Advanced Palladium-Catalyzed Synthesis of N-Acyl Indole Intermediates for Pharmaceutical Manufacturing
Introduction to Next-Generation Indole Synthesis
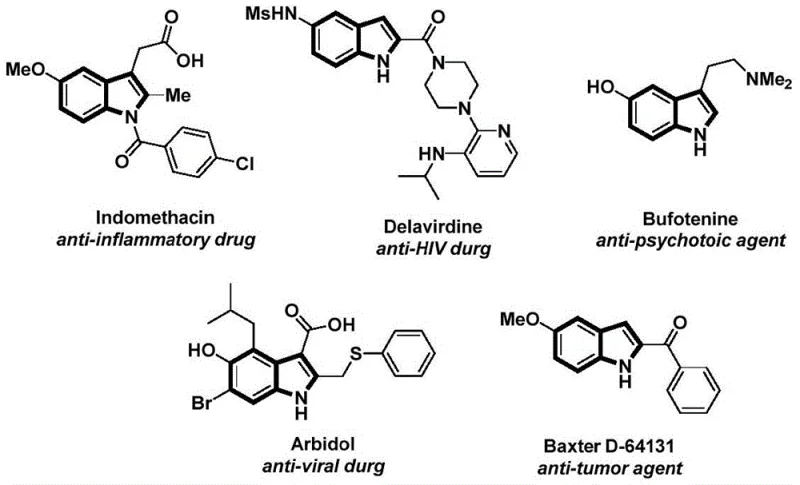
The structural motif of the indole ring serves as a foundational backbone in modern medicinal chemistry, appearing ubiquitously in bioactive natural products and approved pharmaceutical agents ranging from anti-inflammatory drugs like Indomethacin to potent anti-HIV agents such as Delavirdine. As depicted in the visual overview of indole-based therapeutics, the versatility of this heterocyclic system is unmatched, yet the efficient construction of complex N-acyl indole derivatives remains a persistent challenge for process chemists. Patent CN112898192B introduces a transformative preparation method that addresses these synthetic bottlenecks by utilizing a palladium-catalyzed carbonylation cyclization strategy. This novel approach leverages readily available 2-alkynyl anilines and aryl iodides as starting materials, bypassing the traditional limitations associated with gaseous carbon monoxide handling. By integrating a solid carbon monoxide surrogate and a mild oxidative cyclization step, this technology offers a robust pathway for generating high-purity pharmaceutical intermediates with exceptional functional group tolerance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indole compounds via carbonylation has been hindered by significant operational hazards and technical inefficiencies that complicate large-scale manufacturing. Traditional protocols frequently rely on the direct use of carbon monoxide gas, which necessitates specialized high-pressure reactors and rigorous safety protocols to mitigate the risks of toxicity and explosion, thereby inflating capital expenditure and operational complexity. Furthermore, conventional catalytic systems often suffer from poor substrate compatibility, particularly when dealing with electron-deficient or sterically hindered aryl halides, leading to inconsistent yields and difficult purification profiles. The requirement for harsh reaction conditions, such as elevated temperatures exceeding 100°C or the use of strong bases, can also trigger unwanted side reactions and decomposition of sensitive functional groups, ultimately compromising the purity of the final active pharmaceutical ingredient (API) intermediate.
The Novel Approach
In stark contrast to these legacy methods, the disclosed invention employs a sophisticated two-stage tandem reaction sequence that operates under remarkably mild and safe conditions, fundamentally reshaping the cost reduction in pharmaceutical intermediate manufacturing landscape. By substituting hazardous CO gas with 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide source, the process eliminates the need for pressurized gas infrastructure, allowing the reaction to proceed smoothly in standard glassware or stainless steel vessels at a moderate temperature of 60°C. The strategic addition of silver oxide in the second stage facilitates a clean oxidative cyclization, ensuring high conversion rates and minimizing the formation of polymeric byproducts. This streamlined methodology not only simplifies the post-treatment workflow—often requiring only filtration and standard column chromatography—but also dramatically expands the scope of accessible chemical space for drug discovery teams seeking diverse N-acyl indole scaffolds.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The mechanistic elegance of this transformation lies in its sequential palladium-mediated steps, which orchestrate the assembly of the indole core with precision and atom economy. The catalytic cycle initiates with the oxidative addition of the zero-valent palladium species, typically tetrakis(triphenylphosphine)palladium, into the carbon-iodine bond of the aryl iodide substrate to generate a reactive aryl-palladium(II) intermediate. Subsequently, carbon monoxide, which is slowly and controllably released from the thermal decomposition of the TFBen surrogate, inserts into the palladium-carbon bond to form an acyl-palladium complex. This acyl species then undergoes nucleophilic attack by the amino group of the 2-alkynyl aniline, followed by reductive elimination to yield an amide intermediate, regenerating the active palladium catalyst for the next turnover.
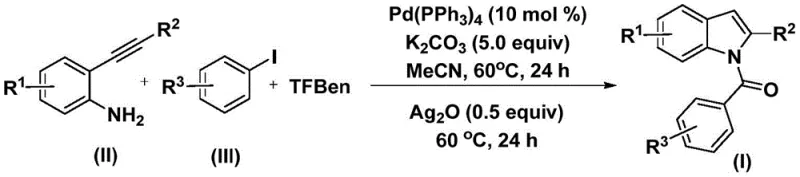
Following the formation of the amide precursor, the reaction trajectory shifts towards cyclization driven by the introduction of silver oxide. The silver species acts as a mild oxidant and Lewis acid promoter, activating the alkyne moiety towards intramolecular nucleophilic attack by the nitrogen atom or facilitating a dehydrogenative coupling process that closes the five-membered pyrrole ring. This dual-catalyst system ensures that the cyclization proceeds with high regioselectivity, avoiding the formation of isomeric byproducts that often plague indole syntheses. The robustness of this mechanism is evidenced by its tolerance to a wide array of substituents, including halogens, alkyl groups, and alkoxy chains, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates where impurity control is paramount.
How to Synthesize N-Acyl Indole Efficiently
The practical execution of this synthesis protocol is designed for maximum reproducibility and ease of operation, making it highly attractive for both laboratory-scale optimization and pilot plant production. The process begins by combining the palladium catalyst, potassium carbonate base, the solid CO source TFBen, the 2-alkynyl aniline, and the aryl iodide in acetonitrile solvent within a sealed reaction vessel. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined below to ensure consistent high-yield results across different batches.
- Mix palladium catalyst, potassium carbonate, TFBen (CO source), 2-alkynyl aniline, and aryl iodide in acetonitrile.
- Heat the mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation.
- Add silver oxide and continue heating at 60°C for another 24 hours to complete the cyclization to the N-acyl indole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond simple yield improvements. By replacing gaseous reagents with stable solids and utilizing commodity chemicals like potassium carbonate and silver oxide, the process significantly reduces the dependency on specialized hazardous material logistics, thereby enhancing supply chain reliability and reducing lead time for high-purity pharmaceutical intermediates. The mild reaction temperature of 60°C lowers energy consumption compared to high-temperature alternatives, contributing to a more sustainable and cost-effective manufacturing footprint that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The elimination of high-pressure CO gas equipment represents a substantial capital saving, as facilities no longer require expensive autoclaves or dedicated gas handling infrastructure to run this chemistry. Furthermore, the use of TFBen as a stoichiometric CO source allows for precise control over the carbonylation step, minimizing waste and maximizing the utilization of expensive palladium catalysts, which drives down the overall cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: All key reagents, including tetrakis(triphenylphosphine)palladium, aryl iodides, and 2-alkynyl anilines, are commercially available from multiple global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions ensures that minor variations in raw material quality do not compromise the batch outcome, providing a stable and predictable production schedule that is critical for meeting tight delivery windows in the pharmaceutical sector.
- Scalability and Environmental Compliance: The simplicity of the post-treatment process, which involves basic filtration and silica gel purification, facilitates easy scale-up from gram to multi-kilogram quantities without the need for complex engineering controls. Additionally, the avoidance of toxic gas emissions and the use of relatively benign solvents like acetonitrile simplify waste stream management, ensuring compliance with increasingly stringent environmental regulations regarding volatile organic compounds and heavy metal discharge.
Frequently Asked Questions (FAQ)
To further clarify the technical specifications and commercial viability of this synthesis route, we have compiled answers to common inquiries based on the experimental data provided in the patent documentation. These insights address critical aspects regarding substrate scope, reaction optimization, and product isolation, offering a transparent view into the process capabilities for potential partners evaluating this technology for their pipeline.
Q: What is the advantage of using TFBen over carbon monoxide gas?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a solid carbon monoxide surrogate, eliminating the need for high-pressure gas equipment and significantly improving operational safety and ease of handling in standard laboratory or plant settings.
Q: What is the role of Silver Oxide (Ag2O) in this reaction?
A: Silver oxide is added in the second stage of the reaction to promote the oxidative cyclization of the intermediate amide, driving the formation of the final N-acyl indole ring structure efficiently under mild thermal conditions.
Q: Is this method compatible with halogenated substrates?
A: Yes, the patent data demonstrates excellent substrate compatibility, successfully tolerating various functional groups including fluoro, chloro, and bromo substituents on both the aniline and aryl iodide components without significant yield loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines and optimizing production costs for our global clientele. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity N-acyl indole intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to leverage this cutting-edge palladium-catalyzed technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can drive value and efficiency in your supply chain.
