Advanced Ir-O-P Catalyzed Diboration for Commercial Scale-Up of Complex Pharmaceutical Intermediates
Advanced Ir-O-P Catalyzed Diboration for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The landscape of organoboron chemistry is undergoing a significant transformation driven by the need for more efficient and versatile C-H activation strategies. A pivotal development in this field is detailed in Chinese Patent CN113980044B, which discloses a novel preparation method for diboronic acid/ester compounds utilizing a unique Ir-O-P type catalyst. This technology represents a substantial leap forward for the synthesis of high-purity pharmaceutical intermediates, addressing long-standing challenges regarding substrate scope and operational complexity. By leveraging an iridium-phosphine-oxygen catalytic system, this method enables the direct diboration of beta-N-aryl substrates under relatively mild thermal conditions. For R&D directors and procurement specialists alike, this innovation signals a new era of accessibility for complex organoboron building blocks, which are critical motifs in modern drug discovery and material science applications.
The core value proposition of this patent lies in its ability to streamline the synthesis of aryl diboronic acid esters through a robust one-pot procedure. Traditionally, accessing these valuable intermediates required multi-step synthetic routes that were often plagued by low atom economy and harsh reaction conditions. The disclosed Ir-O-P catalyst system circumvents these issues by facilitating direct C-H borylation with high regioselectivity. This capability is particularly relevant for the production of reliable pharmaceutical intermediate supplier portfolios, where speed-to-market and structural diversity are paramount. The method's compatibility with a wide array of functional groups ensures that medicinal chemists can rapidly iterate on lead compounds without being constrained by synthetic bottlenecks.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of aryl diboronic acid derivatives was severely restricted by the limitations of available catalytic systems and substrate compatibility. Conventional methods were largely confined to specific classes of compounds, such as arylhydrazone diborides and diborides of 2-phenylpyridine derivatives. This narrow scope meant that a vast chemical space remained inaccessible or required prohibitively expensive and lengthy synthetic pathways to access. Furthermore, traditional iridium-catalyzed borylation often relied on sophisticated, custom-synthesized ligands that drove up the cost of goods significantly. These legacy processes also frequently suffered from sensitivity to air and moisture, necessitating rigorous exclusion techniques that complicate scale-up and increase operational expenditures in a manufacturing setting.
The Novel Approach
The methodology presented in CN113980044B fundamentally disrupts these constraints by introducing a versatile Ir-O-P catalytic cycle that operates effectively with inexpensive, commercially available ligands like triphenylphosphine. This novel approach expands the substrate scope dramatically to include aryl oximes, aryl hydrazones, aryl triazenes, and various 2-phenylpyridine derivatives. Remarkably, the system demonstrates such high intrinsic activity that for aryl triazene-type substrates, the reaction proceeds with high yields even in the absence of added ligands. This simplification of the catalyst system not only reduces raw material costs but also streamlines the purification process, as there are fewer ligand-derived impurities to remove. The ability to achieve these transformations in a one-pot fashion underpins a significant reduction in processing time and waste generation.
Mechanistic Insights into Ir-O-P Catalyzed C-H Activation
The efficacy of this transformation is rooted in the unique electronic and steric properties of the active Ir-O-P catalyst species. As illustrated in the structural data, the active catalyst features a distinct Iridium-Oxygen-Phosphorus coordination environment, formed initially by the reaction of an iridium metal species with a phosphine ligand under air conditions. This specific coordination geometry stabilizes the iridium center against deactivation while maintaining sufficient lability to allow for substrate binding. The catalytic cycle initiates with the oxidative addition of the organic boron source to the metal center, generating a highly reactive iridium-boron species capable of activating inert C-H bonds. This step is critical for overcoming the high bond dissociation energy of aromatic C-H bonds without requiring directing groups that limit structural diversity.
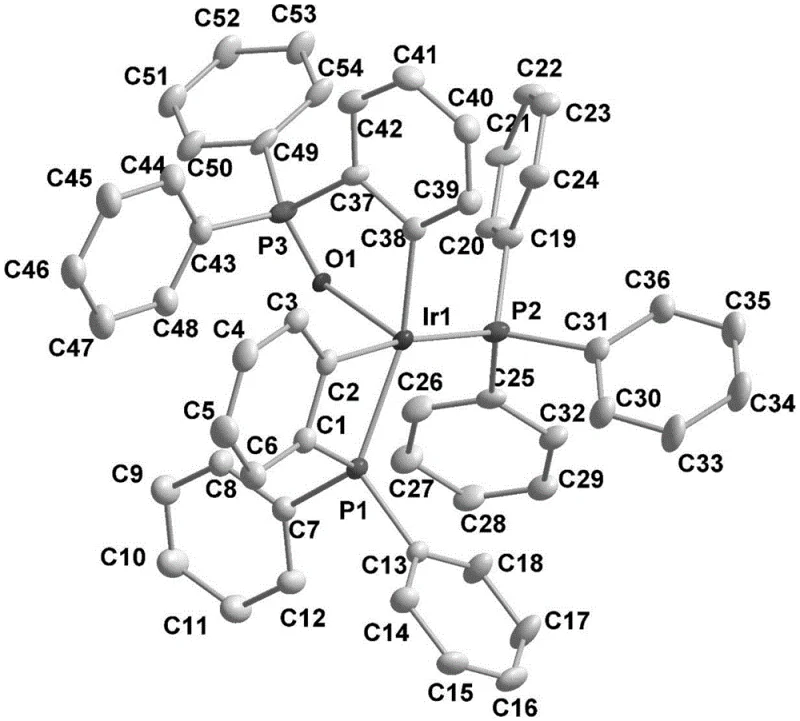
Following C-H activation, the mechanism proceeds through a boron transfer step where the boron moiety is delivered to the ortho-position of the aromatic ring relative to the nitrogen directing group. This regioselectivity is driven by the coordination of the substrate's nitrogen atom to the iridium center, which positions the metal in close proximity to the target C-H bond. The cycle concludes with a reductive elimination step that releases the diborated product and regenerates the active catalyst species. Crucially, the Ir-O-P framework appears to enhance the turnover number and stability of the catalyst, allowing the reaction to proceed efficiently at temperatures between 60-120°C. This mechanistic robustness ensures consistent impurity profiles, a key consideration for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Aryl Diboronic Acid Esters Efficiently
Implementing this synthesis route requires careful attention to the preparation of the active catalyst and the maintenance of anhydrous conditions during the reaction phase. The process begins with the in situ generation of the Ir-O-P catalyst, followed by the addition of the beta-N-aryl substrate and the boron source in a suitable organic solvent such as tetrahydrofuran (THF). The reaction is then heated to facilitate the C-H activation and borylation steps. Detailed standard operating procedures for this synthesis, including specific molar ratios and purification protocols, are outlined in the technical guide below to ensure reproducibility and safety.
- Preparation of the active Ir-O-P catalyst by reacting an iridium metal complex with a phosphine ligand (e.g., triphenylphosphine) under air conditions in an organic solvent.
- Addition of the beta-N-aryl substrate and organic boron source (e.g., bis(pinacolato)diboron) to the catalyst mixture under anhydrous and oxygen-free conditions.
- Heating the reaction mixture to 60-120°C for 12-120 hours, followed by concentration and purification via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this Ir-O-P catalyzed diboration method offers profound benefits for cost reduction in fine chemical manufacturing. The primary driver of economic efficiency is the substitution of expensive, proprietary ligands with commodity chemicals like triphenylphosphine. This shift drastically lowers the bill of materials for the catalyst system, which is often a significant cost center in precious metal catalysis. Additionally, the simplified workup procedure, which typically involves concentration and standard column chromatography or recrystallization, reduces the demand for specialized purification resins and solvents. These factors combine to create a more lean and cost-effective production model that enhances overall margin potential for high-volume intermediates.
- Cost Reduction in Manufacturing: The elimination of complex ligand synthesis and the use of abundant iridium precursors significantly lower the entry barrier for production. By avoiding multi-step ligand preparation, manufacturers can reduce both labor and material costs associated with catalyst preparation. Furthermore, the high yields reported across a broad range of substrates minimize the loss of valuable starting materials, thereby improving the overall atom economy of the process. This efficiency translates directly into a more competitive pricing structure for the final diboronic acid products.
- Enhanced Supply Chain Reliability: The reliance on commercially available raw materials, such as bis(pinacolato)diboron and common iridium salts, mitigates supply chain risks associated with bespoke reagents. Since the key components are standard catalog items, procurement teams can secure multiple sourcing options, ensuring continuity of supply even during market fluctuations. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, further stabilizing the supply chain against disruptions.
- Scalability and Environmental Compliance: The successful demonstration of this method at the gram-level with high yields provides a strong foundation for commercial scale-up of complex pharmaceutical intermediates. The use of standard solvents like THF and toluene allows for easy integration into existing reactor trains without the need for specialized equipment. Moreover, the one-pot nature of the reaction reduces the generation of intermediate waste streams, aligning with green chemistry principles and simplifying environmental compliance and waste disposal logistics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ir-O-P catalyzed diboration technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the key advantages of the Ir-O-P catalyst over traditional iridium systems?
A: The Ir-O-P catalyst described in patent CN113980044B offers superior stability and activity. Unlike traditional systems that often require expensive, specialized ligands, this method utilizes cheap, commercially available triphenylphosphine. Furthermore, the catalyst demonstrates remarkable tolerance, allowing for the diboration of diverse substrates including aryl triazenes and oximes, which were previously difficult to functionalize efficiently.
Q: Can this diboration process be scaled for industrial manufacturing?
A: Yes, the patent explicitly highlights the scalability of this method. It has been successfully demonstrated at the gram-level with high yields, indicating strong potential for kilogram-to-ton scale-up. The use of standard organic solvents like THF and simple post-treatment procedures (concentration and column chromatography) aligns well with existing fine chemical manufacturing infrastructure, facilitating a smoother transition from lab to plant.
Q: What is the substrate scope for this C-H borylation reaction?
A: The method exhibits a broad substrate scope, accommodating various functional groups. It is effective for beta-N-aryl compounds including aryl oximes, aryl hydrazones, aryl triazenes, and 2-phenylpyridine derivatives. The reaction tolerates substituents such as halogens (F, Cl, Br, I), alkyl groups, alkoxy groups, and trifluoromethyl groups, making it highly versatile for synthesizing diverse pharmaceutical building blocks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diboronic Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating drug discovery pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the Ir-O-P catalyzed diboration can be seamlessly transitioned to industrial manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of organoboron intermediate meets the exacting standards required by global pharmaceutical clients. Our commitment to technical excellence ensures that complex synthetic challenges are met with robust, scalable solutions.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce time-to-market for your critical therapeutic candidates.
