Scalable Manufacturing of Trifloxystrobin via Novel Non-Bromination Route
Scalable Manufacturing of Trifloxystrobin via Novel Non-Bromination Route
The global demand for high-efficiency fungicides continues to drive innovation in agrochemical intermediate manufacturing, particularly for broad-spectrum agents like Trifloxystrobin. A significant technological breakthrough in this domain is detailed in patent CN114685313A, which outlines a robust, five-step preparation method that fundamentally restructures the synthetic pathway to eliminate hazardous bromination steps. This novel approach utilizes 3-isochromanone as a starting material, leveraging a sequence of oximation, etherification, chlorination, esterification, and condensation reactions to achieve exceptional atom economy. By replacing traditional toxic reagents with safer alternatives like solid phosgene and methyl nitrite, the process not only enhances environmental compliance but also delivers a final product with purity exceeding 98 percent. For R&D directors and supply chain leaders, this represents a pivotal shift towards more sustainable and cost-effective production of critical crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
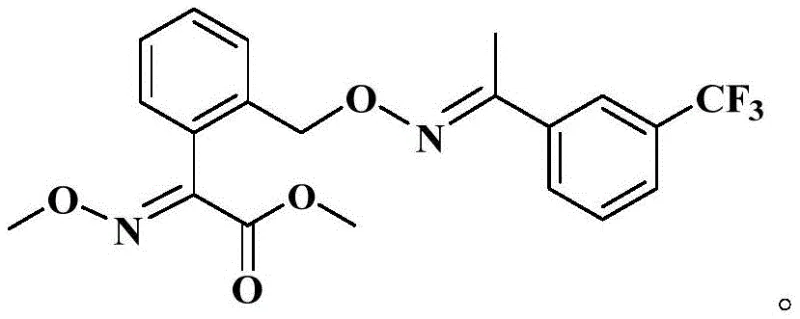
Historically, the industrial synthesis of Trifloxystrobin has been plagued by significant safety and efficiency bottlenecks, primarily stemming from reliance on hazardous halogenation strategies. As illustrated in prior art such as US5221762, traditional routes often commence with o-methylbenzoic acid, proceeding through cyanide-based intermediates and culminating in a bromination step to introduce the necessary side-chain functionality. This legacy approach suffers from inherently low yields and poor selectivity during the bromination phase, resulting in crude products that are notoriously difficult to purify. Furthermore, the use of virulent cyanide raw materials poses severe occupational health risks and necessitates expensive waste treatment protocols. The accumulation of brominated byproducts not only complicates downstream processing but also negatively impacts the content of the target molecule in the final condensation reaction, rendering the entire process economically inefficient and environmentally burdensome for large-scale operations.
The Novel Approach
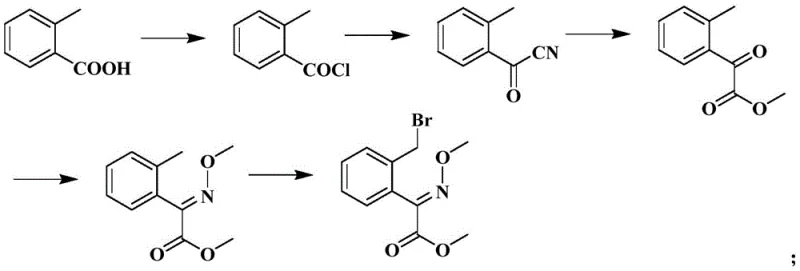
In stark contrast to these legacy methodologies, the process disclosed in CN114685313A introduces a paradigm shift by utilizing a ring-opening strategy that completely bypasses the need for direct bromination. By starting with 3-isochromanone, the synthesis leverages a highly active oximation reaction followed by a controlled etherification to build the core scaffold. The critical innovation lies in the subsequent chlorination step, where solid phosgene is employed to cleave the isochroman ring, simultaneously installing the acetyl chloride functionality required for esterification. This telescoped design allows for the direct conversion of intermediates without complex isolation procedures, drastically reducing solvent consumption and processing time. The result is a streamlined workflow that maintains mild reaction conditions throughout, ensuring high conversion rates while eliminating the generation of sulfur dioxide and hydrogen chloride gases typically associated with thionyl chloride reagents, thereby offering a cleaner and more commercially viable pathway for reliable agrochemical intermediate supplier networks.
Mechanistic Insights into Ring-Opening Chlorination and Condensation
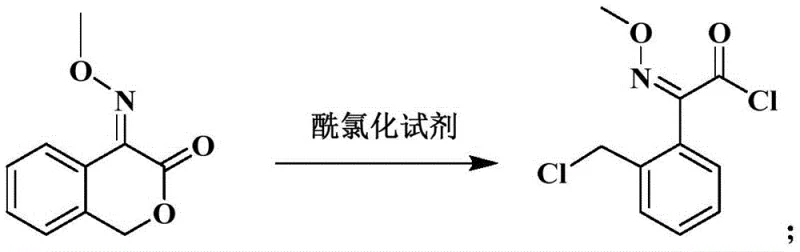
The core chemical ingenuity of this synthesis lies in the precise control of the ring-opening chlorination and the final condensation mechanism. In the third step, the reaction of 3-keto-4-(methoxyimino)isochroman with solid phosgene is catalyzed by an organic base, such as pyridine, to facilitate the nucleophilic attack on the carbonyl carbon. This results in the cleavage of the cyclic ether bond and the formation of 2-(2-(chloromethyl)phenyl)-2-(methoxyimino)acetyl chloride. The patent specifies a "reverse dropping" technique where the substrate is added to the phosgene mixture, a kinetic control measure that maximizes raw material conversion and minimizes side reactions. This is followed by an esterification with methanol under negative pressure, which efficiently removes generated hydrogen chloride, driving the equilibrium towards the formation of the methyl acetate intermediate with a purity of ≥97 percent. This high-purity intermediate is crucial for the success of the final step.
The final condensation reaction couples the methyl acetate intermediate with m-trifluoromethyl acetophenone oxime in the presence of a metal iodide catalyst, such as potassium iodide, and an acid-binding agent like powdered potassium carbonate. The iodide catalyst plays a pivotal role in enhancing the nucleophilicity of the oxime oxygen, overcoming the steric hindrance and low reactivity often observed in similar coupling reactions. Conducted in a polar aprotic solvent like DMF at moderate temperatures (45-55°C), this step achieves a yield of over 93 percent. The mechanistic efficiency here ensures that the stereochemistry is preserved and that the formation of geometric isomers is minimized, directly contributing to the high biological activity of the final fungicide. This level of mechanistic control is essential for achieving the stringent quality standards required in commercial scale-up of complex agrochemical intermediates.
How to Synthesize Trifloxystrobin Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing operational simplicity and safety. The process begins with the preparation of the oxime salt, followed by etherification in an autoclave to manage the pressure of monochloromethane. The subsequent chlorination and esterification steps are designed to be telescoped where possible, minimizing the handling of unstable intermediates. For detailed operational parameters, including specific molar ratios, temperature gradients, and stirring speeds required to replicate these high yields, operators should refer to the standardized technical documentation. The following guide summarizes the critical phases necessary to achieve the reported purity and yield benchmarks.
- Perform oximation on 3-isochromanone using organic metal alkoxide and methyl nitrite to form the nitrous acid metal salt.
- Conduct etherification with monochloromethane to obtain 3-ketone-4-(methoxyimino)isochroman.
- Execute chlorination using solid phosgene to open the ring and form the acetyl chloride intermediate.
- Carry out esterification with methanol under negative pressure to yield the methyl acetate intermediate.
- Finalize with condensation reaction using m-trifluoromethyl acetophenone oxime to obtain Trifloxystrobin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers profound strategic advantages beyond mere technical feasibility. The elimination of bromination reagents and toxic cyanides fundamentally alters the cost structure of manufacturing by removing the need for specialized hazardous waste disposal and expensive heavy metal scavenging processes. Furthermore, the use of readily available starting materials like 3-isochromanone and solid phosgene ensures a stable supply chain that is less susceptible to the regulatory volatility often associated with controlled brominating agents. The process's ability to operate under mild conditions also translates to lower energy consumption and reduced wear on reactor vessels, contributing to substantial cost savings in long-term production cycles.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the purification workflow. Traditional methods require multiple column chromatography separations and extensive washing steps to remove brominated impurities, which consume vast quantities of solvents and silica gel. In contrast, this new route achieves high intermediate purity (≥97%) through simple recrystallization and washing, significantly reducing solvent recovery costs. Additionally, the replacement of thionyl chloride with solid phosgene eliminates the generation of sulfur dioxide tail gas, removing the capital expenditure required for complex scrubbing systems. These factors combine to drastically lower the variable cost per kilogram of the active ingredient, making it a highly competitive option for cost reduction in agrochemical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals that are widely produced and easily sourced globally. Unlike specialized brominating agents which may face shipping restrictions or supply shortages, reagents such as methyl nitrite, methanol, and solid phosgene are standard industrial chemicals with robust logistics networks. The process also reduces the lead time for high-purity agrochemical intermediates by shortening the overall synthesis timeline; the telescoping of the first two steps and the elimination of complex intermediate isolations mean that production batches can be turned around much faster. This agility allows manufacturers to respond more quickly to seasonal demand spikes in the agricultural sector without compromising on quality or delivery schedules.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is inherently designed for green manufacturing. The avoidance of virulent cyanides and the reduction of three-waste (wastewater, waste gas, solid waste) generation align perfectly with increasingly stringent global environmental regulations. The high atom economy ensures that a greater proportion of raw materials end up in the final product rather than as waste, simplifying effluent treatment. This compliance advantage reduces the risk of regulatory shutdowns and fines, ensuring continuous operation. Moreover, the mild reaction temperatures and pressures make the process safer to scale from pilot plants to multi-ton reactors, facilitating the commercial scale-up of complex agrochemical intermediates with minimal engineering modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is critical for evaluating the feasibility of integrating this route into existing production facilities.
Q: Why is the new synthesis route superior to traditional bromination methods?
A: Traditional methods utilize toxic cyanide and bromination reagents which result in low yields, poor selectivity, and difficult purification. The novel route described in CN114685313A avoids bromination entirely, utilizing a ring-opening chlorination strategy that offers higher atom economy and significantly reduces hazardous waste generation.
Q: How does this process improve product purity and yield?
A: By employing solid phosgene instead of thionyl chloride, the process avoids the generation of sulfur dioxide and hydrogen chloride tail gases that complicate separation. The intermediate purity reaches ≥97%, leading to a final condensation yield of over 93% and a final product purity exceeding 98%.
Q: Is this manufacturing process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It features mild reaction conditions (mostly 10-60°C), eliminates the need for complex column chromatography separations, and utilizes a telescoped two-step initial sequence that simplifies operations and enhances safety for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifloxystrobin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN114685313A are fully realized in practice. Our facilities are equipped with state-of-the-art reactors capable of handling pressurized etherification and corrosive chlorination steps safely, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for agrochemical actives.
We invite you to collaborate with us to optimize your supply chain for Trifloxystrobin and related fungicide intermediates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this novel route can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of high-quality crop protection solutions.
