Advanced Palladium-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrole-2-Ketone Intermediates for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrole-2-Ketone Intermediates for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex heterocyclic scaffolds that serve as critical backbones for bioactive molecules. A recent technological breakthrough detailed in patent CN112694430B introduces a highly efficient preparation method for 1,5-dihydro-2H-pyrrole-2-ketone compounds, a structural motif prevalent in numerous natural products and therapeutic agents. This innovation addresses long-standing challenges in carbonylation chemistry by replacing hazardous gaseous carbon monoxide with a solid surrogate, thereby streamlining the synthetic workflow. The significance of this scaffold cannot be overstated, as it appears in potent antibiotics like Althiomycin, hypoglycemic agents such as Glimepiride, and anticancer candidates like Isomalyngamide A, highlighting its versatility across diverse therapeutic areas.
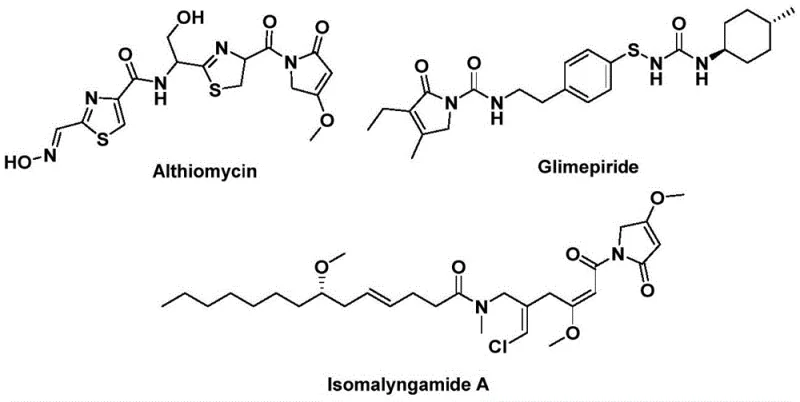
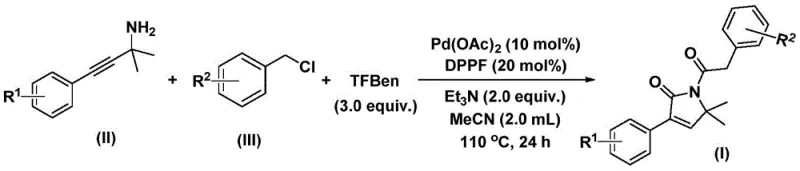
For R&D directors and process chemists, the ability to access these cores reliably is paramount for drug discovery pipelines. The disclosed method leverages a palladium-catalyzed bis-carbonylation strategy that couples readily available propargylamines and benzyl chlorides in a single operational step. By utilizing 1,3,5-tricarboxylic acid phenol ester (TFBen) as an ex situ source of carbon monoxide, the process mitigates the severe safety risks associated with traditional high-pressure CO gas reactions. This shift not only enhances laboratory safety but also simplifies the engineering requirements for scale-up, making it an attractive option for reliable pharmaceutical intermediate supplier networks looking to diversify their catalog with high-value heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles has relied heavily on direct carbonylation using carbon monoxide gas, a reagent that poses significant logistical and safety hurdles in both academic and industrial settings. The requirement for specialized high-pressure equipment, such as autoclaves capable of withstanding substantial PSI, creates a barrier to entry for many contract research organizations and limits the throughput of parallel synthesis campaigns. Furthermore, the toxicity of CO gas necessitates rigorous monitoring systems and specialized ventilation infrastructure, driving up capital expenditure and operational costs. Traditional methods often suffer from poor atom economy or require harsh reaction conditions that limit substrate scope, particularly when sensitive functional groups are present on the starting materials. These factors collectively contribute to extended lead times and increased complexity in the supply chain for critical API intermediates.
The Novel Approach
The methodology described in CN112694430B represents a paradigm shift by employing a solid carbon monoxide surrogate, TFBen, which releases CO in situ under thermal conditions. This approach allows the reaction to proceed in standard glassware, such as Schlenk tubes, at atmospheric pressure, effectively democratizing access to carbonylation chemistry. The reaction system utilizes a palladium catalyst paired with a bidentate phosphine ligand (DPPP) to facilitate the insertion of two carbonyl units into the substrate framework efficiently. This one-pot transformation converts simple propargylamines and benzyl chlorides directly into the desired 1,5-dihydro-2H-pyrrole-2-ketones with remarkable efficiency. The operational simplicity means that cost reduction in pharmaceutical intermediate manufacturing is achievable through reduced equipment overhead and safer handling protocols, while maintaining high chemical fidelity.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
Understanding the catalytic cycle is essential for optimizing this process for commercial production. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride, generating a reactive benzylpalladium intermediate. Subsequently, carbon monoxide, liberated thermally from the TFBen precursor, inserts into the palladium-carbon bond to form an acylpalladium species. This acyl intermediate then undergoes nucleophilic attack or coordination with the propargylamine component, leading to the formation of a five-membered ring palladium complex. A second insertion of carbon monoxide follows, expanding the coordination sphere to a six-membered ring palladium intermediate, which ultimately undergoes reductive elimination to release the final 1,5-dihydro-2H-pyrrole-2-ketone product and regenerate the active catalyst.
From an impurity control perspective, the use of a solid CO source provides a steady, controlled release of carbon monoxide, preventing local concentration spikes that could lead to side reactions or polymerization. The choice of acetonitrile as the solvent further enhances solubility and stability of the polar intermediates involved in the cycle. The tolerance for various substituents (R1 and R2) including halogens, alkyl groups, and trifluoromethyl moieties suggests that the electronic properties of the substrates do not drastically inhibit the catalytic turnover. This robustness ensures that the high-purity pharmaceutical intermediates produced meet stringent quality standards required for downstream drug synthesis, minimizing the burden on purification teams.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-Ketone Efficiently
The experimental protocol outlined in the patent provides a clear roadmap for reproducing these high-yielding transformations. The procedure involves charging a reaction vessel with precise molar ratios of palladium acetate, ligand, base, and the solid CO source, followed by the addition of the organic substrates in acetonitrile. Heating the mixture to 110°C for 24 hours drives the reaction to completion, after which standard workup procedures involving filtration and silica gel treatment are employed. For detailed operational parameters and specific stoichiometric adjustments for different substrates, please refer to the standardized synthesis steps provided below.
- Combine palladium acetate, DPPP ligand, triethylamine, TFBen (CO source), propargylamine, and benzyl chloride in acetonitrile solvent within a Schlenk tube.
- Heat the reaction mixture to 110°C and stir continuously for 24 hours to ensure complete conversion via the bis-carbonylation pathway.
- Filter the reaction mixture, mix with silica gel, and purify the crude product using column chromatography to isolate the target 1,5-dihydro-2H-pyrrole-2-ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from gas-phase carbonylation to this solid-source methodology offers tangible strategic benefits beyond mere chemical yield. The elimination of high-pressure gas cylinders reduces the regulatory burden and insurance costs associated with storing hazardous materials on-site. Additionally, the starting materials—propargylamines and benzyl chlorides—are commodity chemicals that are widely available from global suppliers, ensuring a stable and resilient supply chain不受 geopolitical disruptions affecting specialty gases. The simplified reaction setup allows for easier technology transfer between sites, facilitating the commercial scale-up of complex pharmaceutical intermediates without the need for bespoke reactor engineering.
- Cost Reduction in Manufacturing: The replacement of expensive, specialized high-pressure reactors with standard heating mantles and glassware significantly lowers capital expenditure (CAPEX) for new production lines. Furthermore, the use of TFBen avoids the logistics costs of transporting and storing compressed toxic gases, leading to substantial operational savings. The high reaction efficiency, with yields reaching up to 92% for certain derivatives, minimizes raw material waste and reduces the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: By relying on solid reagents that are stable at room temperature, the risk of supply interruption due to gas cylinder shortages or transport regulations is virtually eliminated. The broad substrate scope means that a single platform technology can be used to generate a diverse library of analogs, allowing for rapid response to changing market demands or clinical trial requirements. This flexibility is crucial for reducing lead time for high-purity pharmaceutical intermediates in fast-paced drug development environments.
- Scalability and Environmental Compliance: The reaction operates in acetonitrile, a solvent that is easily recovered and recycled in industrial distillation columns, aligning with green chemistry principles. The absence of heavy metal contamination issues often associated with heterogeneous catalysts simplifies the purification process, reducing solvent consumption during chromatography. The mild conditions and high selectivity result in fewer byproducts, lowering the environmental footprint of the manufacturing process and easing compliance with increasingly strict waste disposal regulations.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the reaction scope, safety profile, and scalability. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, ensuring accuracy and relevance for process development decisions.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: This method utilizes 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide substitute, eliminating the need for handling toxic, high-pressure CO gas cylinders, thereby significantly enhancing operational safety in industrial settings.
Q: What is the substrate compatibility of this palladium-catalyzed reaction?
A: The process demonstrates excellent functional group tolerance, successfully accommodating substrates with electron-donating groups (like methoxy and methyl) and electron-withdrawing groups (such as fluoro, chloro, bromo, and trifluoromethyl) on both the propargylamine and benzyl chloride components.
Q: What are the typical reaction conditions required for high yield?
A: Optimal results are achieved using palladium acetate (10 mol%) and DPPP (20 mol%) in acetonitrile at 110°C for 24 hours, yielding products with efficiencies often exceeding 80% and reaching up to 92% for specific derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into industrial reality. We are committed to delivering stringent purity specifications for all our intermediates, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and potency.
We invite potential partners to engage with our technical procurement team to discuss how this novel palladium-catalyzed route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your project volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique molecular targets, ensuring a partnership built on transparency, quality, and innovation.
