Advanced Manufacturing of 3-Trifluoromethyl-1,2,4-Triazole Intermediates for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust and cost-efficient pathways for constructing fluorinated heterocycles, which serve as critical scaffolds in modern drug discovery. Patent CN114920707B, published in April 2023, introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds that addresses longstanding challenges in synthetic efficiency and operational complexity. This technology leverages the ubiquitous solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source, enabling a tandem cyclization promoted by molecular iodine. For R&D directors and procurement specialists, this represents a significant shift away from expensive, specialized reagents toward a more sustainable and economically viable manufacturing model. The ability to synthesize these high-value intermediates under air atmosphere without stringent anhydrous conditions drastically simplifies the production workflow, offering a compelling value proposition for reliable pharmaceutical intermediate supplier networks aiming to optimize their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of trifluoromethyl-substituted 1,2,4-triazole cores has relied on multi-step sequences involving harsh reaction conditions and costly precursors. Conventional routes often necessitate the use of specialized trifluoroacetylating agents or pre-functionalized hydrazines that are sensitive to moisture and oxygen, requiring rigorous exclusion of air and water via Schlenk lines or gloveboxes. These operational constraints not only inflate capital expenditure for specialized equipment but also introduce significant bottlenecks in batch processing times. Furthermore, traditional methods frequently suffer from limited substrate scope, where electron-deficient or sterically hindered aromatic amines fail to cyclize efficiently, leading to poor yields and difficult purification profiles. The reliance on stoichiometric amounts of expensive coupling reagents and transition metal catalysts further exacerbates the cost burden, making cost reduction in API manufacturing a persistent challenge for generic drug producers seeking to maintain margin integrity in competitive markets.
The Novel Approach
In stark contrast, the methodology disclosed in CN114920707B utilizes a clever design where the solvent DMF acts as a C1 synthon, effectively merging the roles of reactant and medium. By employing molecular iodine as a mild promoter, the reaction proceeds smoothly at temperatures between 110°C and 130°C under ambient air, eliminating the need for inert gas protection. This approach allows for the direct conversion of readily available trifluoroethyliminohydrazides into the target triazole skeleton with remarkable efficiency. The reaction tolerates a wide array of functional groups, including halogens, alkyls, and alkoxy substituents on the aromatic ring, demonstrating exceptional versatility for generating diverse chemical libraries. As illustrated in the general reaction scheme below, this streamlined process converts simple starting materials into complex heterocycles in a single pot, significantly reducing waste generation and processing time.
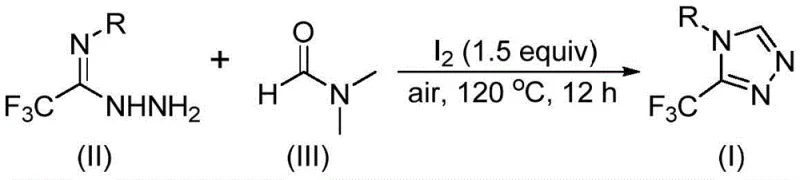
The operational simplicity of this novel route translates directly into enhanced supply chain reliability. Since DMF is one of the most common industrial solvents, sourcing is never a bottleneck, and the elimination of sensitive reagents reduces the risk of batch failures due to environmental exposure. This robustness is particularly valuable for commercial scale-up of complex pharmaceutical intermediates, where consistency and reproducibility are paramount. The method's ability to accommodate various substituents on the aryl ring ensures that medicinal chemists can rapidly access analogues for structure-activity relationship (SAR) studies without redesigning the entire synthetic pathway, thereby accelerating the drug development timeline.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
From a mechanistic perspective, this transformation is a fascinating example of solvent-participatory synthesis. The reaction initiates with the activation of DMF by molecular iodine, which facilitates the cleavage of either the formyl C-H bond or the N-methyl C-H bond. When the formyl group acts as the carbon source, it undergoes condensation with the hydrazide moiety of the substrate to form a hydrazone intermediate. This is followed by an intramolecular cyclization event that eliminates dimethylamine, yielding the aromatized 3-trifluoromethyl-1,2,4-triazole product. Alternatively, when the N-methyl group participates, DMF reacts with iodine to generate an amine salt species, which then undergoes nucleophilic addition with the hydrazide. Subsequent elimination of N-methylformamide generates an azadiene intermediate that cyclizes and oxidizes to the final product. Understanding these dual pathways is crucial for R&D teams optimizing reaction parameters, as subtle changes in temperature or stoichiometry can influence which pathway dominates, although both lead to the desired core structure.
Impurity control is inherently managed by the clean nature of this cyclization. The byproducts, primarily dimethylamine salts or iodine residues, are easily removed during the aqueous workup and subsequent silica gel chromatography. The absence of heavy metal catalysts means there is no risk of toxic metal contamination in the final API, a critical quality attribute for regulatory compliance. The reaction's tolerance to air suggests that oxidative side reactions are minimal or that the iodine promoter effectively manages the redox balance required for aromatization. This mechanistic clarity provides confidence in the process's scalability, as the reaction kinetics are driven by thermal energy and reagent concentration rather than fragile catalytic cycles that might deactivate upon scale-up.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for maximum throughput and minimal technical overhead. The protocol involves simply charging a reaction vessel with the trifluoroethyliminohydrazide substrate, molecular iodine (typically 1.5 equivalents), and DMF. The mixture is heated to the optimal range of 110-130°C and stirred for 10 to 15 hours. Upon completion, standard workup procedures such as filtration, washing, and column chromatography yield the pure product. This straightforward procedure minimizes the need for specialized training for plant operators and reduces the likelihood of human error during setup. For detailed standardized synthesis steps and specific substrate examples, please refer to the guide below.
- Combine trifluoroethyliminohydrazide, molecular iodine (1.5 equivalents), and DMF solvent in a reaction vessel.
- Heat the mixture to 110-130°C under air atmosphere and stir for 10-15 hours to complete the cyclization.
- Perform post-treatment including filtration, washing, and column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this technology are profound. The primary driver of value is the drastic simplification of the bill of materials. By utilizing DMF as a reactant, the process eliminates the need for purchasing separate, expensive C1 building blocks or activating agents. This consolidation of reagents leads to substantial cost savings in raw material procurement. Furthermore, the use of molecular iodine, a commodity chemical, replaces costly transition metal catalysts that often require complex removal steps and generate hazardous waste streams. The elimination of inert atmosphere requirements also reduces utility costs associated with nitrogen or argon consumption, contributing to a leaner manufacturing footprint.
- Cost Reduction in Manufacturing: The integration of the solvent and reactant functions into a single molecule (DMF) fundamentally alters the cost structure of triazole production. There is no need for expensive anhydrous solvents or specialized drying columns, which lowers both CAPEX and OPEX. The high atom economy of the cyclization ensures that a significant portion of the starting mass ends up in the final product, minimizing waste disposal fees. Additionally, the mild reaction conditions reduce energy consumption compared to high-pressure or cryogenic alternatives, further enhancing the overall economic viability of the process for large-scale operations.
- Enhanced Supply Chain Reliability: Sourcing risks are mitigated because all key components—DMF, iodine, and simple aromatic amines—are globally available commodity chemicals with stable supply chains. Unlike proprietary catalysts or exotic reagents that may face geopolitical or logistical disruptions, these materials can be sourced from multiple vendors, ensuring business continuity. The robustness of the reaction against moisture and oxygen means that storage and handling requirements are less stringent, reducing the risk of原料 degradation during transit or warehousing. This reliability is essential for maintaining consistent production schedules and meeting delivery commitments to downstream pharmaceutical clients.
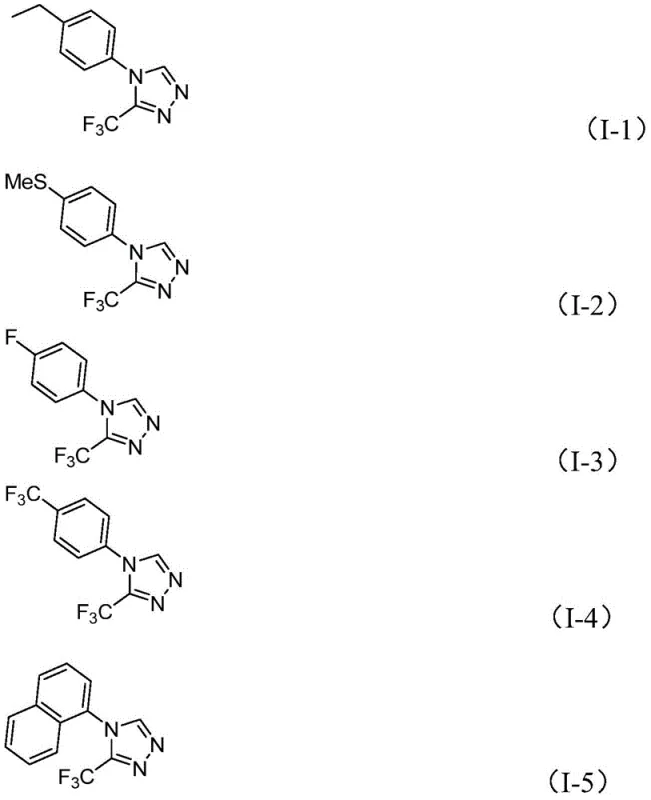
- Scalability and Environmental Compliance: The process is inherently green, aligning with modern sustainability goals. The absence of heavy metals simplifies wastewater treatment and reduces the environmental burden of the facility. Scaling from gram to kilogram or ton quantities does not require re-engineering the reaction setup, as the thermal and mixing requirements are standard for existing reactor infrastructure. The high yields observed across a broad substrate scope, as shown in the product examples below, indicate that the process is robust enough to handle diverse manufacturing campaigns without extensive re-optimization, facilitating rapid response to market demands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. They are derived from the specific advantages and operational details outlined in the patent documentation. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios. The answers reflect the empirical data and mechanistic understanding established during the development of this process.
Q: Does this synthesis require strict anhydrous or anaerobic conditions?
A: No, a key advantage of this patented method (CN114920707B) is that it operates efficiently under standard air atmosphere without the need for expensive inert gas protection or rigorous drying of solvents.
Q: What is the role of DMF in this reaction mechanism?
A: DMF serves a dual function as both the reaction solvent and the carbon source (C1 synthon), utilizing either its formyl or N-methyl group to construct the triazole ring, which significantly reduces raw material costs.
Q: Is this method scalable for industrial production of API intermediates?
A: Yes, the process uses cheap, commercially available starting materials and simple operational conditions (110-130°C), making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocycle synthesis in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries translate seamlessly into industrial reality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole intermediate meets the highest global standards. Our commitment to quality and consistency makes us a trusted partner for multinational pharmaceutical companies seeking to secure their supply chains against volatility.
We invite you to collaborate with us to leverage this advanced iodine-promoted cyclization technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and innovation in your drug development pipeline.
