Advanced Ir-O-P Catalyzed Synthesis of Aryl Diboronic Esters for Pharmaceutical Intermediates
The landscape of organoboron chemistry is undergoing a significant transformation driven by the innovations detailed in Chinese Patent CN113980044B, which introduces a groundbreaking preparation method for diboronic acid and ester compounds utilizing a novel Ir-O-P type catalyst. This technology represents a pivotal advancement in C-H bond activation, addressing long-standing challenges in the synthesis of complex aryl and alkyl diboron species that are critical building blocks for modern drug discovery and material science. Unlike traditional methods that often suffer from narrow substrate scopes and harsh reaction conditions, this patented approach leverages the unique stability and reactivity of an iridium-oxygen-phosphorus coordination environment to facilitate efficient borylation. The process operates effectively under relatively mild thermal conditions ranging from 60-120°C and utilizes a one-pot strategy that streamlines the synthetic workflow, making it an attractive option for the production of high-purity pharmaceutical intermediates. By enabling the direct functionalization of diverse beta-N-aryl and alkane substrates, this methodology offers a robust platform for generating valuable boronate esters that serve as versatile precursors in Suzuki-Miyaura cross-coupling reactions and other downstream transformations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of this Ir-O-P catalytic system, the synthesis of aryl diboronic acid derivatives was heavily constrained by the specific nature of the starting materials and the rigidity of available catalytic cycles. Conventional techniques were largely restricted to synthesizing arylhydrazone diborides and diborides of 2-phenylpyridine derivatives, leaving a significant gap in the chemical space available to medicinal chemists and process developers. These traditional routes often necessitated multi-step synthetic sequences involving sensitive intermediates, which not only increased the overall cost of goods but also introduced opportunities for yield loss and impurity generation at each stage. Furthermore, the reliance on expensive and specialized ligands in older iridium-catalyzed systems often rendered large-scale production economically unfeasible, creating a bottleneck for supply chains requiring kilogram quantities of these specialized intermediates. The inability to efficiently borylate a broader range of nitrogen-containing heterocycles and functionalized aromatics limited the structural diversity accessible to researchers developing new active pharmaceutical ingredients.
The Novel Approach
The methodology disclosed in CN113980044B overcomes these historical barriers by introducing a versatile Ir-O-P type catalyst that exhibits exceptional activity across a wide array of substrate classes, including aryl oximes, aryl hydrazones, aryl triazenes, and 2-phenylpyridine derivatives. A key differentiator of this novel approach is its operational simplicity; it employs a one-pot procedure where the active catalyst is generated in situ or pre-formed, allowing for the direct conversion of readily available starting materials into the desired diboron products without the need for intermediate isolation. Remarkably, the system demonstrates such high intrinsic activity that in the specific case of aryl triazene diboride preparation, the reaction proceeds with high yields even in the absence of added phosphine ligands, further reducing raw material costs. This flexibility allows for the design of complex molecular architectures with high atom economy, providing a reliable pathway for cost reduction in fine chemical manufacturing while maintaining the rigorous purity standards required for regulatory compliance.

Mechanistic Insights into Ir-O-P Catalyzed C-H Borylation
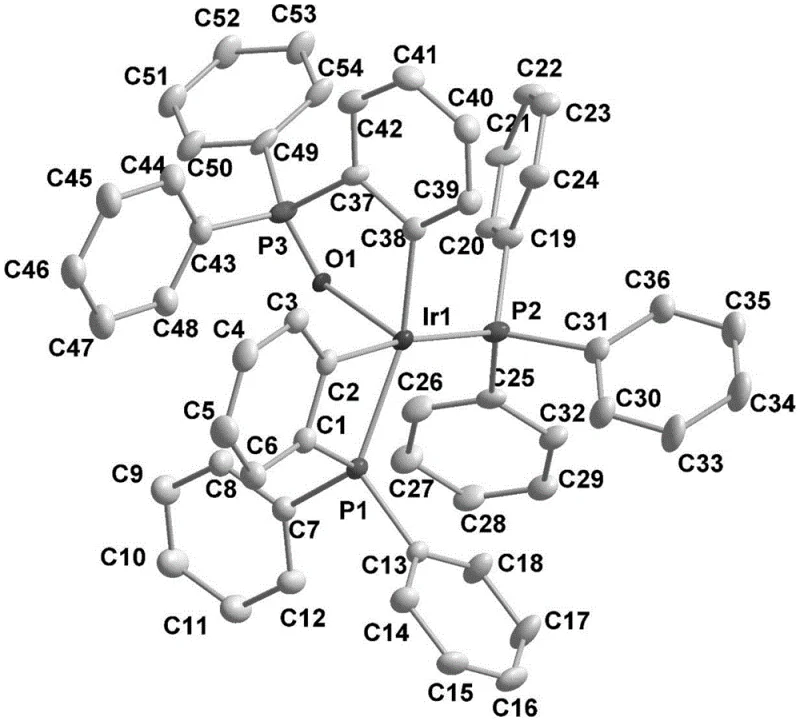
The core of this technological breakthrough lies in the formation and function of the active Ir-O-P catalyst, which features a distinct coordination environment that enhances both stability and reactivity compared to standard iridium complexes. The catalytic cycle initiates with the oxidative addition of the metal iridium species to the organic boron source, facilitated by the unique electronic properties imparted by the oxygen-phosphorus ligand framework. This active species then engages the beta-N-aryl or alkane substrate through a second oxidative addition step, effectively activating the inert carbon-hydrogen bond at the ortho-position relative to the directing group. The subsequent boron transfer and reductive elimination steps are highly efficient, driven by the thermodynamic stability of the resulting C-B bonds and the regeneration of the active iridium species. The presence of the Ir-O-P bond structure, specifically identified in the patent with the molecular formula C54H43IrOP3 when using triphenylphosphine, provides a rigid yet flexible scaffold that accommodates steric bulk while maintaining the necessary electronic density for catalysis.
From an impurity control perspective, the mechanism offers significant advantages by minimizing side reactions that typically plague C-H activation processes, such as over-borylation or decomposition of sensitive functional groups. The specific geometry of the Ir-O-P active site appears to enforce a high degree of regioselectivity, ensuring that borylation occurs predominantly at the desired position adjacent to the nitrogen directing group. This selectivity is crucial for pharmaceutical applications where isomeric impurities can be difficult to separate and may pose toxicological risks. Furthermore, the catalyst's tolerance to air during its formation phase, followed by its operation under anhydrous conditions, suggests a robustness that simplifies handling requirements without compromising the integrity of the reaction mixture. The ability to utilize common solvents like tetrahydrofuran and inexpensive ligands like triphenylphosphine further underscores the practical viability of this mechanism for industrial application, as it avoids the need for exotic reagents that could introduce trace metal contaminants or difficult-to-remove byproducts.
How to Synthesize Aryl Diboronic Esters Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for generating high-quality diboronic acid esters suitable for immediate use in downstream coupling reactions. The process begins with the preparation of the active catalyst, which can be achieved by reacting an iridium precursor such as methoxycyclooctadiene iridium with a phosphine ligand in an organic solvent under air, followed by the introduction of substrates under inert atmosphere. This two-stage addition ensures that the catalyst is fully activated before exposure to the sensitive boron sources and substrates, maximizing turnover numbers and overall yield. The reaction is typically conducted at temperatures between 60-120°C for durations ranging from 12 to 120 hours, depending on the specific steric and electronic nature of the substrate, allowing for fine-tuning of the process to accommodate diverse chemical structures. Detailed standardized synthesis steps for specific examples are provided in the guide below to assist process chemists in replicating these results.
- Form the active Ir-O-P catalyst by reacting an iridium metal complex with a phosphine ligand in an organic solvent under air conditions.
- Under anhydrous and oxygen-free conditions, add the organic boron source, beta-N-aryl substrate, and the active catalyst to the solvent.
- Heat the mixture to 60-120°C for 12-120 hours, then perform post-treatment including concentration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Ir-O-P catalyzed borylation method presents a compelling value proposition centered on cost efficiency, supply reliability, and operational scalability. The shift from multi-step, ligand-intensive processes to a streamlined one-pot reaction significantly reduces the consumption of raw materials and solvents, directly translating to lower manufacturing costs per kilogram of product. By eliminating the need for expensive, specialized ligands in many cases and utilizing commercially available iridium precursors and boron sources, the overall bill of materials is optimized, facilitating substantial cost savings in the production of complex pharmaceutical intermediates. Additionally, the simplified post-treatment process, which involves standard concentration and column chromatography or recrystallization, reduces the burden on purification infrastructure and shortens the overall production cycle time, enhancing the responsiveness of the supply chain to market demands.
- Cost Reduction in Manufacturing: The economic benefits of this technology are driven primarily by the use of inexpensive and readily available reagents, such as triphenylphosphine and common organic solvents like THF, which replace costly proprietary ligands and specialized reaction media. The high atom economy of the one-pot process minimizes waste generation and reduces the volume of solvents required for intermediate isolations, leading to a leaner and more cost-effective manufacturing footprint. Furthermore, the ability to achieve high yields even at gram-scale expansions suggests that the process maintains its efficiency upon scaling, preventing the yield erosion that often plagues the transition from laboratory to pilot plant production. This efficiency ensures that the final cost of the diboronic acid intermediates remains competitive, supporting margin preservation for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: The robustness of the Ir-O-P catalyst system contributes to a more resilient supply chain by reducing the risk of batch failures and ensuring consistent product quality across different production runs. The method's tolerance for a wide range of substrates means that a single catalytic platform can be used to produce a diverse portfolio of intermediates, simplifying inventory management and reducing the need for multiple specialized production lines. The use of stable, commercially sourced starting materials mitigates the risk of supply disruptions associated with custom-synthesized reagents, ensuring continuity of supply even in volatile market conditions. This reliability is critical for maintaining the production schedules of active pharmaceutical ingredients where delays can have significant commercial consequences.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process aligns well with green chemistry principles by operating at moderate temperatures and utilizing solvents that are easier to recover and recycle compared to more hazardous alternatives. The reduction in synthetic steps inherently lowers the E-factor (mass of waste per mass of product), simplifying waste treatment protocols and reducing the environmental footprint of the manufacturing facility. The scalability of the reaction, demonstrated by its successful expansion to gram-level quantities with maintained high yields, indicates a clear path to multi-kilogram and ton-scale production without the need for fundamental process redesign. This scalability ensures that the technology can meet the growing demand for boron-containing intermediates in the pharmaceutical and agrochemical sectors while adhering to increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ir-O-P catalyzed borylation technology, drawing directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational parameters and potential advantages for partners considering this methodology for their own synthesis campaigns. Understanding these nuances is essential for evaluating the feasibility of integrating this process into existing manufacturing workflows and for assessing its alignment with specific project goals regarding purity, cost, and timeline.
Q: Is a phosphine ligand always required for this Ir-O-P catalyzed borylation?
A: While triphenylphosphine is the preferred ligand for optimal yields, the patent indicates that for aryl triazene-type diborides, the target compound can be obtained in high yield even without adding a ligand, showcasing the robustness of the iridium system.
Q: What types of substrates are compatible with this C-H borylation method?
A: The method demonstrates excellent functional group tolerance, successfully borylating aryl oximes, aryl hydrazones, aryl triazenes, and 2-phenylpyridine derivatives, significantly expanding the scope beyond traditional limitations.
Q: Can this synthesis method be scaled up for industrial production?
A: Yes, the patent explicitly states that the method has been successfully expanded to gram-level reactions with very high yields, indicating strong potential for commercial scale-up due to its simple one-pot operation and easy post-treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diboronic Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Ir-O-P catalyzed synthesis method for producing high-value diboronic acid and ester compounds, and we are uniquely positioned to leverage this technology for our global clientele. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results observed in the patent can be reliably translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of diboronic acid intermediate we deliver meets the exacting standards required for clinical and commercial drug substance manufacturing. We understand that consistency and quality are paramount in the pharmaceutical supply chain, and our commitment to process excellence ensures that we can deliver these complex intermediates with the reliability your projects demand.
We invite you to engage with our technical team to explore how this advanced borylation methodology can optimize your specific synthesis routes and drive value for your organization. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this technology offers compared to your current sourcing or manufacturing strategies. We encourage you to contact our technical procurement team today to discuss your specific requirements, request specific COA data for relevant compounds, and obtain route feasibility assessments tailored to your unique molecular targets. Let us partner with you to accelerate your development timelines and secure a robust supply of critical building blocks for your next generation of therapeutics.
