Advanced Recyclable Ligand Technology for Scalable Paclitaxel and Docetaxel Manufacturing
Advanced Recyclable Ligand Technology for Scalable Paclitaxel and Docetaxel Manufacturing
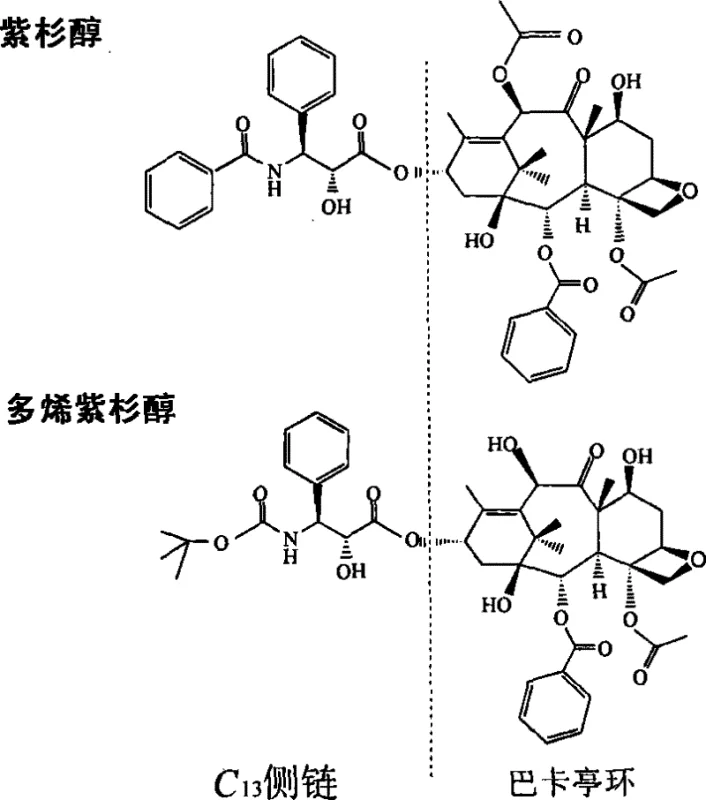
The global demand for potent anticancer agents continues to surge, placing immense pressure on the supply chains of critical pharmaceutical intermediates. Patent CN101029049B introduces a transformative approach to synthesizing the C13 side chains of paclitaxel and docetaxel, two of the most widely used chemotherapy drugs in oncology. This technology leverages recyclable and reusable cinchona alkaloid derivative ligands, addressing the longstanding economic and ecological challenges associated with traditional semi-synthetic routes. By shifting from single-use expensive catalysts to a recoverable system derived from abundant quinine, this innovation offers a robust pathway for reliable pharmaceutical intermediates supplier networks to enhance efficiency. The core breakthrough lies in the molecular design of the ligand, which balances high catalytic activity with facile recovery mechanisms, ensuring that the production of these life-saving drugs remains both economically viable and environmentally sustainable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of paclitaxel and docetaxel has been fraught with significant bottlenecks that hinder cost reduction in API manufacturing. The primary source of paclitaxel was initially the extraction from the bark of the Pacific Yew tree, a process that is ecologically devastating due to the low natural content of approximately 0.006% and the destruction of rare forest resources. While semi-synthesis from baccatin III extracted from yew needles alleviated some ecological pressure, the synthesis of the crucial C13 side chain remained a costly hurdle. Conventional asymmetric catalytic methods typically rely on precious metal catalysts paired with expensive chiral ligands that cannot be recovered. These ligands are consumed in stoichiometric or near-stoichiometric amounts, leading to exorbitant raw material costs and complex waste streams. Furthermore, the inability to recycle these high-value chiral auxiliaries means that every batch of production incurs the full cost of fresh ligand synthesis, creating a volatile cost structure that is sensitive to fluctuations in the supply of rare chemical precursors.
The Novel Approach
The methodology outlined in the patent data presents a paradigm shift by utilizing cinchona alkaloid derivatives that are specifically engineered for recovery and reuse. Instead of relying on non-recoverable systems, this approach starts with quinine, a cheap and readily available natural alkaloid, and modifies it through substitution and oxidation reactions to create a specialized chiral ligand. The genius of this design is the incorporation of polar functional groups, such as hydroxyl or sulfonyl groups, into the ligand structure. These groups ensure the ligand is soluble in the reaction medium during the asymmetric dihydroxylation (AD) step, facilitating high catalytic activity and excellent stereocontrol. However, upon completion of the reaction, the addition of a non-polar solvent induces the precipitation of the ligand, allowing it to be separated via simple filtration. This 'homogeneous reaction, two-phase separation' strategy effectively decouples the high performance of homogeneous catalysis from the ease of heterogeneous recovery, significantly reducing the synthesis cost of the C13 side chain while maintaining rigorous quality standards.
Mechanistic Insights into Asymmetric Dihydroxylation with Recyclable Ligands
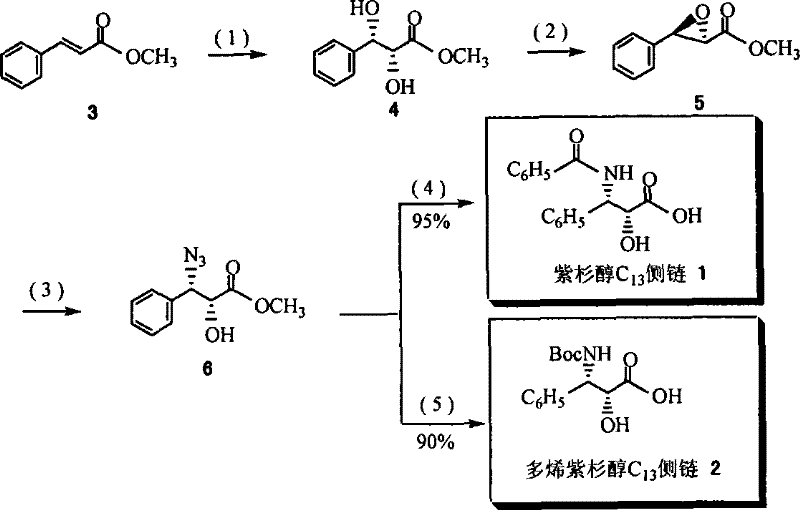
The heart of this synthetic route is the asymmetric dihydroxylation (AD) of olefins, specifically transforming methyl cinnamate into the chiral diol precursor required for the C13 side chain. The general structural formula (I) of the ligand plays a pivotal role in directing the stereochemistry of this transformation. The ligand coordinates with the osmium tetroxide catalyst to form a chiral environment that favors the formation of one enantiomer over the other with high precision. The presence of the quinoline ring and the quinuclidine nitrogen in the cinchona backbone provides the necessary steric bulk and electronic properties to differentiate between the prochiral faces of the alkene substrate. Crucially, the variable R2 group, which can be a hydroxyl, sulfide, or sulfonyl moiety, dictates the solubility profile. During the reaction in polar solvents like acetone-water mixtures, the ligand remains in solution, ensuring maximum interaction with the catalyst and substrate. This solubility is essential for achieving the high turnover numbers and enantiomeric excess values observed, often exceeding 99% ee in the initial cycles. The mechanism ensures that the chirality is transferred efficiently from the ligand to the product, minimizing the formation of unwanted isomers that would require difficult and yield-reducing purification steps downstream.
Impurity control is inherently built into this mechanistic design through the recovery phase. In traditional processes, trace metals and ligand residues often contaminate the final product, requiring extensive chromatography or crystallization steps to meet stringent pharmaceutical purity specifications. In this novel system, the precipitation of the ligand allows for its physical removal from the reaction mixture before the product isolation steps begin. Since the ligand contains the bulk of the nitrogenous and sulfur-containing impurities potential, removing it early simplifies the purification of the diol intermediate. Furthermore, the stability of the ligand upon recovery means that degradation products, which could act as catalyst poisons or new impurity sources in subsequent runs, are minimized. The data indicates that the recovered ligand retains its activity and stereoselectivity over multiple cycles, suggesting that the molecular integrity of the chiral pocket is preserved. This robustness is critical for commercial scale-up of complex pharmaceutical intermediates, as it ensures batch-to-batch consistency and reduces the risk of off-specification production runs caused by catalyst variability.
How to Synthesize Paclitaxel C13 Side Chain Efficiently
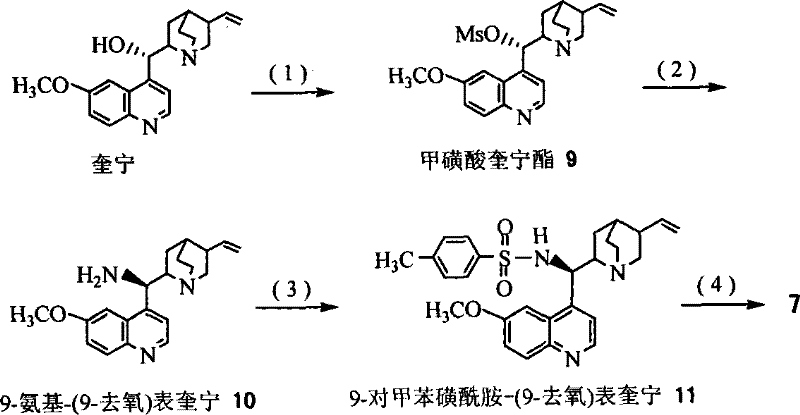
The synthesis of the target C13 side chains involves a multi-step sequence starting from the preparation of the ligand itself, followed by the key asymmetric dihydroxylation and subsequent functional group transformations. The process begins with the modification of quinine to introduce the necessary polar handles for recovery, such as the tosylamide and diol functionalities found in Ligand 7. Once the ligand is prepared, it is employed in the AD reaction of methyl cinnamate in the presence of osmium tetroxide and an oxidant like NMO. The resulting chiral diol is then subjected to a series of transformations including esterification, cyclization, ring-opening, and amination to yield the final N-protected phenylisoserine derivatives. These derivatives are the direct precursors that couple with baccatin III to form the final active pharmaceutical ingredients. The detailed standardized synthesis steps for implementing this efficient route are provided in the guide below.
- Prepare the recyclable cinchona alkaloid derivative ligand from quinine through substitution and oxidation reactions to ensure polar group functionality.
- Conduct the asymmetric dihydroxylation (AD) reaction of methyl cinnamate using the prepared ligand and osmium tetroxide catalyst in a homogeneous system.
- Recover the ligand by adding a non-polar solvent to precipitate it, followed by filtration, allowing the ligand to be reused for subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this recyclable ligand technology translates into tangible strategic advantages that go beyond simple chemistry. The primary value proposition is the drastic simplification of the cost structure associated with chiral synthesis. By eliminating the need to purchase fresh, expensive chiral ligands for every batch, manufacturers can achieve substantial cost savings in raw materials. The ability to recover the ligand with high efficiency means that the effective cost per kilogram of the C13 side chain is significantly lowered, making the final API more competitive in the global market. Moreover, the reliance on quinine, a commodity chemical with a stable supply chain, mitigates the risks associated with sourcing specialized, low-volume chiral reagents that are often subject to long lead times and supply disruptions. This shift enhances the overall resilience of the supply chain, ensuring continuous production even when market conditions for fine chemicals are volatile.
- Cost Reduction in Manufacturing: The economic impact of ligand recovery cannot be overstated in the context of high-value pharmaceutical intermediates. Traditional asymmetric syntheses often see the chiral ligand constitute a major portion of the variable cost. By implementing a system where the ligand is recovered via simple filtration and reused, the consumption of this high-value component is minimized. This qualitative reduction in material usage directly improves the gross margin of the manufacturing process. Additionally, the simplified workup procedure reduces the consumption of solvents and energy required for complex purification steps like column chromatography, further driving down operational expenditures. The elimination of transition metal catalyst residues also reduces the cost associated with metal scavenging technologies, streamlining the downstream processing workflow.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for life-saving medications. This technology leverages quinine, a naturally occurring alkaloid with a well-established global supply network, as the starting material for the ligand. Unlike synthetic chiral ligands that may depend on niche suppliers with limited capacity, quinine is produced at scale for various industries. This abundance ensures that the production of the ligand itself is not a bottleneck. Furthermore, the robustness of the ligand allows it to be stockpiled or regenerated on-site, reducing dependency on just-in-time deliveries of sensitive reagents. The stability of the recovered ligand over multiple cycles means that inventory management becomes more predictable, allowing for better planning and reduced safety stock requirements for critical chiral auxiliaries.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process offers a cleaner alternative to traditional methods. The 'homogeneous reaction, two-phase separation' technique minimizes waste generation by avoiding the use of large volumes of silica or other stationary phases typically required for ligand removal. The ability to recycle the ligand reduces the overall chemical footprint of the synthesis, aligning with green chemistry principles that are increasingly mandated by regulatory bodies. Scalability is enhanced because the separation method (filtration) is easily transferable from laboratory to pilot and commercial scales without the need for specialized equipment. This ease of scale-up reduces the time and capital investment required to bring new capacity online, ensuring that manufacturing can respond quickly to surges in demand for paclitaxel and docetaxel.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recyclable ligand technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of what partners can expect when adopting this methodology. Understanding these nuances is essential for R&D teams evaluating process feasibility and procurement teams assessing vendor capabilities. The answers reflect the specific advantages of the cinchona-derived system over conventional non-recyclable alternatives.
Q: How does the recyclable ligand reduce production costs compared to traditional methods?
A: Traditional methods often use expensive chiral ligands that are lost after a single use. This patent utilizes cheap quinine to create a ligand that can be recovered via simple filtration and reused at least 10 times without significant loss of activity or stereoselectivity, drastically lowering raw material costs.
Q: What is the stereoselectivity achieved with this novel ligand system?
A: The process achieves high optical purity, with experimental data showing product enantiomeric excess (ee) values reaching up to 99% in the initial runs and maintaining high levels (above 92% ee) even after multiple recycling cycles.
Q: Is this synthesis method scalable for industrial manufacturing?
A: Yes, the method is designed for scalability. It employs a 'homogeneous reaction, two-phase separation' strategy where the ligand dissolves during the reaction for high activity but precipitates upon adding non-polar solvents, simplifying the separation process and making it suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel C13 Side Chain Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN101029049B can be realized on an industrial scale. Our facilities are equipped with state-of-the-art reactors and rigorous QC labs capable of meeting stringent purity specifications required for oncology intermediates. We understand the critical nature of chiral purity and the complexities involved in osmium-catalyzed reactions, and our team is dedicated to optimizing these parameters to deliver high-quality C13 side chains consistently. Our commitment to quality assurance ensures that every batch meets the exacting standards necessary for subsequent coupling with baccatin III.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced technology for your supply chain. By working together, we can conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for the recyclable ligand process. Whether you are looking to secure a long-term supply of high-purity pharmaceutical intermediates or optimize an existing synthetic route, NINGBO INNO PHARMCHEM is ready to provide the technical support and manufacturing capacity needed to succeed in the competitive oncology market.
