Advanced Palladium-Catalyzed Bis-Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrole-2-Ketone Production
Introduction to Next-Generation Pyrrolinone Synthesis
The structural motif of 1,5-dihydro-2H-pyrrole-2-ketone serves as a critical pharmacophore found in a diverse array of bioactive natural products and therapeutic agents. As illustrated in the chemical landscape, this core scaffold is integral to molecules such as Althiomycin, which exhibits potent antibacterial properties, Glimepiride, a widely prescribed hypoglycemic agent for diabetes management, and Isomalyngamide A, known for its potential anticancer activity. 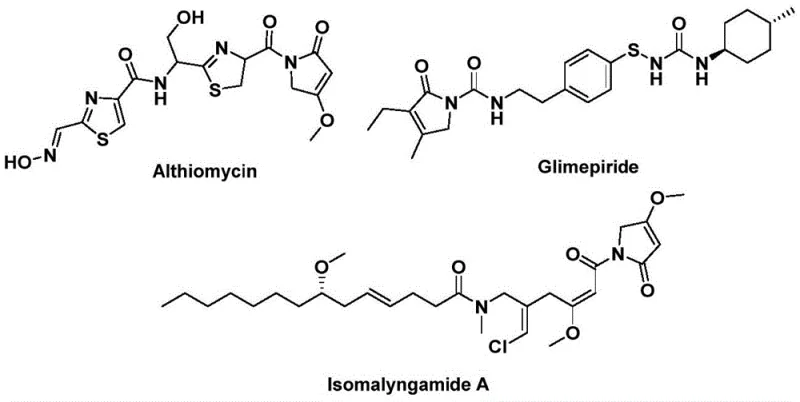 . Recognizing the immense pharmaceutical value of this heterocyclic system, recent advancements detailed in patent CN112694430B have introduced a transformative preparation method that addresses long-standing synthetic challenges. This novel approach leverages a palladium-catalyzed bis-carbonylation strategy, utilizing readily available propargyl amines and benzyl chlorides as starting materials to construct the complex five-membered lactam ring in a single operational step. For research and development teams seeking reliable pharmaceutical intermediate suppliers, this technology represents a significant leap forward in accessing high-value scaffolds with improved efficiency and reduced environmental footprint compared to traditional multi-step sequences.
. Recognizing the immense pharmaceutical value of this heterocyclic system, recent advancements detailed in patent CN112694430B have introduced a transformative preparation method that addresses long-standing synthetic challenges. This novel approach leverages a palladium-catalyzed bis-carbonylation strategy, utilizing readily available propargyl amines and benzyl chlorides as starting materials to construct the complex five-membered lactam ring in a single operational step. For research and development teams seeking reliable pharmaceutical intermediate suppliers, this technology represents a significant leap forward in accessing high-value scaffolds with improved efficiency and reduced environmental footprint compared to traditional multi-step sequences.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,5-dihydro-2H-pyrrol-2-one skeleton has relied on methodologies that often suffer from significant operational drawbacks and limited substrate scope. Traditional routes frequently necessitate the use of hazardous gaseous carbon monoxide under high pressure, requiring specialized autoclaves and stringent safety protocols that complicate scale-up and increase capital expenditure. Furthermore, many existing synthetic pathways involve multiple discrete steps, including separate protection and deprotection sequences, which inevitably lead to cumulative yield losses and generate substantial quantities of chemical waste. The reliance on stoichiometric organometallic reagents or harsh cyclization conditions can also result in poor functional group tolerance, limiting the diversity of derivatives that can be accessed for structure-activity relationship (SAR) studies. These inefficiencies create bottlenecks in the supply chain for high-purity pharmaceutical intermediates, driving up costs and extending lead times for drug development programs that depend on these critical building blocks.
The Novel Approach
In stark contrast to these legacy methods, the invention disclosed in patent CN112694430B offers a streamlined, one-pot solution that dramatically simplifies the manufacturing process. By employing a solid carbon monoxide surrogate, specifically 1,3,5-tricarboxylic acid phenol ester (TFBen), the new protocol obviates the need for handling toxic CO gas, thereby enhancing workplace safety and reducing regulatory burdens associated with high-pressure operations. The reaction proceeds efficiently in acetonitrile at moderate temperatures ranging from 100°C to 120°C, utilizing a robust palladium catalytic system that demonstrates exceptional compatibility with a wide array of functional groups. This methodology allows for the direct coupling of substituted propargyl amines and benzyl chlorides, facilitating the rapid assembly of diverse pyrrolinone libraries with high atom economy. For procurement managers focused on cost reduction in API manufacturing, this shift towards safer, telescoped processes translates directly into lower operational costs and a more resilient supply chain capable of meeting demanding production schedules without compromising on quality or purity standards.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation
The mechanistic pathway underpinning this transformation is a sophisticated example of transition metal-catalyzed carbonylative cyclization, driven by the unique reactivity of the palladium center. The catalytic cycle initiates with the oxidative addition of the active palladium(0) species into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzyl-palladium intermediate. Subsequently, carbon monoxide, which is liberated in situ from the thermal decomposition of the phenol 1,3,5-tricarboxylate surrogate, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the propargyl amine, triggering an intramolecular cyclization event that constructs the five-membered nitrogen-containing ring. 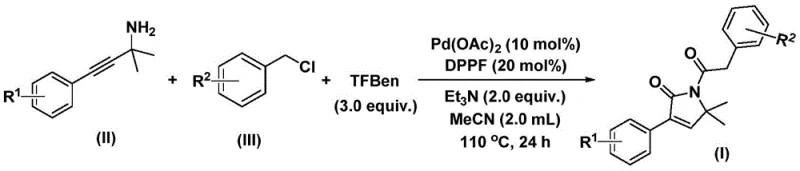 . Following the initial ring closure, a second molecule of carbon monoxide inserts into the newly formed organopalladium bond, expanding the coordination sphere before a final reductive elimination step releases the target 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active catalyst. This dual-carbonylation sequence is meticulously controlled by the choice of ligand, typically 1,1'-bis(diphenylphosphino)ferrocene (DPPP), which stabilizes the palladium center and facilitates the sequential insertion events necessary for forming the complex lactam architecture.
. Following the initial ring closure, a second molecule of carbon monoxide inserts into the newly formed organopalladium bond, expanding the coordination sphere before a final reductive elimination step releases the target 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active catalyst. This dual-carbonylation sequence is meticulously controlled by the choice of ligand, typically 1,1'-bis(diphenylphosphino)ferrocene (DPPP), which stabilizes the palladium center and facilitates the sequential insertion events necessary for forming the complex lactam architecture.
From an impurity control perspective, the use of a solid CO source provides a distinct advantage by ensuring a steady, low-concentration release of carbon monoxide throughout the reaction duration. This controlled release mechanism minimizes the formation of side products often associated with excess CO pressure, such as over-carbonylated species or polymerization byproducts. Furthermore, the mild basic conditions provided by triethylamine help to neutralize the hydrochloric acid byproduct generated during the oxidative addition step, preventing acid-catalyzed degradation of the sensitive pyrrolinone ring or the starting amine. The high selectivity of this catalytic system ensures that the final crude mixture contains predominantly the desired regioisomer, significantly simplifying downstream purification efforts. For quality assurance teams, this inherent selectivity means that achieving stringent purity specifications requires less aggressive chromatographic separation, preserving overall yield and reducing the consumption of silica gel and solvents during the isolation phase.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-Ketone Efficiently
The practical implementation of this synthesis is designed for ease of execution in standard laboratory and pilot plant settings, requiring only common glassware and heating equipment. The process begins by charging a reaction vessel with the palladium catalyst, the bidentate phosphine ligand, the solid CO surrogate, the base, and the two primary organic substrates dissolved in acetonitrile. The mixture is then heated to the optimized temperature range and stirred for a period of 24 to 48 hours, allowing the bis-carbonylation cascade to proceed to completion. Detailed standardized operating procedures regarding specific molar ratios, workup techniques, and purification parameters are essential for reproducibility and are outlined below for technical reference.
- Combine palladium acetate, DPPP ligand, triethylamine, solid CO surrogate (TFBen), propargyl amine, and benzyl chloride in acetonitrile solvent within a sealed vessel.
- Heat the reaction mixture to a temperature range of 100-120°C and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target pyrrol-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel synthetic route offers profound strategic benefits for organizations managing the procurement of complex heterocyclic intermediates. The shift from hazardous gaseous reagents to stable solid surrogates fundamentally alters the risk profile of the manufacturing process, allowing for production in facilities that may not be equipped for high-pressure gas handling. This flexibility expands the pool of potential contract manufacturing partners and reduces the logistical complexity associated with transporting dangerous goods. Moreover, the use of commercially available starting materials, such as substituted benzyl chlorides and propargyl amines, ensures a stable and competitive supply base, mitigating the risk of raw material shortages that can plague specialty chemical markets. The robustness of the reaction conditions further enhances supply chain reliability by minimizing batch-to-batch variability and reducing the likelihood of costly campaign failures due to sensitive reaction parameters.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the telescoping of multiple synthetic steps into a single pot operation. By eliminating the need for intermediate isolation, purification, and the associated solvent exchanges, the overall processing time and labor costs are drastically reduced. Additionally, the avoidance of high-pressure equipment lowers capital depreciation costs and maintenance expenses, while the use of a solid CO surrogate removes the need for expensive gas containment infrastructure. The high reaction efficiency observed across a broad substrate scope means that valuable starting materials are converted into product with minimal waste, optimizing the cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like acetonitrile, triethylamine, and simple benzyl halides ensures that the supply chain is not dependent on exotic or single-source reagents. This diversification of raw material sources protects against market volatility and geopolitical disruptions that can impact the availability of specialized precursors. Furthermore, the operational simplicity of the reaction allows for rapid scale-up from gram to kilogram scales without the need for extensive process re-engineering, ensuring that supply can be ramped up quickly to meet sudden increases in demand from downstream pharmaceutical clients.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this methodology aligns perfectly with modern green chemistry principles. The replacement of toxic carbon monoxide gas with a benign solid precursor significantly reduces the potential for accidental exposure and environmental release. The high atom economy of the bis-carbonylation reaction minimizes the generation of chemical waste, lowering the costs associated with waste disposal and treatment. As regulatory pressures on pharmaceutical manufacturers continue to intensify, adopting such inherently safer and cleaner processes provides a competitive edge by simplifying regulatory filings and demonstrating a commitment to sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and optimization studies presented in the patent literature, providing clarity on reaction parameters and substrate limitations. Understanding these nuances is crucial for process chemists evaluating the feasibility of this route for specific target molecules.
Q: What is the primary advantage of using TFBen over gaseous CO in this synthesis?
A: Using 1,3,5-trimethoxybenzene phenol ester (TFBen) as a solid carbon monoxide surrogate eliminates the need for high-pressure gas cylinders and specialized equipment, significantly enhancing operational safety and simplifying the reaction setup for scale-up.
Q: Does this protocol tolerate electron-withdrawing groups on the benzyl chloride substrate?
A: Yes, the methodology demonstrates excellent substrate compatibility, successfully accommodating various functional groups including halogens (F, Cl, Br), trifluoromethyl groups, and cyano groups without compromising reaction efficiency or yield.
Q: What are the typical reaction conditions required for optimal conversion?
A: Optimal conversion is achieved using acetonitrile as the solvent at temperatures between 110°C and 120°C for approximately 24 hours, utilizing a catalytic system of Pd(OAc)2 and DPPP ligand with triethylamine as the base.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your project from early-stage discovery through to full-scale manufacturing. We are committed to delivering products that meet stringent purity specifications, backed by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced catalytic technologies like the Pd-catalyzed bis-carbonylation described herein, we offer our partners a distinct advantage in terms of speed to market and cost efficiency.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific project requirements. Whether you need a Customized Cost-Saving Analysis for an existing route or require specific COA data and route feasibility assessments for a new target, our experts are ready to provide the support you need. Contact us today to explore how our innovative synthesis platforms can accelerate your drug development timeline and optimize your supply chain performance.
