockage and crystallization. The absence of metal ions means that even in the presence of moisture or carbon dioxide impurities, the system remains homogeneous and free-flowing, allowing for uninterrupted continuous production runs that were previously impossible. Furthermore, these organocatalysts exhibit remarkable thermal stability and tunable electronic properties, enabling precise control over the reaction kinetics and selectivity without the environmental burden of heavy metal waste streams. This transition to metal-free catalysis not only solves the mechanical reliability issues of the plant but also aligns with increasingly stringent global environmental regulations regarding wastewater discharge and hazardous waste management.
Mechanistic Insights into NHC-Catalyzed Carbonylation
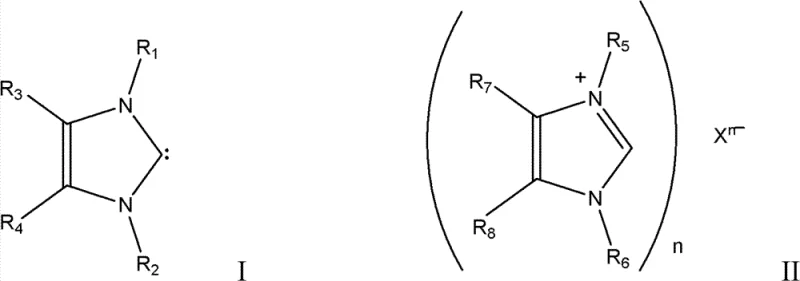
The efficacy of this new synthetic route lies in the unique electronic structure of the N-heterocyclic carbene (NHC) catalyst, which acts as a potent nucleophile capable of activating carbon monoxide under mild conditions. Unlike traditional bases that rely on ionic interactions prone to side reactions with acidic impurities, the NHC catalyst engages in a reversible coordination with the carbonyl species, facilitating its insertion into the amine bond with high specificity. The general structural framework of these catalysts, as depicted in the patent formulas, allows for extensive modular modification of the substituents (R groups) on the nitrogen atoms and the backbone. This modularity enables chemists to fine-tune the steric bulk and electron-donating capacity of the catalyst to optimize performance for specific amine substrates, ranging from simple aliphatic amines to complex aromatic derivatives. Such structural versatility ensures that the catalytic cycle remains robust even under the elevated pressures and temperatures required for industrial throughput, providing a level of control that inorganic catalysts simply cannot match.
From an impurity control perspective, the mechanism inherently suppresses the generation of non-volatile residues that typically contaminate the final product. In the conventional sodium methoxide process, the accumulation of sodium salts not only blocks pipes but also complicates the downstream purification steps, often requiring extensive washing and filtration that lower overall yield. With the NHC system, the reaction mixture remains largely free of solid particulates, meaning the crude product stream is significantly cleaner prior to distillation. This purity advantage translates directly into reduced energy consumption during the separation phase, as there is no need to manage slurry flows or dissolve stubborn salt cakes. Additionally, the patent highlights the potential for catalyst recovery; since the catalyst is an organic molecule with distinct solubility properties compared to the product, it can be separated and recycled through simple extraction and evaporation techniques, further enhancing the atom economy and sustainability profile of the entire manufacturing operation.
How to Synthesize N,N-Dimethylformamide Efficiently
Implementing this advanced catalytic technology requires a precise understanding of the operational parameters that maximize conversion while maintaining catalyst longevity. The patent outlines a scalable batch process where the amine substrate and the selected NHC or imidazolium catalyst are first dissolved in a compatible solvent system, such as methanol, ethanol, or even water, depending on the specific solubility requirements of the reagents. This homogenous mixture is then charged into a high-pressure autoclave reactor, setting the stage for the introduction of the carbon source. The flexibility in solvent choice is a significant advantage, allowing manufacturers to utilize greener solvents like water for certain applications, thereby reducing the volatile organic compound (VOC) footprint of the facility. Once the reactor is sealed, the system is pressurized with carbon monoxide, initiating the carbonylation sequence that transforms the amine into the desired formamide derivative with high efficiency.
- Prepare the reaction mixture by dissolving the amine compound (such as dimethylamine) and the NHC or imidazolium salt catalyst in a suitable solvent like methanol or water within a high-pressure autoclave.
- Introduce carbon monoxide gas into the reactor, maintaining a controlled pressure between 0.2 MPa and 8 MPa, preferably around 3 to 5 MPa for optimal conversion rates.
- Heat the reaction mixture to a temperature range of 50°C to 150°C while stirring vigorously, allowing the carbonylation to proceed until pressure stabilization indicates completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free catalytic technology offers profound strategic benefits that extend far beyond simple chemical yield improvements. The primary value proposition lies in the drastic simplification of plant maintenance schedules and the elimination of unplanned downtime caused by equipment fouling. In the traditional process, the relentless accumulation of sodium salts forces facilities into a cycle of frequent cleaning and eventual shutdown, creating bottlenecks that ripple through the entire supply network and jeopardize delivery commitments. By switching to the NHC-catalyzed route, manufacturers can achieve extended continuous operation cycles, effectively decoupling production capacity from maintenance constraints and ensuring a steady, reliable flow of finished goods to customers. This operational stability is critical for meeting the just-in-time delivery expectations of major pharmaceutical and electronics clients who cannot afford supply interruptions.
- Cost Reduction in Manufacturing: The economic impact of eliminating inorganic salt formation is multifaceted, touching upon both direct operational expenditures and capital efficiency. By preventing the crystallization of sodium formate and carbonates, the facility avoids the recurring costs associated with aggressive cleaning protocols, including the consumption of cleaning agents, water, and the labor hours required for manual intervention. Moreover, the preservation of heat exchanger efficiency ensures that energy transfer remains optimal throughout the production campaign, preventing the energy spikes that occur when equipment becomes insulated by salt deposits. Although the initial cost of the organic catalyst may differ from commodity sodium methoxide, the total cost of ownership is significantly lowered through reduced waste disposal fees, minimized product loss during cleaning, and the potential for catalyst recycling, creating a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the predictability of production outputs, a metric that is severely compromised by the variability of the traditional salt-forming process. The novel catalytic method provides a consistent and predictable reaction environment, removing the stochastic element of equipment blockages that can halt production without warning. This reliability allows supply chain planners to optimize inventory levels with greater confidence, reducing the need for excessive safety stock that ties up working capital. Furthermore, the ability to operate with a wider range of feedstock qualities, including carbon monoxide streams with higher impurity tolerances, reduces the risk of supply disruptions caused by raw material specification variances, making the entire value chain more robust against external shocks.
- Scalability and Environmental Compliance: As regulatory frameworks regarding industrial emissions and wastewater quality become increasingly rigorous, the environmental profile of a chemical process becomes a key license to operate. The metal-free nature of this synthesis eliminates the generation of saline wastewater, which is notoriously difficult and expensive to treat to meet discharge standards. This inherent cleanliness facilitates easier permitting for capacity expansions and reduces the liability associated with environmental compliance. From a scalability standpoint, the absence of solid handling issues means that scaling from pilot plants to multi-ton commercial reactors does not require complex engineering solutions for slurry management or solid removal, allowing for a smoother and faster scale-up trajectory that accelerates time-to-market for new products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic system, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this technology into existing infrastructure or new greenfield projects. The answers provided reflect the specific capabilities and limitations observed during the development of the NHC and imidazolium salt catalyzed processes, offering a realistic view of what can be achieved in a commercial setting.
Q: How does the NHC catalyst solve the blockage issues found in traditional sodium methoxide processes?
A: Traditional methods generate insoluble sodium salts (formate, carbonate) from CO2 impurities, which crystallize and block equipment. The metal-free NHC catalyst prevents salt formation entirely, ensuring smooth continuous flow.
Q: Can the N-heterocyclic carbene catalyst be recycled after the reaction?
A: Yes, the patent demonstrates that the catalyst residue can be recovered via extraction and drying, allowing for reuse in subsequent batches, which significantly lowers long-term catalyst costs.
Q: What are the typical reaction conditions for this metal-free DMF synthesis?
A: The process typically operates at moderate temperatures (100°C to 150°C) and pressures (3 MPa to 6 MPa) using solvents like methanol or water, offering a safer profile than extreme high-pressure alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-Dimethylformamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic technologies requires a partner with deep technical expertise and a proven track record in process optimization. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102190592B are fully realized in practical, large-scale operations. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of N,N-Dimethylformamide meets the exacting standards required by the global pharmaceutical and electronics industries. We are committed to leveraging our R&D capabilities to refine these metal-free synthesis routes, delivering products that are not only high in quality but also produced with a minimal environmental footprint.
We invite forward-thinking organizations to collaborate with us to explore how this innovative synthesis method can transform their supply chain dynamics and cost structures. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and sustainability in your chemical sourcing strategy.
