Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Production
Advanced Palladium-Catalyzed Carbonylation for Scalable N-Acyl Indole Production
The synthesis of complex heterocyclic scaffolds remains a cornerstone of modern medicinal chemistry, particularly for the development of potent therapeutic agents. A recent technological breakthrough detailed in patent CN112898192B introduces a highly efficient and operationally simple method for preparing N-acyl indole compounds. This innovation addresses critical bottlenecks in traditional synthetic routes by utilizing a palladium-catalyzed carbonylative cyclization strategy that employs safe, solid carbon monoxide surrogates. Indole derivatives are ubiquitous in pharmacology, serving as the structural backbone for a wide array of bioactive molecules including anti-inflammatory, anti-tumor, and anti-viral drugs, as illustrated by prominent examples such as Indomethacin and Delavirdine.
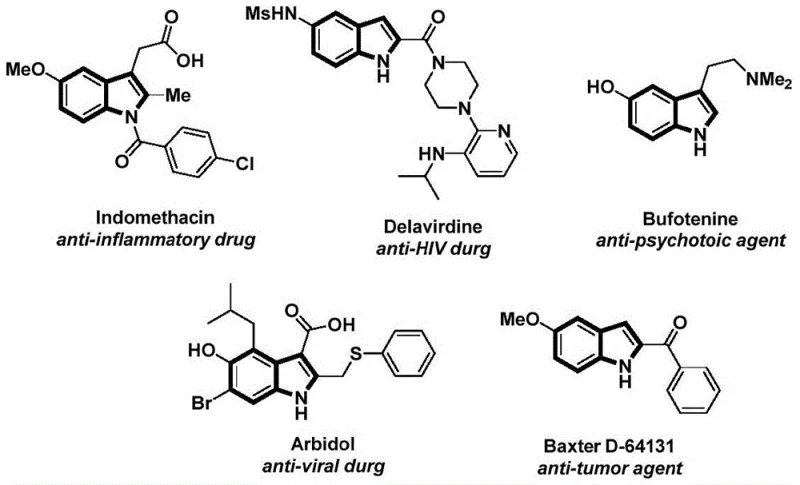
This novel methodology not only streamlines the construction of the indole core but also integrates the acylation step seamlessly, offering a distinct advantage for process chemists aiming to reduce step counts. By leveraging 2-alkynyl anilines and aryl iodides as readily available starting materials, the process achieves high atom economy and excellent substrate compatibility. For R&D directors and procurement specialists in the pharmaceutical sector, this represents a significant opportunity to optimize the supply chain for high-value intermediates, ensuring both cost-effectiveness and regulatory compliance through safer processing conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of N-acyl indoles via carbonylation has been fraught with significant operational hazards and technical complexities. Conventional carbonylation reactions typically rely on the use of carbon monoxide (CO) gas, which is highly toxic, flammable, and requires specialized high-pressure equipment to handle safely. This necessity for pressurized reactors imposes substantial capital expenditure and strict safety protocols, often limiting the feasibility of these reactions in standard laboratory or pilot plant settings. Furthermore, controlling the stoichiometry of gaseous CO can be challenging, leading to inconsistent reaction rates and potential side reactions that complicate downstream purification. The reliance on harsh conditions often results in poor functional group tolerance, restricting the scope of substrates that can be effectively utilized without extensive protection-deprotection sequences.
The Novel Approach
In stark contrast, the method disclosed in CN112898192B revolutionizes this landscape by replacing hazardous CO gas with 1,3,5-tricarboxylic acid phenol ester (TFBen), a stable and easy-to-handle solid surrogate. This substitution fundamentally alters the risk profile of the synthesis, allowing the reaction to proceed under atmospheric pressure in standard glassware or stainless steel reactors without the need for specialized gas handling infrastructure. The process operates at a moderate temperature of 60°C, which is energy-efficient and gentle enough to preserve sensitive functional groups on the aromatic rings. By integrating the carbonylation and cyclization into a streamlined one-pot sequence, this approach drastically reduces solvent consumption and waste generation, aligning perfectly with green chemistry principles while delivering high-purity products suitable for pharmaceutical applications.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The elegance of this synthesis lies in its sophisticated yet robust catalytic cycle, which orchestrates multiple bond-forming events with high precision. The reaction initiates with the oxidative addition of the palladium(0) catalyst into the carbon-iodine bond of the aryl iodide substrate, generating a reactive aryl-palladium(II) intermediate. Subsequently, carbon monoxide is released in situ from the decomposition of TFBen and inserts into the palladium-carbon bond, forming an acyl-palladium species. This key intermediate then undergoes nucleophilic attack by the amine group of the 2-alkynyl aniline, followed by reductive elimination to yield an amide precursor. The presence of silver oxide in the second stage is critical, as it promotes the intramolecular cyclization of this amide intermediate, ultimately forging the N-acyl indole skeleton with high regioselectivity.
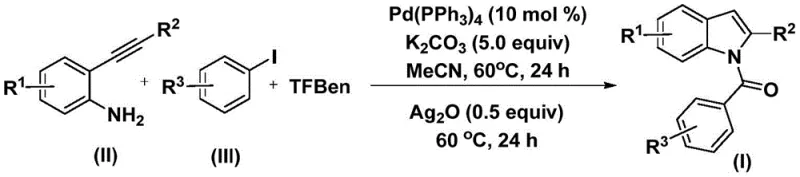
From an impurity control perspective, the use of TFBen ensures a steady and controlled release of carbon monoxide, preventing the accumulation of unreacted gas that could lead to over-carbonylation or polymerization side products. The mild basic conditions provided by potassium carbonate facilitate the deprotonation steps necessary for cyclization without inducing hydrolysis of the sensitive acyl groups. Furthermore, the choice of acetonitrile as the solvent optimizes the solubility of both organic substrates and inorganic bases, ensuring a homogeneous reaction environment that maximizes conversion rates. This mechanistic clarity allows process engineers to fine-tune parameters such as catalyst loading and reaction time to achieve optimal yields, typically ranging from moderate to excellent depending on the electronic nature of the substituents.
How to Synthesize N-Acyl Indole Compounds Efficiently
Implementing this synthesis protocol requires careful attention to reagent quality and reaction sequencing to maximize yield and purity. The process is designed to be scalable, moving seamlessly from milligram-scale discovery to kilogram-scale production with minimal modification. The standardized procedure involves a two-stage heating protocol where the initial coupling is followed by an oxidative cyclization, ensuring complete conversion of the starting materials. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Combine palladium catalyst (Pd(PPh3)4), potassium carbonate, 1,3,5-tricarboxylic acid phenol ester (TFBen), 2-alkynyl aniline, and aryl iodide in acetonitrile solvent.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and coupling steps.
- Add silver oxide (Ag2O) to the mixture and continue heating at 60°C for an additional 24 hours to complete the cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits that extend beyond mere chemical yield. The shift from gaseous reagents to solid surrogates fundamentally de-risks the manufacturing process, reducing the likelihood of production stoppages due to safety incidents or regulatory inspections related to hazardous gas storage. This enhanced operational stability ensures a more predictable delivery schedule for critical intermediates, allowing downstream API manufacturers to maintain leaner inventory levels without compromising on security of supply. Additionally, the simplicity of the workup procedure, which involves basic filtration and chromatography, reduces the demand for specialized labor and complex separation equipment, further driving down the overall cost of goods sold.
- Cost Reduction in Manufacturing: The elimination of high-pressure reactors and specialized CO handling systems results in significant capital expenditure savings and lower maintenance costs. By utilizing commercially available and inexpensive reagents like aryl iodides and TFBen, the raw material cost profile is optimized, making the final N-acyl indole products highly competitive in the global market. The high atom economy of the reaction minimizes waste disposal costs, contributing to a more sustainable and economically viable production model.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable solid reagents mitigates the risks associated with the transportation and storage of hazardous gases, which are often subject to strict logistical regulations. This flexibility allows for a more agile supply chain capable of responding rapidly to fluctuations in demand. The broad substrate scope demonstrated in the patent means that a single production line can be adapted to synthesize a diverse library of indole derivatives, enhancing the versatility of the manufacturing asset.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic gas emissions align perfectly with increasingly stringent environmental regulations. Scaling this process from bench to plant does not require exponential increases in safety infrastructure, facilitating a smoother technology transfer. The reduced solvent usage and simplified purification steps lower the environmental footprint, supporting corporate sustainability goals and ensuring long-term regulatory compliance in major pharmaceutical markets.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into existing workflows. The following questions address common inquiries regarding the practical application, safety profile, and scalability of this patented method. These insights are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for implementation.
Q: What is the primary advantage of using TFBen over carbon monoxide gas in this synthesis?
A: Using 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide surrogate eliminates the need for handling hazardous high-pressure CO gas, significantly improving operational safety and simplifying equipment requirements for industrial scale-up.
Q: What represents the typical yield range for this palladium-catalyzed cyclization?
A: According to the experimental data in the patent, the reaction demonstrates robust efficiency with isolated yields ranging from 44% to 82% across various substrates, indicating good tolerance for different functional groups like halogens and alkoxy groups.
Q: Why is silver oxide added in the second stage of the reaction?
A: Silver oxide acts as a crucial promoter in the second stage, facilitating the oxidative cyclization of the intermediate amide to form the final N-acyl indole core structure under mild thermal conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN112898192B for accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale innovation to market-ready product is seamless. Our state-of-the-art facilities are equipped to handle complex palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this efficient route can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive edge in the global pharmaceutical supply chain.
