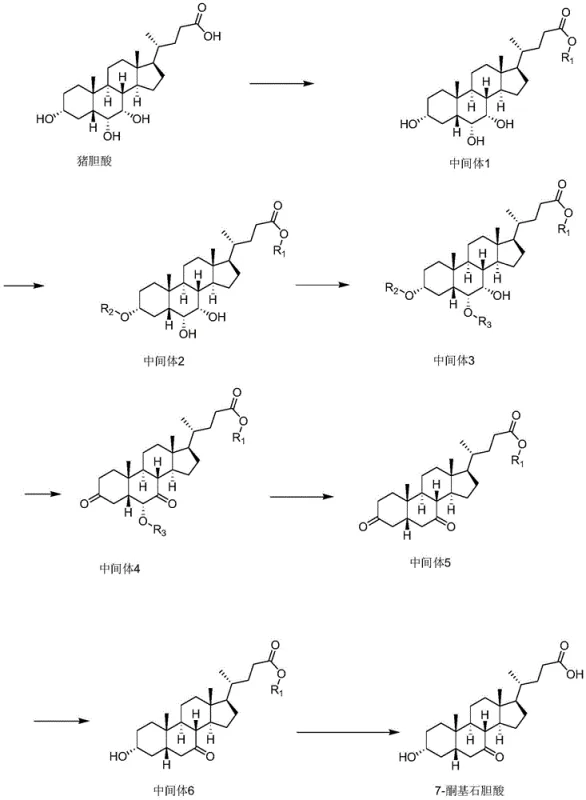
Revolutionizing Bile Acid Synthesis: A Novel Route to High-Purity 7-Ketolithocholic Acid Intermediates
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for synthesizing critical bile acid derivatives, and the technology disclosed in patent CN110423261B represents a significant breakthrough in this domain. This innovative preparation process focuses on the production of 7-ketolithocholic acid, a pivotal intermediate for manufacturing hepatobiliary drugs such as ursodeoxycholic acid and obeticholic acid. Traditionally, the reliance on chenodeoxycholic acid as a starting material has presented challenges regarding raw material availability and impurity profiles. However, this new methodology leverages hyocholic acid, a component often underutilized in pig bile processing, to create a streamlined synthetic route. By transforming a previously discarded resource into a high-value pharmaceutical intermediate, this technology not only addresses supply chain constraints but also aligns with green chemistry principles. The strategic shift from extraction-dependent methods to this sophisticated chemical synthesis ensures a more stable and scalable supply of high-purity 7-ketolithocholic acid for global drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 7-ketolithocholic acid has been heavily dependent on the extraction and oxidation of chenodeoxycholic acid from animal bile, a process fraught with inherent inefficiencies and supply volatility. The conventional chemical synthesis routes often suffer from poor selectivity during the oxidation stages, leading to the formation of unwanted 3-position oxidized impurities that are difficult to separate. Furthermore, the incomplete oxidation of chenodeoxycholic acid can leave behind raw material impurities that compromise the final product's quality, necessitating costly and time-consuming purification steps. These traditional methods also struggle with yield consistency, as reported yields in older literature can be as low as 38%, making them economically unviable for large-scale commercial production. The reliance on specific animal bile sources also introduces variability in raw material composition, which complicates quality control and regulatory compliance for pharmaceutical grade intermediates. Consequently, the industry has long needed a more robust and selective synthetic strategy to overcome these persistent bottlenecks.
The Novel Approach
The novel approach detailed in the patent data introduces a paradigm shift by utilizing hyocholic acid as the primary starting material, effectively turning a waste product into a valuable precursor for critical medications. This method employs a highly selective silane protecting group strategy that specifically targets the 3-hydroxyl group while leaving the 6 and 7-hydroxyl groups unaffected, a level of precision that significantly simplifies the overall synthetic sequence. By exploiting the unique spatial structure of hyocholic acid, the process allows for the selective connection of a strong leaving group at the 6-position without requiring complex or harsh operational conditions. This strategic manipulation of molecular geometry eliminates the need for multiple redundant protection and deprotection steps that typically plague steroid synthesis. The result is a drastically simplified reaction route that not only improves overall yield but also enhances the purity profile of the final 7-ketolithocholic acid product, making it an ideal candidate for modern pharmaceutical manufacturing standards.
Mechanistic Insights into Selective Silane Protection and Oxidation
The core of this technological advancement lies in the meticulous control of stereochemistry and functional group reactivity throughout the synthesis pathway. The process begins with the esterification of hyocholic acid, followed by the critical step of protecting the 3-hydroxyl group using a silane protecting group reagent such as trimethylchlorosilane or dimethyl tert-butylchlorosilane. This protection step exhibits exceptional selectivity, ensuring that the 3-position is shielded while the 6 and 7-hydroxyl groups remain available for subsequent transformations. This selectivity is crucial because it prevents the formation of byproducts that would otherwise arise from non-specific reactions at multiple hydroxyl sites. Following protection, the specific spatial arrangement of the 6,7-hydroxyl system is leveraged to introduce a strong leaving group, such as a methanesulfonyl or p-toluene sulfonyl group, exclusively at the 6-position. This precise functionalization sets the stage for the subsequent oxidation and elimination reactions that ultimately define the ketone structure at the 7-position.
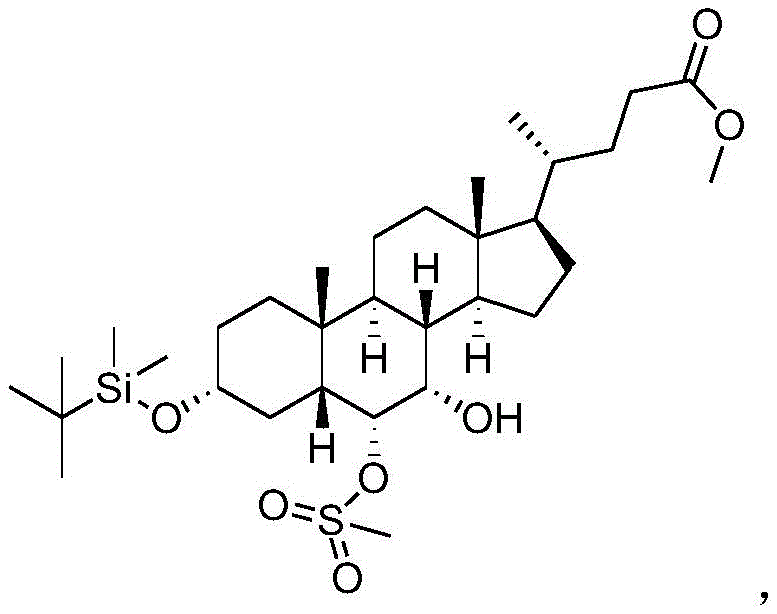
Impurity control is inherently built into the mechanism of this synthesis, primarily due to the avoidance of direct oxidation on the 3-position which is a common source of contamination in traditional routes. By starting with hyocholic acid and employing selective protection, the process bypasses the generation of 3-position oxidized impurities that are characteristic of chenodeoxycholic acid oxidation. The oxidation step itself, utilizing reagents like chromium trioxide or NBS, is performed on an intermediate where the 3-position is already secured, ensuring that the oxidative force is directed solely towards the intended target sites. Subsequent removal of the leaving group and reduction steps are conducted under mild conditions that minimize degradation or racemization of the steroid backbone. The final hydrolysis step removes the protecting groups to reveal the target 7-ketolithocholic acid with an HPLC detection purity exceeding 99.5%. This rigorous control over reaction pathways ensures that the impurity spectrum is tightly managed, meeting the stringent requirements of regulatory bodies for active pharmaceutical ingredients.
How to Synthesize 7-Ketolithocholic Acid Efficiently
The synthesis of this high-value intermediate follows a logical progression of esterification, selective protection, functionalization, and final conversion, designed for reproducibility and scale. The initial phase involves dissolving the pig bile acid in an alcohol solvent with an acid catalyst to form the ester intermediate, setting the foundation for subsequent selective reactions. Detailed standardized synthesis steps see the guide below.
- Perform esterification of hyocholic acid in alcohol with an acid catalyst to obtain Intermediate 1.
- Protect the 3-hydroxyl group selectively using a silane protecting group reagent to form Intermediate 2.
- Introduce a strong leaving group at the 6-position to generate the key Intermediate 3.
- Execute oxidation, removal of the leaving group, reduction, and final hydrolysis to yield 7-ketolithocholic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers profound strategic advantages that extend beyond simple chemical efficiency. The primary benefit stems from the utilization of hyocholic acid, a raw material that is currently underutilized and often discarded, which fundamentally alters the cost structure of the supply chain. By shifting demand to this abundant byproduct, manufacturers can mitigate the price volatility associated with traditional bile acid extracts like chenodeoxycholic acid. This transition not only secures a more stable raw material base but also aligns with sustainability goals by reducing waste in the animal byproduct processing industry. The simplified synthetic route further translates to reduced processing time and lower consumption of reagents and solvents, contributing to substantial cost savings in manufacturing operations without compromising on quality or yield.
- Cost Reduction in Manufacturing: The elimination of complex multi-step protection and deprotection sequences directly reduces the consumption of expensive reagents and solvents, leading to a leaner production cost model. By avoiding the need for harsh reaction conditions and specialized equipment required for traditional oxidation methods, capital expenditure and operational costs are significantly optimized. The high selectivity of the reaction minimizes the formation of byproducts, which reduces the burden on downstream purification processes and lowers waste disposal costs. Furthermore, the ability to use readily available and inexpensive starting materials like hyocholic acid ensures that the overall cost of goods sold remains competitive even in fluctuating market conditions. These cumulative efficiencies allow for a more aggressive pricing strategy while maintaining healthy profit margins for suppliers.
- Enhanced Supply Chain Reliability: Diversifying the raw material base to include hyocholic acid reduces dependency on single-source extracts that are subject to seasonal and geographical variations. The robustness of the chemical synthesis route ensures consistent production output regardless of fluctuations in natural bile availability, thereby stabilizing lead times for downstream pharmaceutical customers. The scalability of the process means that production volumes can be ramped up quickly to meet surges in demand for hepatobiliary medications without the long lead times associated with biological extraction methods. This reliability is critical for maintaining continuous manufacturing schedules for life-saving drugs and preventing stockouts in the global supply chain. Suppliers adopting this technology can offer more reliable delivery commitments, strengthening their partnerships with major pharmaceutical companies.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of standard organic solvents make this process highly amenable to scale-up from pilot plant to full commercial production without significant engineering hurdles. The reduction in hazardous waste generation, due to higher selectivity and fewer purification steps, simplifies compliance with increasingly stringent environmental regulations. The process avoids the use of extremely toxic or restricted reagents where possible, facilitating easier permitting and operation in diverse geographic locations. This environmental friendliness enhances the corporate social responsibility profile of the manufacturer, appealing to eco-conscious stakeholders and investors. Ultimately, the combination of scalability and compliance ensures long-term viability and reduces the risk of regulatory shutdowns or fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis technology. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation to ensure accuracy and relevance. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this intermediate into their supply chains.
Q: What is the primary advantage of using hyocholic acid over chenodeoxycholic acid?
A: Hyocholic acid is often a discarded byproduct in pig bile processing, making it a cost-effective raw material compared to chenodeoxycholic acid. Additionally, this route avoids 3-position oxidized impurities common in traditional methods.
Q: How does the new process ensure high purity levels?
A: The process utilizes highly selective silane protection at the 3-position and specific spatial structure utilization at the 6,7-positions, minimizing side reactions and achieving HPLC purity greater than 99.5%.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the reaction conditions are mild, reagents are commercially available, and the steps avoid harsh operations, making it highly suitable for commercial scale-up of complex steroid intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Ketolithocholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthesis route and are fully equipped to bring this technology to commercial reality for our global partners. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are designed to handle complex steroid chemistry with stringent purity specifications, supported by rigorous QC labs that verify every batch against the highest industry standards. We understand that the transition to a new synthetic route requires confidence in the partner's capability, and our track record in process optimization and scale-up serves as a testament to our commitment to excellence. By leveraging our infrastructure, you can accelerate the time-to-market for your bile acid-based therapeutics while maintaining the highest levels of quality assurance.
We invite you to engage with our technical procurement team to discuss how this innovative process can be tailored to your specific project requirements. Request a Customized Cost-Saving Analysis to understand the economic impact of switching to this hyocholic acid-based route for your manufacturing needs. Our team is ready to provide specific COA data and route feasibility assessments to demonstrate the viability and advantages of this technology for your supply chain. Let us collaborate to optimize your production of high-purity 7-ketolithocholic acid and drive value across your entire pharmaceutical portfolio.
