Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Manufacturing
Advanced Mo/Cu Co-Catalyzed Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for Pharmaceutical Manufacturing
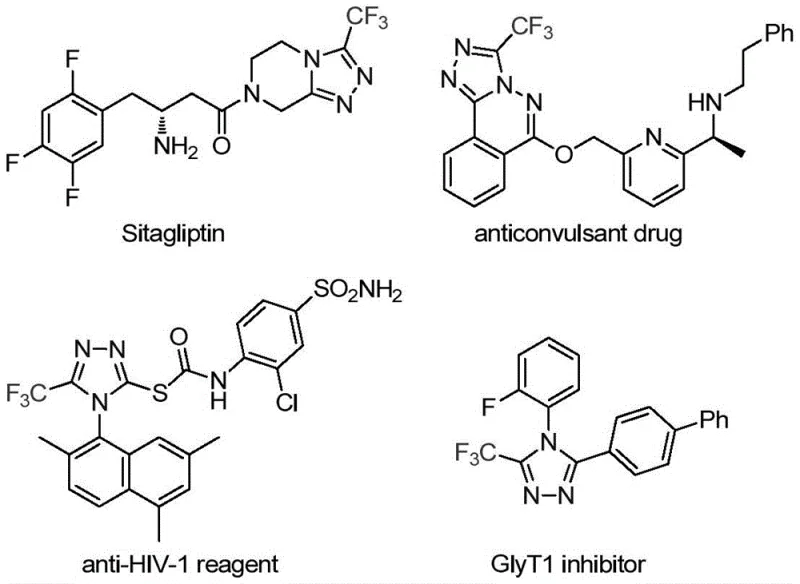
The rapid evolution of medicinal chemistry demands increasingly sophisticated heterocyclic scaffolds, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. A groundbreaking development in this sector is detailed in patent CN113307778A, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a significant leap forward for the production of critical pharmaceutical intermediates, addressing long-standing challenges in yield and operational simplicity. The 1,2,4-triazole core is a privileged structure found in numerous blockbuster drugs, including antidiabetic agents like Sitagliptin and antiviral compounds, making the ability to synthesize these motifs efficiently a top priority for R&D teams globally.
This novel methodology leverages a unique dual-catalytic system involving Molybdenum Hexacarbonyl and Cuprous Acetate to drive the cycloaddition of trifluoroethylimidoyl chloride with functionalized isonitriles. By operating under mild thermal conditions and utilizing readily accessible reagents, this process not only simplifies the synthetic route but also opens new avenues for designing diverse libraries of bioactive molecules. For procurement and supply chain leaders, understanding the nuances of this technology is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazoles has been fraught with synthetic inefficiencies and safety concerns. Traditional literature reports predominantly rely on the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl-substituted 1,2,4-oxazolinones. These classical routes often suffer from harsh reaction conditions, requiring extreme temperatures or strongly acidic/basic environments that can degrade sensitive functional groups. Furthermore, alternative copper-catalyzed multi-component reactions utilizing diazonium salts and trifluorodiazoethane introduce significant safety hazards due to the explosive nature of diazo compounds, posing severe risks during commercial scale-up.
Another prevalent limitation in existing methodologies is the restricted substrate scope, particularly regarding the synthesis of 3,4-disubstituted 1,2,4-triazoles. Many established protocols are optimized for fully substituted triazoles, leaving a gap in the efficient production of specific substitution patterns required for modern drug design. The reliance on unstable intermediates and the generation of difficult-to-remove byproducts further complicate downstream purification, leading to lower overall yields and increased waste generation. These factors collectively contribute to higher manufacturing costs and longer lead times, creating bottlenecks for companies seeking cost reduction in API manufacturing.
The Novel Approach
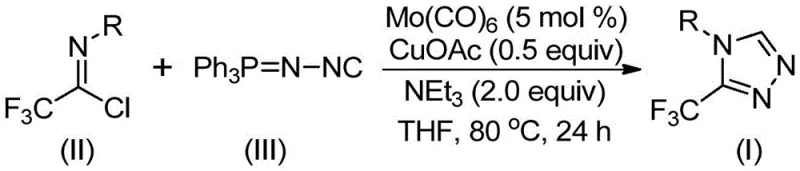
In stark contrast to these legacy methods, the invention disclosed in CN113307778A introduces a streamlined, co-catalytic strategy that fundamentally reshapes the synthetic landscape. By employing trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as starting materials, the process bypasses the need for hazardous diazo reagents entirely. The reaction proceeds smoothly in common aprotic solvents like THF at moderate temperatures ranging from 70°C to 90°C, typically completing within 18 to 30 hours. This mildness allows for exceptional functional group tolerance, enabling the incorporation of diverse substituents such as halogens, alkyl groups, and electron-withdrawing nitro groups without compromising the integrity of the triazole ring.
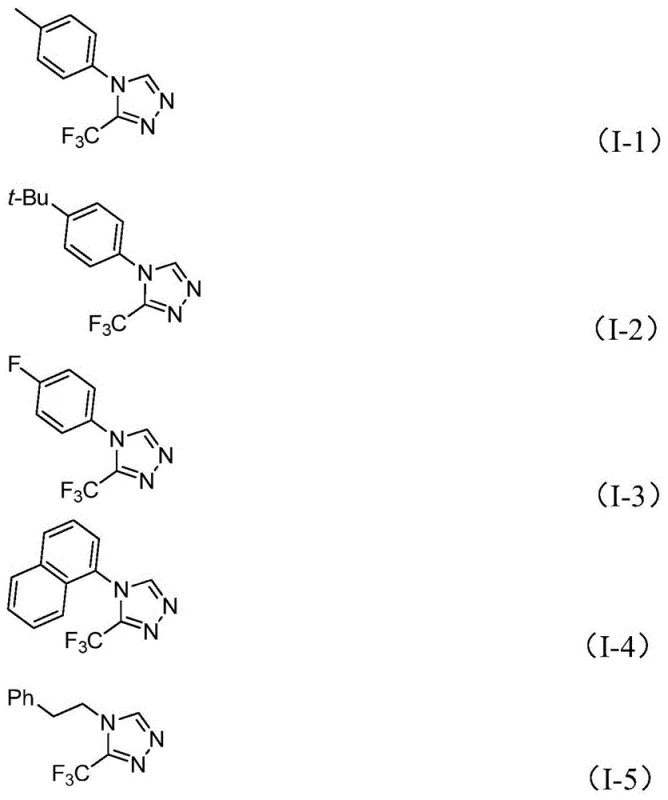
The versatility of this approach is evidenced by its ability to generate a wide array of derivatives, as demonstrated by the successful synthesis of compounds I-1 through I-15 with yields ranging significantly, reaching up to 99% in optimized cases. The use of cheap and easily obtainable starting materials, combined with a simple post-treatment procedure involving filtration and standard column chromatography, drastically reduces the operational complexity. This robustness makes the method highly attractive for the commercial scale-up of complex pharmaceutical intermediates, offering a viable pathway to produce high-value heterocycles with minimal environmental impact and maximum economic efficiency.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The success of this transformation lies in the synergistic interaction between the molybdenum and copper catalysts, which orchestrate a precise sequence of bond-forming events. Mechanistically, the reaction is initiated by the activation of the functionalized isonitrile by Molybdenum Hexacarbonyl [Mo(CO)₆]. This metal complex formation enhances the nucleophilicity of the isonitrile carbon, priming it for the subsequent cycloaddition step. Simultaneously, the Cuprous Acetate [CuOAc] acts as a promoter, facilitating the [3+2] cycloaddition between the activated isonitrile species and the trifluoroethylimidoyl chloride. This concerted process leads to the formation of a five-membered ring intermediate, effectively constructing the core 1,2,4-triazole skeleton in a single pot.
Following the ring closure, the system undergoes a crucial elimination step where triphenylphosphine oxide is removed, driven by the presence of water within the reaction system or during workup. This step is vital for aromatizing the intermediate and yielding the final stable 3-trifluoromethyl-substituted 1,2,4-triazole product. The choice of Triethylamine as the base plays a dual role: it neutralizes the hydrochloric acid byproduct generated during the reaction and helps maintain the optimal pH for the catalytic cycle to proceed uninterrupted. Understanding this mechanistic pathway is critical for R&D directors aiming to optimize reaction parameters for high-purity pharmaceutical intermediates, as slight deviations in catalyst loading or base equivalents can influence the rate of triphenylphosphine oxide elimination and overall conversion.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized stoichiometry and reaction conditions outlined in the patent data. The process is designed to be user-friendly, utilizing standard Schlenk techniques to manage the air-sensitive nature of the molybdenum catalyst while maintaining an inert atmosphere. The following guide summarizes the critical operational parameters derived from the experimental examples, ensuring reproducibility and high yield. For detailed standard operating procedures and safety data sheets, please refer to the technical documentation provided below.
- Combine Molybdenum hexacarbonyl (5 mol %), Cuprous acetate (0.5 equiv), Triethylamine (2.0 equiv), molecular sieves, trifluoroethylimidoyl chloride, and functionalized isonitrile in an organic solvent such as THF.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target 3-trifluoromethyl-1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Mo/Cu co-catalyzed method offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Unlike methods requiring custom-synthesized diazonium salts or unstable hydrazines, this protocol relies on trifluoroethylimidoyl chloride and functionalized isonitriles, which are either commercially available or can be synthesized in situ from abundant precursors like aromatic amines and triphenylphosphine. This accessibility ensures a steady flow of inputs, reducing lead time for high-purity pharmaceutical intermediates and mitigating the risk of production stoppages due to material shortages.
- Cost Reduction in Manufacturing: The economic profile of this synthesis is exceptionally favorable due to the use of earth-abundant and inexpensive metal catalysts. Cuprous Acetate is significantly cheaper than noble metal alternatives like Palladium or Rhodium, and Molybdenum Hexacarbonyl is used in low catalytic loadings (5 mol %). Furthermore, the elimination of hazardous reagents reduces the costs associated with specialized containment, waste disposal, and safety compliance. The high atom economy and the ability to achieve near-quantitative yields in certain substrates mean less raw material is wasted, directly translating to substantial cost savings per kilogram of finished product.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more resilient supply chain. Because the reaction tolerates a wide range of functional groups and proceeds in common solvents like THF, there is no dependency on exotic or hard-to-source reagents that might be subject to geopolitical supply constraints. The scalability demonstrated in the patent, moving seamlessly from milligram to gram scales, indicates that the process can be transferred to large-scale reactors with minimal re-optimization. This predictability allows supply chain planners to forecast production timelines with greater accuracy, ensuring consistent delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with green chemistry principles. The avoidance of explosive diazo compounds and the use of relatively benign solvents reduce the facility's environmental footprint. The post-treatment process is straightforward, involving simple filtration and chromatography, which minimizes the generation of complex aqueous waste streams. As regulatory bodies impose stricter limits on heavy metal residues and solvent emissions, adopting a cleaner synthesis route future-proofs the manufacturing process, ensuring long-term compliance and reducing the burden of environmental remediation costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for their own pipelines. Understanding these details is crucial for assessing the feasibility of integrating this method into existing manufacturing workflows.
Q: What are the key advantages of the Mo/Cu co-catalyzed method over traditional triazole synthesis?
A: Unlike traditional methods that often require harsh cyclization conditions or unstable diazonium salts, this novel approach utilizes mild temperatures (70-90°C) and commercially available starting materials. It offers superior functional group tolerance and eliminates the need for complex multi-step sequences, significantly streamlining the production of pharmaceutical intermediates.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent data explicitly demonstrates that the reaction can be expanded to gram-level scales with high efficiency. The use of inexpensive catalysts like Cuprous Acetate and standard solvents like THF suggests a straightforward path to kilogram and ton-scale manufacturing without requiring exotic equipment.
Q: What is the substrate scope for the R-group in this triazole synthesis?
A: The method exhibits broad substrate scope, successfully accommodating various aryl groups including phenyl, naphthyl, and phenethyl derivatives. It tolerates diverse substituents such as methyl, methoxy, fluoro, chloro, nitro, and tert-butyl groups at ortho, meta, or para positions, allowing for the synthesis of a wide library of analogues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the Mo/Cu co-catalyzed pathway described in CN113307778A and possesses the technical expertise to execute this synthesis with precision. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the highest industry standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug discovery programs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive quotations. Let us help you accelerate your timeline to market with a reliable, cost-effective, and scalable supply of high-quality triazole intermediates.
