Revolutionizing Boronate Ester Production: A Mild n-Butyllithium Catalyzed Route for Commercial Scale
Revolutionizing Boronate Ester Production: A Mild n-Butyllithium Catalyzed Route for Commercial Scale
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more efficient, cost-effective, and scalable methodologies. A significant breakthrough in this domain is documented in patent CN111320644A, which details a novel method for preparing boronate esters via hydroboration reactions. This technology leverages the high activity of commercial n-butyllithium to catalyze the reaction between imines and boranes under remarkably mild conditions. For R&D directors and process chemists, this represents a paradigm shift away from苛刻 (harsh) traditional methods towards a streamlined, atom-economic process. The ability to achieve yields exceeding 90% at room temperature using a commodity reagent fundamentally alters the economic and operational feasibility of producing these critical pharmaceutical intermediates. This report analyzes the technical depth and commercial implications of this innovation for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the hydroboration of unsaturated C=N bonds has presented significant challenges compared to the more facile hydroboration of C=O bonds. Prior art solutions often relied on catalytic systems involving main group elements such as magnesium, calcium, sodium, rhenium, or zinc. These conventional catalysts suffer from substantial drawbacks that hinder industrial adoption. Firstly, many of these catalytic species are expensive to synthesize or purchase, driving up the raw material costs for the final API intermediate. Secondly, the reaction conditions required are often severe, necessitating high temperatures and extended reaction times, which increases energy consumption and complicates process safety. Furthermore, some existing systems exhibit low yields or poor selectivity, leading to difficult purification processes and increased waste generation. The complexity of ligand design for these metal centers also adds a layer of intellectual property risk and supply chain fragility for manufacturers relying on proprietary catalysts.
The Novel Approach
In stark contrast to the complexities of prior art, the method disclosed in CN111320644A utilizes commercial n-butyllithium as a highly efficient catalyst. This approach simplifies the synthetic route dramatically by eliminating the need for specialized, hard-to-prepare catalyst precursors. The reaction proceeds smoothly at room temperature, typically completing within 1 to 2 hours, which significantly reduces the thermal load on manufacturing equipment. As illustrated in the reaction scheme below, the transformation of imines to boronic esters is achieved with exceptional efficiency.

The use of n-butyllithium, a standard reagent in organic synthesis, ensures consistent quality and availability. Moreover, the protocol tolerates a wide range of substrates, maintaining high yields even with varying electronic environments on the aromatic rings. This robustness is critical for process chemistry, where batch-to-batch consistency is paramount. By shifting to this milder, reagent-based catalysis, manufacturers can bypass the bottlenecks associated with transition metal or complex main-group catalyst sourcing.
Mechanistic Insights into n-Butyllithium Catalyzed Hydroboration
The success of this methodology lies in the unique reactivity of n-butyllithium towards the imine-borane system. While organolithium reagents are traditionally viewed as stoichiometric bases or nucleophiles, this patent elucidates their potential as catalytic activators for hydroboration. The mechanism likely involves the initial coordination or deprotonation events that activate the borane species, facilitating its addition across the C=N double bond. This activation lowers the energy barrier for the hydroboration step, allowing it to proceed rapidly at ambient temperatures. Understanding this mechanistic pathway is vital for R&D teams aiming to optimize the process further or adapt it to continuous flow reactors. The simplicity of the catalytic cycle suggests a low propensity for catalyst deactivation, which supports long run-times and high turnover numbers in a production setting.
Furthermore, the specificity of the catalyst is highlighted by comparative studies within the patent data. For instance, substituting n-butyllithium with other lithium amide compounds, such as the structure shown in Formula I, fails to yield the desired product.
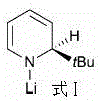
This negative result underscores the precise electronic and steric requirements for effective catalysis in this system. It confirms that not just any strong base will suffice; the specific nature of the n-butyl group is essential for the observed reactivity. From an impurity control perspective, the mild conditions minimize side reactions such as polymerization or over-reduction, which are common pitfalls in high-temperature hydroborations. The resulting crude product profile is cleaner, reducing the burden on downstream purification units like crystallization or chromatography. This level of control over the reaction trajectory is a key indicator of a mature, scalable process suitable for GMP manufacturing environments.
The substrate scope is another area where this mechanism shines. The system accommodates imines with diverse substituents, including electron-withdrawing halogens and electron-donating methoxy or methyl groups.
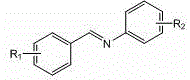
This versatility implies that the catalytic cycle is robust against electronic perturbations on the aromatic rings. For process developers, this means a single set of operating parameters can potentially cover a library of analogues, streamlining the development of new drug candidates. The ability to handle both electron-rich and electron-poor substrates without modifying the catalyst loading or temperature profile simplifies the tech transfer package significantly.
How to Synthesize Boronate Ester Efficiently
Implementing this synthesis route requires strict adherence to anhydrous and oxygen-free conditions to maintain catalyst integrity. The general procedure involves charging a dried reaction vessel with the imine substrate and tetrahydrofuran solvent under an inert atmosphere. Following this, pinacolborane is added, and the mixture is homogenized before the introduction of the n-butyllithium catalyst. The reaction is allowed to stir at room temperature for a short duration, typically 1 to 2 hours, after which it is quenched by exposure to air.
- Prepare the reaction vessel under anhydrous and oxygen-free conditions with an inert gas atmosphere, then add the imine substrate and organic solvent such as THF.
- Introduce pinacolborane to the mixture followed by the addition of the n-butyllithium catalyst, ensuring thorough mixing for uniform reaction initiation.
- Stir the reaction at room temperature for 1 to 2 hours, terminate by exposure to air, and remove the solvent under reduced pressure to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this n-butyllithium catalyzed process offers tangible strategic benefits beyond mere chemical elegance. The primary advantage stems from the utilization of commodity chemicals. n-Butyllithium and pinacolborane are widely produced and available from multiple global suppliers, mitigating the risk of single-source dependency that often plagues projects using bespoke catalysts. This abundance translates directly into cost stability and negotiating power for purchasing departments. Additionally, the elimination of expensive transition metals or complex ligand systems removes a significant cost center from the bill of materials. The reduction in raw material costs is compounded by the operational efficiencies gained from running reactions at room temperature.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the drastic simplification of the input materials. By replacing costly, specialized catalysts with inexpensive n-butyllithium, the direct material cost per kilogram of product is significantly lowered. Furthermore, the mild reaction conditions eliminate the need for energy-intensive heating or cooling cycles, resulting in substantial utility savings. The high yields reported, often reaching up to 99%, mean that less raw material is wasted, improving the overall mass balance and reducing the cost of goods sold. This efficiency allows for more competitive pricing strategies in the final API market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of standard reagents that are not subject to the same geopolitical or manufacturing bottlenecks as exotic catalysts. Since n-butyllithium is a staple in the chemical industry, lead times for raw materials are generally shorter and more predictable. This reliability ensures that production schedules can be met consistently, reducing the risk of delays in delivering critical pharmaceutical intermediates to clients. The robustness of the process also means that scale-up from pilot plant to commercial production is less risky, as there are fewer variables related to catalyst performance that could fluctuate between batches.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, the process aligns well with green chemistry principles. The use of THF as a solvent is standard and manageable with existing recovery infrastructure. The absence of heavy metals simplifies waste treatment protocols and reduces the regulatory burden associated with metal residue limits in pharmaceutical products. The short reaction time of 1 to 2 hours increases the throughput capacity of existing reactor trains, allowing facilities to produce more volume without capital expansion. This scalability is essential for meeting the growing demand for boron-containing drugs in the oncology and infectious disease sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. They are derived from the specific experimental data and claims found in the patent literature to provide accurate guidance for potential partners.
Q: What are the advantages of using n-butyllithium over traditional catalysts for imine hydroboration?
A: Unlike traditional catalysts based on magnesium, calcium, or zinc which often require high temperatures and complex preparation, n-butyllithium is a commercially available reagent that operates efficiently at room temperature with high yields up to 99%.
Q: Does this catalytic system tolerate various electronic substituents on the imine substrate?
A: Yes, the patent demonstrates excellent universality for imines with different substitution positions and electronic effects, including electron-withdrawing groups like halogens and electron-donating groups like methyl and methoxy.
Q: What are the typical reaction conditions and catalyst loading required?
A: The reaction proceeds under mild conditions at room temperature for 1 to 2 hours using tetrahydrofuran as the solvent, with a low catalyst loading of only 4 to 5 mol% relative to the imine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boronate Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the n-butyllithium catalyzed hydroboration technology described in CN111320644A. As a leading CDMO, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this efficient route to market. Our facilities are equipped with rigorous QC labs and stringent purity specifications to ensure that every batch of boronate ester meets the highest international standards. We understand that consistency and quality are non-negotiable for our partners in the pharmaceutical industry, and our infrastructure is designed to deliver exactly that.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule. Please contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can optimize your supply chain and accelerate your time to market with high-quality, cost-effective intermediates.
