Advanced Manufacturing Route for High-Purity Fingolimod Pharmaceutical Intermediates
Advanced Manufacturing Route for High-Purity Fingolimod Pharmaceutical Intermediates
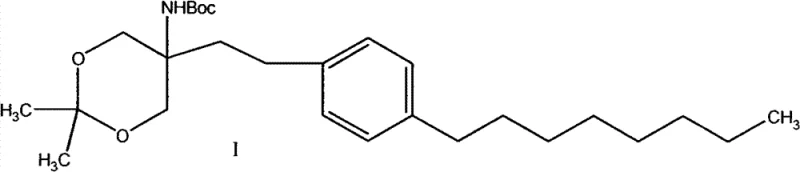
The pharmaceutical landscape for multiple sclerosis treatments has been significantly shaped by the approval of Fingolimod, necessitating robust and scalable supply chains for its critical precursors. Patent CN102850319A introduces a transformative preparation method for {5-[2-(4-n-octylphenyl)ethyl]-2,2-dimethyl-1,3-dioxane-5-yl} carbamic acid tert-butyl ester, a pivotal intermediate in the synthesis of this blockbuster drug. This technical disclosure addresses long-standing inefficiencies in prior art by establishing a route that prioritizes cost-effectiveness, operational safety, and high total yield. By shifting away from precious metal catalysts and hazardous solvents, this methodology offers a compelling value proposition for generic drug manufacturers and active pharmaceutical ingredient (API) producers seeking to optimize their production economics. The strategic implementation of iron-catalyzed cross-coupling and classical Wittig olefination ensures that the process remains accessible while maintaining the stringent purity standards required for regulatory approval.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of this specific dioxane derivative was plagued by significant economic and safety hurdles that hindered industrial scalability. Existing literature, such as the work by Kim et al., relied heavily on 4-n-octyl iodobenzene as a starting material, a reagent that is not only prohibitively expensive but also difficult to source in bulk quantities suitable for commercial manufacturing. Furthermore, these legacy processes often incorporated hazardous azide chemistry, introducing severe safety risks related to potential explosivity and toxicity that require specialized containment infrastructure. Another critical bottleneck identified in earlier methods, specifically those described by Balasubramaniam, was the reliance on carbon tetrachloride, a solvent now heavily restricted due to its ozone-depleting potential and carcinogenic properties. Additionally, the widespread use of palladium-catalyzed cross-coupling in traditional routes imposed a substantial financial burden due to the high cost of the metal and the complex downstream processing required to remove trace heavy metal residues to meet pharmacopeial limits.
The Novel Approach
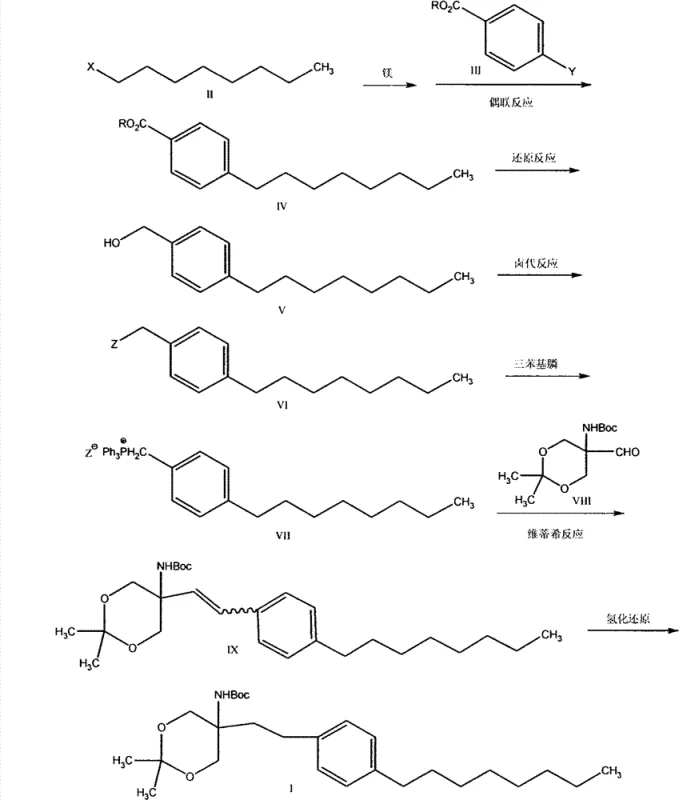
The methodology outlined in patent CN102850319A fundamentally reengineers the synthetic pathway to overcome these specific bottlenecks through clever retrosynthetic analysis and reagent selection. Instead of expensive aryl halides, the process initiates with readily available and low-cost 1-halo-n-octane, which is converted into a Grignard reagent for subsequent coupling. This shift allows for the use of iron catalysis rather than palladium, drastically reducing the catalyst cost profile and simplifying the purification workflow. The route elegantly constructs the carbon skeleton through a sequence of reduction, halogenation, and phosphonium salt formation, culminating in a Wittig reaction that efficiently installs the necessary two-carbon linker. By avoiding toxic solvents like carbon tetrachloride and dangerous azide intermediates, this approach aligns perfectly with modern green chemistry principles, thereby reducing the environmental footprint and regulatory compliance costs associated with waste disposal and worker safety monitoring.
Mechanistic Insights into Iron-Catalyzed Cross-Coupling and Wittig Olefination
The cornerstone of this cost-reduction strategy lies in the initial cross-coupling reaction, where an iron catalyst facilitates the union of the Grignard reagent derived from 1-halo-n-octane and a 4-halo-benzoate. Unlike palladium cycles which involve expensive oxidative addition and reductive elimination steps requiring ligand stabilization, the iron-catalyzed mechanism operates through a more economical redox cycle that tolerates a broader range of functional groups without the need for rigorous exclusion of air in some variations, although inert atmosphere is preferred for consistency. This step effectively builds the lipophilic octyl chain onto the aromatic core with high fidelity, as evidenced by the reported yields exceeding 86 percent in experimental examples. The subsequent reduction of the ester moiety to the benzyl alcohol utilizes a sodium borohydride and lithium chloride system, a modification that enhances the reducing power of the borohydride species, allowing for milder conditions compared to traditional lithium aluminum hydride reductions which pose greater safety risks on a large scale.
Following the conversion of the alcohol to a benzyl halide, the synthesis employs a classic Wittig reaction to couple the lipophilic tail with the protected amino-dioxane headgroup. The generation of the phosphonium ylide from the benzyl halide and triphenylphosphine is a robust transformation that proceeds with high conversion rates. The reaction with the aldehyde component, (5-formyl-2,2-dimethyl-1,3-dioxane-5-yl) carbamic acid tert-butyl ester, forms the alkene intermediate with defined stereochemistry, although the subsequent hydrogenation step renders the cis/trans ratio less critical for the final saturated product. The final catalytic hydrogenation using palladium on carbon is a standard yet highly effective method for saturating the double bond, ensuring the removal of any olefinic impurities and delivering the target molecule with exceptional purity. This mechanistic sequence ensures that impurities are either volatile or easily separable via crystallization or extraction, minimizing the need for complex chromatographic purifications that drive up manufacturing costs.
How to Synthesize Fingolimod Intermediate Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and minimize byproduct formation, particularly during the Grignard formation and the Wittig olefination steps. The process begins with the careful initiation of the magnesium turnings to ensure complete consumption of the alkyl halide, followed by the controlled addition to the iron-catalyst mixture to manage exotherms. Detailed standard operating procedures regarding temperature gradients, stoichiometry, and quenching protocols are essential for reproducibility at the multi-kilogram scale. For a comprehensive breakdown of the specific molar ratios, solvent volumes, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Prepare Grignard reagent from 1-halo-n-octane and react with 4-halo-benzoate via iron-catalyzed cross-coupling to form the ester.
- Reduce the ester to 4-n-octylbenzyl alcohol using sodium borohydride and lithium chloride.
- Convert the alcohol to a benzyl halide, react with triphenylphosphine to form the phosphonium salt, and perform a Wittig reaction with the protected aldehyde.
- Hydrogenate the resulting alkene intermediate using palladium carbon to obtain the final saturated target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this patented route offers a distinct competitive advantage by decoupling production costs from the volatile pricing of precious metals and specialty aryl halides. The substitution of palladium with iron represents a direct and significant reduction in raw material expenditure, as iron salts are orders of magnitude cheaper and more abundant than palladium complexes. Furthermore, the elimination of azide chemistry removes the need for specialized safety equipment and explosive handling certifications, which translates into lower overhead costs for facility maintenance and insurance. The use of commodity chemicals like 1-bromo-n-octane and methyl p-chlorobenzoate ensures a stable supply chain that is less susceptible to the shortages often experienced with custom-synthesized building blocks. This stability allows for better long-term planning and inventory management, securing the continuity of supply for downstream API manufacturing.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with inexpensive iron catalysts fundamentally alters the cost structure of the synthesis, removing the need for costly metal scavenging resins often required to meet strict residual metal specifications in pharmaceuticals. Additionally, the high yields observed in the reduction and hydrogenation steps, reaching up to 99 percent in some cases, minimize material loss and reduce the cost of goods sold per kilogram of finished intermediate. The avoidance of complex purification techniques like preparative HPLC, relying instead on standard extractions and crystallizations, further drives down operational expenses and solvent consumption.
- Enhanced Supply Chain Reliability: By utilizing starting materials that are widely produced for other industrial applications, such as long-chain alkyl halides and simple benzoates, the risk of supply disruption is significantly mitigated compared to routes relying on niche reagents. The robustness of the chemical transformations, which tolerate minor variations in reaction conditions without catastrophic failure, ensures consistent batch-to-batch quality. This reliability is crucial for maintaining Just-In-Time inventory systems and meeting the rigorous delivery schedules demanded by global pharmaceutical clients who cannot afford production delays.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, avoiding exothermic runaways and hazardous reagents that limit reactor size. The removal of carbon tetrachloride and azides simplifies the environmental permitting process and reduces the cost of waste treatment, as the effluent streams are less toxic and easier to remediate. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the corporate social responsibility profile of the supply chain, a factor increasingly weighted in vendor selection criteria by major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the comparative data and experimental results presented in the patent documentation. These insights are intended to clarify the operational benefits and technical feasibility for potential partners evaluating this technology for their own production lines. Understanding these nuances is critical for making informed decisions about technology transfer and process validation.
Q: Why is this new synthesis route preferred over previous methods reported by Kim et al.?
A: Previous methods relied on expensive 4-n-octyl iodobenzene and dangerous azides, alongside costly palladium catalysts. This new route utilizes inexpensive 1-halo-n-octane and iron catalysis, significantly lowering raw material costs and improving safety profiles for large-scale production.
Q: How does this process address environmental and safety concerns found in earlier literature?
A: Earlier protocols, such as those by Balasubramaniam, utilized highly toxic carbon tetrachloride as a solvent. The patented method eliminates this hazard by employing greener solvents like tetrahydrofuran and toluene, ensuring better compliance with modern environmental regulations and worker safety standards.
Q: What are the yield expectations for the key steps in this manufacturing process?
A: The patent demonstrates robust yields across the sequence, with the initial cross-coupling achieving approximately 86.7%, the reduction step reaching 95%, and the final hydrogenation step yielding up to 99%, indicating a highly efficient and commercially viable pathway.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fingolimod Intermediate Supplier
The technical superiority of this synthesis route positions it as an ideal candidate for commercial scale-up, and NINGBO INNO PHARMCHEM stands ready to leverage this innovation for your supply chain needs. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every batch of {5-[2-(4-n-octylphenyl)ethyl]-2,2-dimethyl-1,3-dioxane-5-yl} carbamic acid tert-butyl ester meets the highest international standards for pharmaceutical intermediates.
We invite you to engage with our technical procurement team to discuss how this optimized route can specifically benefit your project timelines and budget constraints. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this greener, more efficient methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique volume requirements, ensuring a partnership built on transparency, quality, and mutual growth.
