Advanced Photocatalytic Synthesis of Fluoroalkyl Indolines for Commercial Scale-up
Advanced Photocatalytic Synthesis of Fluoroalkyl Indolines for Commercial Scale-up
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to introduce fluorine atoms into heterocyclic scaffolds, driven by the profound impact of fluorination on metabolic stability and lipophilicity. Patent CN111138338A discloses a groundbreaking synthesis method for photocatalytic fluoroalkyl indolines, utilizing visible light catalysis to achieve efficient cyclization and fluorination simultaneously. This technology represents a significant paradigm shift from traditional thermal methods, leveraging Blue LEDs as a clean energy source and Eosin Y Na as an inexpensive, metal-free photocatalyst. For R&D directors and process chemists, this approach offers a compelling alternative to transition metal-catalyzed routes, particularly for the production of high-purity pharmaceutical intermediates where residual metal limits are stringent. The method operates under remarkably mild conditions, typically at room temperature, utilizing a MeCN/H2O mixed solvent system that adheres to modern green chemistry standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluoroalkyl indolines has relied heavily on transition metal catalysis, often involving copper or palladium complexes that necessitate harsh reaction conditions. These conventional protocols frequently require high-temperature heating to overcome activation energy barriers, which inadvertently induces unwanted side reactions and decomposition of sensitive functional groups. Furthermore, the reliance on expensive heterocyclic fluorination reagents and specialized ligands significantly inflates the raw material costs, making large-scale manufacturing economically challenging. A critical bottleneck in these traditional pathways is the difficulty in removing trace transition metals from the final product, a step that often requires additional purification stages and increases the overall lead time. The use of toxic organic solvents in these processes also poses environmental hazards and complicates waste disposal, conflicting with the increasing regulatory pressure for sustainable manufacturing practices in the fine chemical sector.
The Novel Approach
In stark contrast, the novel photocatalytic method described in the patent utilizes visible light to drive the reaction at ambient temperatures, effectively eliminating the thermal stress on the substrate. By employing Eosin Y Na, a commercially available organic dye, the process avoids the introduction of heavy metals entirely, thereby simplifying the downstream purification workflow and ensuring a cleaner impurity profile. The reaction system is elegantly simple, requiring only standard laboratory equipment such as Schlenk flasks and Blue LED arrays, which facilitates easy translation from bench scale to pilot plant operations. The integration of water as a co-solvent not only reduces the consumption of volatile organic compounds but also enhances the safety profile of the reaction medium. This approach enables the efficient construction of the indoline core with diverse fluoroalkyl chains, as illustrated in the general reaction scheme below, demonstrating broad substrate compatibility and high yields.
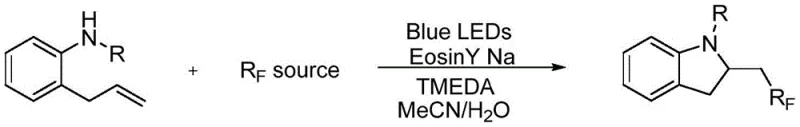
Mechanistic Insights into Visible-Light Photocatalytic Cyclization
The core of this innovation lies in the photoredox catalytic cycle initiated by the excitation of Eosin Y Na under blue light irradiation. Upon absorbing photons, the photocatalyst enters an excited state capable of engaging in single-electron transfer (SET) processes with the fluorinating reagent, generating reactive fluoroalkyl radicals. These radicals subsequently add to the terminal alkene of the 2-allylaniline substrate, initiating a cascade that leads to intramolecular cyclization and the formation of the indoline ring. The use of TMEDA as an additive plays a crucial role in modulating the reaction environment, likely assisting in the stabilization of intermediate species or facilitating proton transfer steps essential for the catalytic turnover. This radical-mediated pathway is distinct from ionic mechanisms found in traditional acid or base-catalyzed reactions, offering unique regioselectivity and chemoselectivity that are difficult to achieve otherwise. The mild nature of the radical generation ensures that sensitive functional groups on the aromatic ring or the nitrogen protecting group remain intact throughout the transformation.
From an impurity control perspective, the room temperature operation significantly suppresses thermal degradation pathways that often plague high-temperature syntheses. The absence of transition metals removes a major source of potential contamination, which is particularly advantageous for producing intermediates intended for oncology or CNS drugs where metal limits are parts-per-million. The reaction demonstrates excellent tolerance for various protecting groups on the nitrogen atom, including Ts, Cbz, and Boc, allowing for flexibility in synthetic planning. Moreover, the method accommodates a wide range of fluoroalkyl sources, from simple trifluoromethyl groups to longer perfluoroalkyl chains like heptafluoropropyl, expanding the chemical space accessible to medicinal chemists. The structural diversity achievable with this method is exemplified by the successful synthesis of complex derivatives, such as the perfluoroalkyl-substituted indoline shown below, highlighting the robustness of the catalytic system.
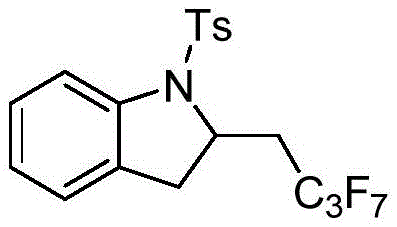
How to Synthesize Fluoroalkyl Indoline Efficiently
The operational simplicity of this photocatalytic protocol makes it highly attractive for process development teams aiming to establish robust manufacturing procedures. The synthesis begins with the preparation of the reaction mixture under an inert atmosphere to prevent oxygen quenching of the excited photocatalyst, followed by the sequential addition of substrates and reagents. Detailed standardized operating procedures regarding stoichiometry, light intensity, and workup protocols are critical for ensuring reproducibility and maximizing yield across different batches. The following guide outlines the fundamental steps derived from the patent examples, serving as a baseline for further optimization and scale-up studies in a GMP environment.
- Charge a reaction vessel with 2-allylaniline substrate, Eosin Y Na photocatalyst, and TMEDA base in a MeCN/H2O solvent mixture under nitrogen atmosphere.
- Add the fluorinating reagent (RF source) and irradiate the mixture with Blue LEDs (10W) at room temperature (5-30°C) while stirring until TLC indicates completion.
- Extract the product with DCM, dry over anhydrous magnesium sulfate, concentrate, and purify via column chromatography using petroleum ether/ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this photocatalytic technology offers tangible benefits in terms of cost structure and operational reliability. The shift away from precious metal catalysts and expensive ligands directly impacts the bill of materials, resulting in substantial cost savings on raw materials without compromising product quality. The use of commodity chemicals like Eosin Y and common solvents ensures a stable supply chain, mitigating the risks associated with sourcing specialized reagents that may have long lead times or limited availability. Furthermore, the energy efficiency of using LED light sources compared to prolonged heating cycles contributes to lower utility costs and a reduced carbon footprint for the manufacturing facility. These factors collectively enhance the economic viability of producing fluoroalkyl indolines, making them more accessible for inclusion in drug discovery pipelines and commercial formulations.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive metal scavenging resins and complex filtration steps, streamlining the production process. By utilizing visible light as the driving force, the process avoids the high energy consumption associated with maintaining elevated temperatures over extended periods. The use of water as a co-solvent reduces the volume of organic waste generated, lowering waste disposal costs and environmental compliance burdens. Overall, the simplified reaction setup and cheaper reagent profile contribute to a significantly reduced cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as 2-allylanilines and common fluorinating agents ensures a resilient supply chain less susceptible to geopolitical disruptions. The robustness of the reaction conditions allows for flexible scheduling and batch processing, enabling manufacturers to respond quickly to fluctuating demand from downstream clients. Additionally, the stability of the photocatalyst and reagents simplifies inventory management, as there is no need for specialized storage conditions often required for air-sensitive metal complexes. This reliability translates to consistent delivery schedules and reduced risk of production delays for key pharmaceutical intermediates.
- Scalability and Environmental Compliance: The photochemical nature of the reaction is inherently scalable through the use of flow chemistry or multi-lamp reactor arrays, facilitating the transition from gram-scale optimization to kilogram-scale production. The adherence to green chemistry principles, specifically the use of aqueous solvents and metal-free catalysis, aligns with increasingly strict environmental regulations and corporate sustainability goals. Simplified post-treatment procedures, such as direct extraction and chromatography, minimize the generation of hazardous byproducts and simplify the permitting process for new manufacturing lines. This environmental compatibility positions the technology as a future-proof solution for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for stakeholders evaluating this technology for their specific applications. Understanding these nuances is essential for making informed decisions about process adoption and supplier selection.
Q: What are the advantages of using Eosin Y Na over transition metal catalysts?
A: Eosin Y Na is an organic dye that eliminates the risk of heavy metal contamination in the final API, simplifying purification and reducing costs associated with metal scavenging.
Q: Can this method tolerate diverse fluoroalkyl groups?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully incorporating CF3, CF2COOH, C3F7, and other perfluoroalkyl chains into the indoline skeleton.
Q: Is the MeCN/H2O solvent system scalable?
A: Absolutely. The use of water as a co-solvent aligns with green chemistry principles, reduces toxicity, and simplifies waste treatment compared to purely organic solvent systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroalkyl Indoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like visible-light photocatalysis in delivering high-value intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries are seamlessly translated into reliable industrial processes. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of fluoroalkyl indoline meets the exacting standards required by top-tier pharmaceutical companies. Our commitment to quality and consistency makes us a trusted partner for long-term supply agreements.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized photocatalytic process can enhance your supply chain efficiency and reduce overall manufacturing costs.
