Scalable Synthesis of Fingolimod Key Intermediates via Optimized Suzuki Coupling for Commercial API Production
Introduction to Next-Generation Fingolimod Intermediate Manufacturing
The pharmaceutical landscape for immunosuppressants has been revolutionized by Fingolimod (FTY720), the first oral therapy approved for relapsing-remitting multiple sclerosis. As global demand for this critical active pharmaceutical ingredient (API) continues to surge, the efficiency of its supply chain relies heavily on the robustness of its key intermediate synthesis. Patent CN103145689A introduces a groundbreaking five-step synthetic methodology that addresses the longstanding bottlenecks in producing Compound 8, the pivotal precursor to Fingolimod. This innovative approach leverages a convergent strategy combining Wittig olefination, catalytic hydrogenation, and Suzuki-Miyaura cross-coupling to achieve an overall yield of 36.6%, demonstrating superior efficiency compared to historical benchmarks. For R&D directors and procurement strategists, this patent represents a vital opportunity to optimize the commercial scale-up of complex pharmaceutical intermediates while adhering to stricter environmental regulations. By shifting away from stoichiometric Lewis acid reactions toward catalytic cycles, this technology offers a pathway to cost reduction in API manufacturing that is both scientifically sound and economically viable for high-volume production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Fingolimod intermediates has been plagued by inefficient routes that impose severe burdens on both cost and environmental compliance. One prominent prior art method, illustrated in the reaction scheme below, initiates with toluene and relies heavily on Friedel-Crafts acylation to construct the carbon skeleton. This traditional approach necessitates the use of stoichiometric amounts of aluminum chloride or similar Lewis acids, resulting in the generation of massive quantities of corrosive aluminum salt waste that requires expensive neutralization and disposal protocols. Furthermore, this route suffers from low overall yields due to the harsh reaction conditions which often promote side reactions and decomposition of sensitive functional groups. The reliance on such archaic chemistry not only inflates the cost of goods sold (COGS) but also creates significant supply chain risks associated with waste management regulatory compliance. Consequently, manufacturers relying on these legacy processes face diminishing margins and increased operational complexity, making them less competitive in the global reliable pharmaceutical intermediate supplier market.
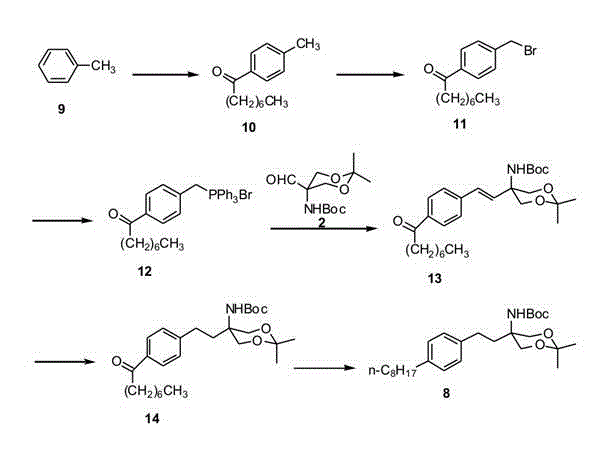
The Novel Approach
In stark contrast to the pollution-intensive legacy methods, the novel pathway disclosed in CN103145689A utilizes a modern organometallic strategy that prioritizes atom economy and operational simplicity. By employing a Wittig reaction to establish the initial carbon-carbon double bond followed by a highly selective Suzuki coupling to attach the alkyl chain, this method completely bypasses the need for Friedel-Crafts reagents. The use of palladium catalysis allows for milder reaction conditions, typically operating between 70°C and 120°C, which preserves the integrity of the protecting groups and minimizes the formation of difficult-to-remove impurities. This strategic shift from stoichiometric reagents to catalytic cycles fundamentally alters the economic model of production, enabling high-purity pharmaceutical intermediates to be generated with significantly reduced downstream purification requirements. The result is a streamlined process that is not only chemically elegant but also inherently safer and more scalable for industrial applications, directly addressing the pain points of modern API manufacturing.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling and Radical Boronation
The core of this technological advancement lies in the sophisticated application of palladium-catalyzed cross-coupling mechanics, specifically the Suzuki-Miyaura reaction, which serves as the convergence point for the two major molecular fragments. The mechanism begins with the oxidative addition of the aryl iodide (Compound 6) to the Pd(0) species, forming a reactive aryl-palladium complex. This intermediate then undergoes transmetallation with the organoboron species (Compound 5), which is generated in situ via a radical-mediated boronation process involving bis(pinacolato)diboron, benzoyl peroxide (BPO), and tert-butyl nitrite. This radical boronation step is particularly noteworthy as it allows for the direct functionalization of the benzylic position under mild conditions (10-50°C), avoiding the need for cryogenic temperatures or hazardous lithiation reagents. The subsequent reductive elimination releases the coupled product (Compound 7) and regenerates the active Pd(0) catalyst, closing the catalytic cycle. This mechanistic elegance ensures high turnover numbers and minimizes the residual palladium content in the final product, a critical parameter for meeting stringent stringent purity specifications required by regulatory bodies.
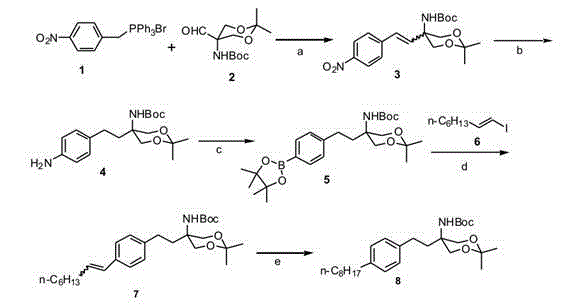
Impurity control is another critical aspect where this mechanism excels, particularly regarding the stereochemistry of the olefinic intermediates. The initial Wittig reaction produces a mixture of cis and trans isomers (Compound 3), which could potentially complicate downstream processing. However, the subsequent hydrogenation step using Pd/C effectively saturates the double bond, erasing the stereochemical distinction and converging both isomers into the single saturated intermediate (Compound 4). This 'stereo-convergence' is a powerful tool for yield maximization, as it prevents the loss of half the material due to isomer separation. Furthermore, the use of heterogeneous Pd/C catalysts in the hydrogenation steps facilitates easy filtration and removal of the catalyst, preventing metal contamination in the final API. This robust control over the impurity profile ensures that the final high-purity pharmaceutical intermediates meet the rigorous quality standards necessary for clinical and commercial use, thereby reducing the risk of batch rejection and ensuring supply continuity.
How to Synthesize Fingolimod Intermediate Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize the efficiency of each transformation, particularly the sensitive boronation and coupling steps. The process begins with the condensation of the phosphonium salt and aldehyde under basic conditions, followed by a careful workup to remove phosphine oxide byproducts. The subsequent hydrogenation and boronation steps must be monitored to prevent over-reaction or decomposition of the boronate ester, which can be sensitive to hydrolysis. Finally, the Suzuki coupling demands anhydrous conditions and rigorous degassing to maintain the activity of the palladium catalyst. For a detailed breakdown of the specific reagent ratios, solvent systems, and temperature profiles required to replicate this high-yielding process, please refer to the standardized technical guide below.
- Perform Wittig reaction between compound 1 and compound 2 using inorganic bases like K2CO3 in THF/DMF solvent mixture at 70-150°C to generate olefin compound 3.
- Execute catalytic hydrogenation of compound 3 using Pd/C to obtain compound 4, followed by radical boronation with bis(pinacolato)diboron, BPO, and t-BuONO to yield boronate ester compound 5.
- Conduct Suzuki coupling between compound 5 and iodide 6 using palladium catalysts and base, followed by final hydrogenation of compound 7 to afford the target intermediate 8.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers transformative benefits for procurement managers and supply chain directors seeking to optimize their sourcing strategies for Fingolimod precursors. The elimination of expensive and hazardous reagents, such as the phosphonates and azides required in alternative routes, results in a substantial reduction in raw material expenditure. Moreover, the simplified workflow reduces the number of unit operations and isolation steps, which directly correlates to lower labor costs and reduced consumption of solvents and energy. This efficiency translates into a more resilient supply chain capable of withstanding market fluctuations in raw material pricing. By partnering with a manufacturer utilizing this technology, clients can secure a more stable supply of critical intermediates, mitigating the risk of production delays caused by reagent shortages or complex waste disposal bottlenecks.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the substitution of high-cost specialty reagents with commodity chemicals. Traditional routes often rely on custom-synthesized building blocks that carry significant price premiums due to low production volumes and complex preparation. In contrast, this method utilizes widely available starting materials and catalytic amounts of palladium, which can often be recovered or recycled in industrial settings. Additionally, the avoidance of aluminum salt waste eliminates the hidden costs associated with hazardous waste treatment and environmental compliance fees. These factors combine to create a significantly leaner cost structure, allowing for more competitive pricing models without sacrificing margin, ultimately delivering substantial cost savings to the end customer.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability and stability of raw materials. The reagents employed in this novel synthesis, such as pinacolborane derivatives and common aryl iodides, are produced on a multi-ton scale by numerous global suppliers, ensuring a diversified and secure sourcing base. Unlike routes dependent on niche reagents that may be subject to single-source supply risks, this method leverages a robust chemical ecosystem. Furthermore, the operational simplicity of the process—characterized by ambient pressure hydrogenations and standard heating protocols—reduces the likelihood of equipment failure or process upsets. This reliability ensures consistent lead times and enables manufacturers to maintain healthy inventory levels, providing clients with the confidence of uninterrupted supply for their API production schedules.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden challenges related to heat transfer and mixing, but this route is inherently designed for scalability. The exothermic nature of the reactions is manageable within standard reactor configurations, and the use of heterogeneous catalysts simplifies the separation process, avoiding the need for complex distillation or chromatography on a large scale. From an environmental standpoint, the reduction in toxic waste streams aligns with the growing global emphasis on green chemistry and sustainable manufacturing. By minimizing the generation of heavy metal waste and volatile organic compounds, this process facilitates easier permitting and regulatory approval in jurisdictions with strict environmental laws. This compliance advantage future-proofs the supply chain against tightening regulations, ensuring long-term viability and commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its potential impact on their production pipelines. The following questions address common inquiries regarding the practical implementation, cost implications, and quality attributes of this patented method. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making. Whether you are concerned about impurity profiles or the feasibility of technology transfer, the answers below clarify how this innovation stands apart from conventional methodologies.
Q: How does this new synthesis route compare to traditional Friedel-Crafts methods in terms of environmental impact?
A: Unlike conventional routes utilizing Friedel-Crafts acylation which generate significant aluminum salt waste and require complex disposal, this patented method employs a Wittig reaction and Suzuki coupling sequence that eliminates heavy metal salt byproducts, drastically reducing environmental pollution and waste treatment costs.
Q: What are the primary cost drivers eliminated in this manufacturing process?
A: This process avoids the use of expensive reagents such as dimethyl (2-oxypropyl)phosphonate and p-toluenesulfonyl azide found in prior art. By utilizing commercially available and cheaper starting materials alongside efficient palladium catalysis, the overall raw material cost is substantially lowered without compromising yield.
Q: Is this synthetic pathway suitable for large-scale industrial production?
A: Yes, the methodology is specifically designed for industrial scalability. It features simple operational conditions, mild temperatures ranging from 0°C to 150°C, and robust catalytic systems like Pd/C that are easily filtered and managed, ensuring consistent quality and safety during commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fingolimod Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this synthesis are fully realized in practice. Our state-of-the-art facilities are equipped with advanced reactors capable of handling the specific thermal and pressure requirements of hydrogenation and coupling reactions, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards. We understand that in the pharmaceutical industry, consistency is key, and our robust quality management systems are designed to deliver that reliability batch after batch.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced synthetic technology for their Fingolimod supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this route for your specific volume requirements. We encourage you to reach out today to discuss your project needs,索取 specific COA data, and review our comprehensive route feasibility assessments. Let us help you optimize your API manufacturing strategy with a solution that balances cost, quality, and sustainability.
