Advanced Steroid Deacetoxylation: Scaling High-Purity Intermediates for Global Pharma
Advanced Steroid Deacetoxylation: Scaling High-Purity Intermediates for Global Pharma
The synthesis of complex steroid pharmaceuticals often hinges on the efficient manipulation of the D-ring side chain, particularly the removal of protecting groups to reveal active functionalities. A pivotal advancement in this domain is documented in Chinese Patent CN110407902B, which discloses a highly efficient method for removing the 17-acetoxy group from steroid compounds. This transformation is critical because the resulting sterene compounds serve as indispensable intermediates in the production of various corticosteroids and hormonal therapies. The patent addresses long-standing inefficiencies in traditional elimination protocols by introducing a novel catalytic system that utilizes specific organic bases in conjunction with low-boiling point solvents. This technological shift not only enhances the chemical yield of the reaction but also fundamentally alters the economic and environmental profile of the manufacturing process, making it exceptionally suitable for large-scale industrial applications where solvent recovery and energy consumption are paramount concerns for supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the deacetoxylation of 17α-acetoxy steroids has been performed using inorganic bases such as potassium acetate dissolved in high-boiling polar aprotic solvents like dimethylformamide (DMF) or dimethyl sulfoxide (DMSO). While chemically feasible, this conventional route presents severe logistical and economic bottlenecks for commercial manufacturing. The primary drawback lies in the physical properties of the solvents; DMF and DMSO possess extremely high boiling points, making their removal from the reaction mixture energy-intensive and time-consuming. Furthermore, these solvents are notoriously difficult to recycle efficiently, often leading to significant waste generation and increased disposal costs. From a purity perspective, the harsh conditions sometimes required to drive the reaction to completion in these viscous media can lead to the formation of stubborn impurities that complicate downstream purification. The reliance on such problematic solvents creates a fragile supply chain link, as any disruption in solvent availability or tightening of environmental regulations regarding volatile organic compound (VOC) emissions can halt production lines.
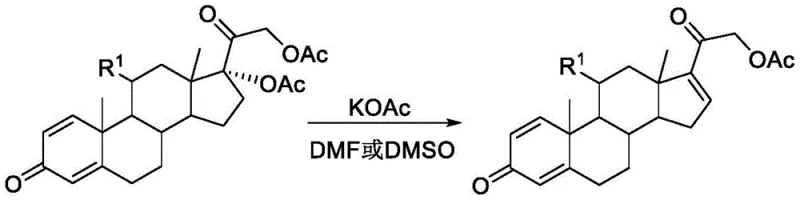
The Novel Approach
In stark contrast to the legacy methods, the technology described in CN110407902B introduces a paradigm shift by employing strong organic bases, specifically bicyclic guanidine derivatives such as TBD (1,5,7-triazabicyclo[4.4.0]dec-5-ene) or MTBD (7-methyl-TBD), within low-boiling solvent systems. This innovative combination allows the elimination reaction to proceed under much milder thermal conditions while achieving superior conversion rates. By shifting the solvent landscape to options like tetrahydrofuran (THF), dioxane, or even aromatic hydrocarbons, the process dramatically simplifies the work-up procedure. These low-boiling solvents can be easily stripped off under reduced pressure at lower temperatures, significantly reducing the thermal load on the product and minimizing the risk of degradation. Moreover, the ease of distillation facilitates high-efficiency solvent recovery loops, turning a waste stream into a reusable asset. This approach effectively decouples the reaction efficiency from the use of hazardous, high-boiling media, offering a cleaner, faster, and more economically robust pathway for generating high-purity steroid intermediates.
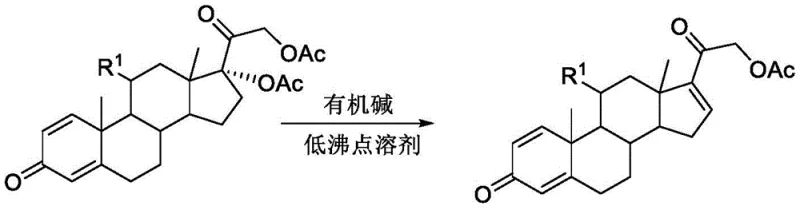
Mechanistic Insights into Organic Base Catalyzed Elimination
The core of this technological breakthrough lies in the unique basicity and steric profile of the chosen organic catalysts. Unlike potassium acetate, which acts as a relatively weak base requiring high temperatures and polar stabilization to function effectively, organic super-bases like TBD and MTBD possess exceptional proton affinity. In the context of the steroid skeleton, these bases facilitate an E2-type elimination mechanism where the base abstracts the acidic proton at the C16 position. This abstraction occurs concurrently with the departure of the acetoxy leaving group at the C17 position, resulting in the formation of the desired C16-C17 double bond. The non-nucleophilic nature of these bulky guanidine bases is crucial, as it prevents unwanted side reactions such as nucleophilic attack on the carbonyl groups present elsewhere in the steroid framework, thereby preserving the integrity of the molecule. This selectivity ensures that the reaction trajectory is funneled almost exclusively towards the desired elimination product, minimizing the generation of regio-isomers or addition by-products that would otherwise burden the purification team.
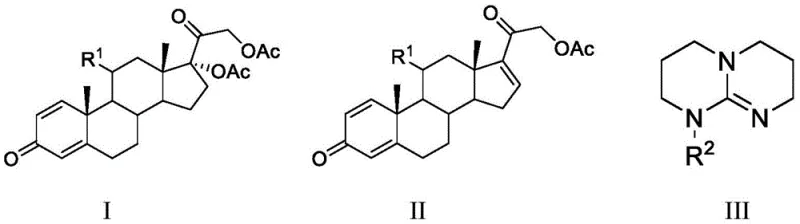
Furthermore, the choice of solvent plays a synergistic role in this mechanistic pathway. In traditional DMF/DMSO systems, the high polarity stabilizes the transition state but also solvates the base too strongly, potentially reducing its effective reactivity. In the new low-boiling solvent systems like THF or dioxane, the solvation shell around the organic base is less restrictive, allowing for greater kinetic freedom and faster reaction rates. The patent data indicates that maintaining a molar ratio of substrate to base between 1:2 and 1:3 optimizes this interaction, ensuring that there is sufficient base to drive the equilibrium forward without excessive reagent waste. This precise control over the reaction environment allows manufacturers to achieve yields as high as 95%, as demonstrated in the experimental examples, representing a substantial improvement over the often variable and lower yields associated with inorganic base protocols. The result is a process that is not only chemically elegant but also rigorously controlled, providing the consistency required for GMP manufacturing environments.
How to Synthesize 17-Deacetoxy Steroid Compounds Efficiently
Implementing this novel deacetoxylation protocol requires careful attention to reagent quality and atmospheric control to maximize the benefits of the organic base catalysis. The process begins with the dissolution of the 17-acetoxy steroid precursor in a dry, low-boiling solvent such as tetrahydrofuran or dioxane, typically under an inert nitrogen atmosphere to prevent moisture interference which could deactivate the sensitive organic base. Once the substrate is fully solubilized, the organic base (Structure III) is introduced, and the mixture is heated to reflux. The progress of the reaction is conveniently monitored via thin-layer chromatography (TLC), allowing operators to quench the reaction immediately upon completion to prevent over-reaction. Following the reaction, a simple acidic quench followed by extraction and solvent removal yields the target sterene compound with high purity. For a comprehensive understanding of the specific operational parameters and safety considerations, the detailed standardized synthesis steps are outlined in the guide below.
- Dissolve the 17-acetoxy steroid substrate (Structure I) in a low-boiling solvent such as tetrahydrofuran (THF) or dioxane under inert atmosphere.
- Add a stoichiometric amount of a strong organic base, specifically MTBD or TBD (Structure III), to the reaction mixture.
- Heat the mixture to reflux temperature until TLC monitoring confirms complete consumption of the starting material, then quench and isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from the conventional DMF-based process to this new organic base-mediated method represents a strategic opportunity to optimize operational expenditures and mitigate supply risks. The most immediate impact is observed in the realm of utility costs and waste management. By eliminating the need for high-boiling solvents that require extensive energy input for removal and specialized treatment for disposal, the overall cost of goods sold (COGS) for the intermediate is significantly reduced. The ability to recover and reuse low-boiling solvents like THF or dioxane through standard distillation infrastructure further amplifies these savings, creating a circular economy within the production facility that minimizes raw material consumption. Additionally, the higher reaction yields reported in the patent mean that less starting material is required to produce the same amount of final product, directly lowering the demand on upstream supply chains and reducing the inventory burden on the warehouse.
- Cost Reduction in Manufacturing: The substitution of expensive and difficult-to-handle high-boiling solvents with commodity low-boiling solvents drastically simplifies the downstream processing workflow. Since solvents like THF and dioxane have much lower boiling points than DMF or DMSO, the energy required for solvent stripping and product drying is substantially lower, leading to direct reductions in utility bills. Furthermore, the improved yield efficiency means that the effective cost per kilogram of the active intermediate decreases, as fewer batches are needed to meet production targets, and the loss of valuable steroid starting material is minimized. This cumulative effect results in a leaner manufacturing process with a healthier margin profile, allowing for more competitive pricing in the global market without sacrificing quality standards.
- Enhanced Supply Chain Reliability: Relying on traditional solvents like DMSO can sometimes introduce bottlenecks due to their classification as reproductive toxins in certain jurisdictions, which complicates logistics and storage. By shifting to widely available industrial solvents like toluene, THF, or dioxane, the supply chain becomes more resilient and less susceptible to regulatory shocks. The use of robust organic bases that are commercially available in bulk quantities further secures the supply line. This flexibility ensures that production schedules can be maintained consistently, reducing the lead time for high-purity pharmaceutical intermediates and enabling manufacturers to respond more agilely to fluctuating market demands from downstream API producers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this new method offers a clearer path to compliance with increasingly stringent global environmental regulations. The reduction in hazardous waste volume, driven by the elimination of persistent high-boiling solvents, lowers the facility's environmental footprint and reduces the costs associated with waste disposal permits and treatments. The process is inherently safer to scale because the lower operating temperatures and pressures associated with low-boiling solvents reduce the thermal risk profile of the reactor. This makes the technology ideal for commercial scale-up of complex steroid intermediates, allowing facilities to increase batch sizes confidently while maintaining strict adherence to green chemistry principles and corporate sustainability goals.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of adopting this deacetoxylation technology, we have compiled answers to common inquiries based on the specific technical disclosures found in the patent literature. These questions address the critical variables of reagent selection, solvent compatibility, and process optimization that are essential for successful technology transfer. Understanding these nuances is vital for R&D directors planning pilot runs and for quality assurance teams establishing specification limits for the new process. The following insights are derived directly from the experimental data and claims presented in the intellectual property documentation.
Q: What are the primary advantages of using organic bases over potassium acetate in this elimination reaction?
A: According to patent CN110407902B, organic bases like MTBD and TBD provide superior catalytic activity compared to traditional potassium acetate. This results in significantly higher reaction yields (up to 95% in examples) and allows the use of low-boiling solvents, which simplifies downstream processing.
Q: Which solvents are recommended for this deacetoxylation process to ensure industrial viability?
A: The patent explicitly recommends replacing high-boiling, difficult-to-remove solvents like DMF and DMSO with low-boiling alternatives. Preferred solvents include tetrahydrofuran (THF), dioxane, halogenated hydrocarbons, or aromatic hydrocarbons, which facilitate easier solvent recovery and reduce energy consumption during concentration.
Q: What is the optimal molar ratio of substrate to organic base for maximum efficiency?
A: Experimental data within the patent indicates that a molar ratio of substrate to organic base between 1:1 and 1:5 is effective. However, for optimal balance between cost and conversion, a ratio of 1:2 to 1:3 is preferred to ensure complete elimination of the 17-acetoxy group.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Steroid Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic methodologies like the one described in CN110407902B requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our facilities are equipped with state-of-the-art reactors capable of handling sensitive organic base chemistries under strictly controlled inert atmospheres, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of steroid intermediate meets the exacting standards of the global pharmaceutical industry. We are committed to delivering not just a chemical product, but a reliable supply solution that integrates cutting-edge process chemistry with operational excellence.
We invite you to leverage our technical proficiency to optimize your supply chain for steroid drug development. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements, demonstrating how this novel elimination route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data for our steroid portfolio and to discuss route feasibility assessments for your upcoming campaigns. Let us collaborate to bring your next-generation steroid therapies to market faster and more efficiently.
