Revolutionizing Paclitaxel Side Chain Synthesis with Recyclable Cinchona Alkaloid Ligands for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust solutions for the synthesis of complex anticancer agents, particularly paclitaxel and docetaxel, which remain cornerstone treatments for various malignancies. Patent CN101029049B introduces a transformative approach to this challenge by detailing the application of recyclable and reusable cinchona alkaloid derivative ligands in the synthesis of the critical C13 side chains for these drugs. This technology addresses the longstanding economic and logistical bottlenecks associated with asymmetric catalysis, specifically the high cost and waste generation of traditional chiral ligands. By leveraging cheap and readily available quinine as a starting material, the inventors have developed a catalytic system that not only maintains high stereoselectivity but also allows for efficient recovery and reuse. For R&D directors and procurement strategists, this represents a pivotal shift towards more sustainable and cost-effective pharmaceutical intermediate manufacturing, promising to stabilize supply chains for these high-demand oncology therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for paclitaxel and docetaxel C13 side chains often rely on asymmetric dihydroxylation or aminohydroxylation reactions that utilize expensive, single-use chiral ligands and precious metal catalysts. These conventional methods suffer from significant economic drawbacks, primarily due to the inability to recover the chiral inducers after the reaction is complete. Furthermore, the separation of the product from the catalyst system can be cumbersome, often requiring complex chromatographic purification steps that reduce overall yield and increase processing time. The reliance on non-recyclable materials also generates substantial chemical waste, posing environmental compliance challenges and inflating the cost of goods sold (COGS). For large-scale API manufacturing, these inefficiencies translate into higher prices and potential supply vulnerabilities, making the search for a more circular and efficient catalytic system a top priority for forward-thinking chemical enterprises.
The Novel Approach
The innovation described in the patent circumvents these issues by introducing a specialized cinchona alkaloid derivative ligand featuring a polar functional group, such as a hydroxyl or sulfonyl group, at a specific position on the molecular scaffold. This structural modification is the key to the ligand's unique solubility profile: it remains soluble during the homogeneous reaction phase to ensure high catalytic activity and stereocontrol, yet precipitates out of the solution when a non-polar solvent is added post-reaction. This "homogeneous reaction, two-phase separation" mechanism allows for the ligand to be recovered via simple filtration with a recovery rate exceeding 92%. 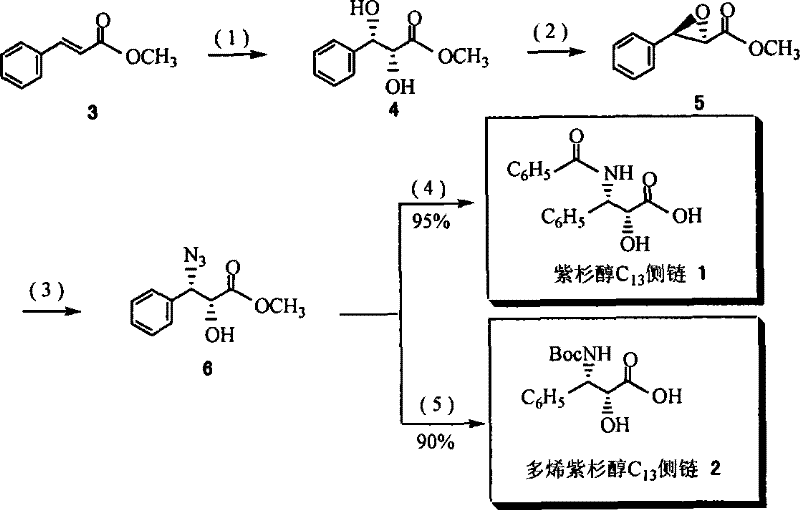 This recyclability means the same batch of ligand can be used repeatedly without significant degradation in performance, fundamentally altering the cost structure of the synthesis. This approach offers a compelling pathway for cost reduction in pharmaceutical intermediate manufacturing by minimizing raw material consumption and simplifying downstream processing.
This recyclability means the same batch of ligand can be used repeatedly without significant degradation in performance, fundamentally altering the cost structure of the synthesis. This approach offers a compelling pathway for cost reduction in pharmaceutical intermediate manufacturing by minimizing raw material consumption and simplifying downstream processing.
Mechanistic Insights into Asymmetric Dihydroxylation with Recyclable Ligands
The core of this technology lies in the Asymmetric Dihydroxylation (AD) reaction, where the recyclable ligand coordinates with osmium tetroxide to facilitate the enantioselective addition of hydroxyl groups across the double bond of cinnamate esters. The general structural formula (I) of the ligand, derived from quinine, provides a rigid chiral environment that dictates the facial selectivity of the oxidant attack. The presence of the polar R2 group (such as -OH or -SO2(CH2)nOH) is critical; it ensures the ligand dissolves in the polar reaction medium (often acetone-water mixtures), allowing for intimate contact between the catalyst and the substrate. This solubility is essential for achieving the high reaction rates and excellent enantiomeric excess (up to 99% ee) observed in the experimental data. Without this polar anchor, the ligand might phase-separate prematurely, leading to poor conversion and selectivity.
Furthermore, the mechanism of recovery is elegantly simple yet chemically profound. Once the dihydroxylation is complete, the addition of a non-polar solvent like ether disrupts the solvation shell around the polar ligand, causing it to crash out of the solution while the organic product remains dissolved. This physical separation avoids the need for destructive workups or complex extractions that could degrade the sensitive chiral catalyst. The recovered ligand retains its structural integrity and catalytic potency, as evidenced by its ability to be reused for at least 10 cycles with minimal loss in optical purity or yield. This robustness is a testament to the stability of the cinchona alkaloid backbone and the strategic placement of the functional groups, providing R&D teams with a reliable tool for high-purity pharmaceutical intermediate production that aligns with green chemistry principles.
How to Synthesize Paclitaxel C13 Side Chain Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing both paclitaxel and docetaxel side chains starting from inexpensive methyl cinnamate. The process begins with the preparation of the recyclable ligand itself, such as Ligand 7 or Ligand 8, which involves a sequence of mesylation, nucleophilic substitution with azide, catalytic hydrogenation, and sulfonamide formation, followed by a final oxidation step to install the polar diol or sulfone tail. 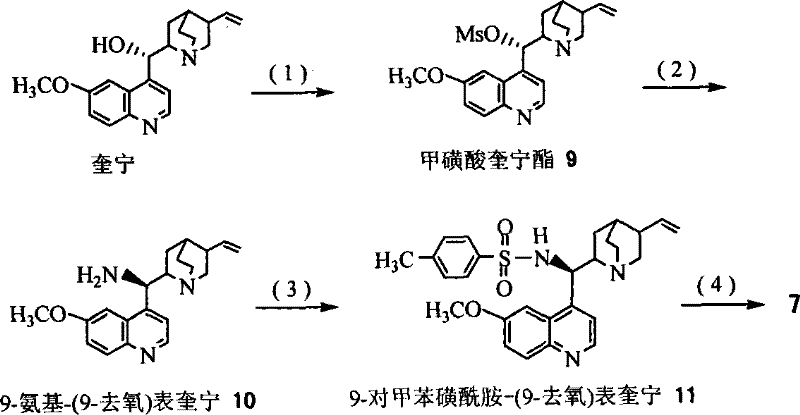 Once the ligand is prepared, it is employed in the key AD reaction with osmium tetroxide and NMO as the co-oxidant. The resulting chiral diol is then subjected to a series of transformations including epoxidation, ring-opening with azide, and final protection/deprotection steps to yield the target C13 side chain. The detailed standardized synthesis steps for this efficient pathway are provided below.
Once the ligand is prepared, it is employed in the key AD reaction with osmium tetroxide and NMO as the co-oxidant. The resulting chiral diol is then subjected to a series of transformations including epoxidation, ring-opening with azide, and final protection/deprotection steps to yield the target C13 side chain. The detailed standardized synthesis steps for this efficient pathway are provided below.
- Synthesize the recyclable ligand (e.g., Ligand 7 or 8) starting from quinine through mesylation, azide substitution, reduction, and sulfonamide formation followed by oxidation.
- Perform the Asymmetric Dihydroxylation (AD) reaction on methyl cinnamate using the recyclable ligand and osmium tetroxide catalyst to form the chiral diol intermediate.
- Recover the ligand by precipitation with non-polar solvents, then convert the diol intermediate into the final C13 side chain through epoxidation, ring opening, and protection steps.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this recyclable ligand technology offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage is the drastic simplification of the supply chain for chiral catalysts. Instead of sourcing expensive, proprietary ligands for every batch, manufacturers can produce their own ligand from commodity chemicals like quinine and reuse it indefinitely. This vertical integration capability significantly de-risks the supply chain against vendor lock-in and price volatility in the fine chemicals market. Moreover, the simplified separation process reduces the demand for extensive solvent usage and chromatography media, leading to lower operational expenditures and a smaller environmental footprint, which is increasingly critical for regulatory compliance in modern chemical manufacturing.
- Cost Reduction in Manufacturing: The ability to recover and reuse the chiral ligand eliminates the recurring cost of purchasing expensive catalysts for each production run. Since the ligand is derived from quinine, a relatively abundant natural product, the initial material cost is low, and the amortization of this cost over multiple cycles results in substantial savings. Additionally, the simplified workup procedure reduces labor hours and utility costs associated with complex purification steps, further driving down the overall production cost of the C13 side chain intermediates.
- Enhanced Supply Chain Reliability: By utilizing a ligand that can be synthesized in-house from readily available starting materials, companies can mitigate the risk of supply disruptions often associated with specialized reagents. The robustness of the ligand, which maintains its activity over multiple reuse cycles, ensures consistent production throughput without the need for frequent re-qualification of new catalyst batches. This stability is crucial for maintaining continuous manufacturing schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: The "precipitation and filtration" recovery method is inherently scalable, avoiding the bottlenecks of column chromatography which are difficult to implement on a multi-ton scale. This ease of scale-up facilitates the transition from pilot plant to commercial production with minimal process re-engineering. Furthermore, the reduction in chemical waste and solvent consumption aligns with stringent environmental regulations, reducing the burden on waste treatment facilities and enhancing the company's sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recyclable ligand technology in industrial settings. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, aiming to clarify the practical implications for potential adopters.
Q: How does the recyclable ligand improve cost efficiency in paclitaxel production?
A: The ligand, derived from inexpensive quinine, can be recovered with over 92% efficiency and reused multiple times without significant loss of stereoselectivity, drastically reducing the cost of chiral catalysts compared to traditional single-use ligands.
Q: What is the optical purity achieved with this novel catalytic system?
A: The process consistently achieves high optical purity, with reported enantiomeric excess (ee) values reaching up to 99% for the dihydroxylation products, ensuring the high quality required for pharmaceutical intermediates.
Q: Is this synthesis method scalable for industrial manufacturing?
A: Yes, the method utilizes a 'homogeneous reaction, two-phase separation' strategy where the ligand precipitates upon adding non-polar solvents, allowing for simple filtration and recovery, which is highly advantageous for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel C13 Side Chain Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value oncology intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the recyclable ligand system described in CN101029049B can be successfully translated into robust manufacturing operations. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify optical purity and impurity profiles. Our expertise allows us to navigate the complexities of chiral synthesis, ensuring that every batch of pharmaceutical intermediate meets the exacting standards required for downstream API synthesis.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your operational efficiency and reduce your total cost of ownership for critical cancer therapy intermediates.
