Advanced Bidentate Phosphine Ligands Enabling Efficient Butadiene Hydroformylation for Industrial Adipaldehyde Production
The chemical industry is currently witnessing a pivotal shift in the synthesis of C6 dicarbonyl compounds, driven by the urgent need for more sustainable and efficient pathways to produce key polymer precursors like adipaldehyde. Patent CN113004326B introduces a groundbreaking class of bidentate phosphine ligands characterized by a rigid dibenzobarrelene backbone, which fundamentally alters the landscape of butadiene hydroformylation. These novel structures, defined by General Formula 1, are engineered to overcome the historical bottlenecks of low selectivity and harsh reaction conditions that have plagued this technology for over six decades. By precisely tuning the steric and electronic properties of the phosphine substituents, this innovation enables the direct conversion of abundant 1,3-butadiene into 1,6-hexanedial with unprecedented efficiency.
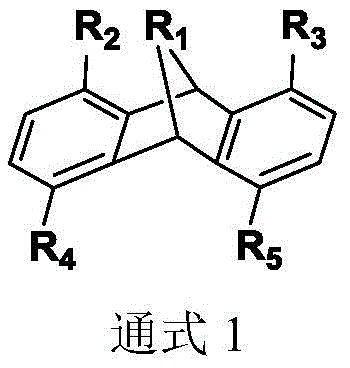
For R&D directors and process engineers, the implications of this technology are profound, offering a reliable phosphine ligand supplier pathway to access high-purity intermediates essential for nylon and polyurethane manufacturing. The structural versatility allows for the attachment of various aryl, alkyl, and heterocyclic groups at the phosphorus centers, facilitating fine-tuning for specific catalytic cycles. This level of molecular precision is critical for minimizing byproduct formation and maximizing atom economy, addressing the core challenges of modern green chemistry in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of adipaldehyde has relied heavily on the multi-step oxidation of 1,6-cyclohexanediol or cyclohexene, processes that are inherently inefficient and environmentally burdensome. These conventional routes suffer from the unavailability of cheap raw materials, the high cost of stoichiometric oxidants, and the significant difficulty in recycling these oxidants, leading to substantial waste generation. Furthermore, early attempts at direct butadiene hydroformylation, dating back to the 1960s, were commercially non-viable due to extreme reaction conditions requiring synthesis gas pressures exceeding 750 bar. Even under such dangerous and energy-intensive conditions, the selectivity for the desired linear adipaldehyde product remained dismally low, often below 10%, rendering the process economically unfeasible for cost reduction in fine chemical manufacturing.
The Novel Approach
The technology disclosed in CN113004326B represents a paradigm shift by introducing a new generation of bidentate phosphine ligands that operate effectively under significantly milder conditions. Unlike the single-site catalysts of the past, these ligands create a well-defined coordination environment around the rhodium center, enforcing a specific geometry that favors the formation of the linear dialdehyde. The patent demonstrates that with these optimized ligands, reaction pressures can be reduced to a manageable 2 MPa to 5 MPa range while achieving adipaldehyde selectivity approaching 60%. This dramatic improvement in performance metrics transforms the hydroformylation of butadiene from a laboratory curiosity into a viable industrial alternative to the hydronitrile route, promising substantial cost savings and supply chain reliability.
Mechanistic Insights into Rhodium-Catalyzed Hydroformylation
The exceptional performance of these ligands stems from their unique ability to control the regioselectivity of the catalytic cycle through steric hindrance and electronic modulation. The rigid dibenzobarrelene scaffold prevents the free rotation of the phosphine arms, locking the metal center into a conformation that disfavors the formation of branched isomers. During the catalytic cycle, the coordination of the bulky phosphine groups directs the insertion of the alkene into the metal-hydride bond specifically at the terminal position, which is the critical step for generating the linear 1,6-dialdehyde rather than internal isomers. This mechanistic control is further enhanced by the electron-donating or withdrawing nature of the substituents on the phosphorus atoms, which modulates the electron density at the rhodium center to optimize the rates of oxidative addition and reductive elimination steps.
From an impurity control perspective, the design of these ligands minimizes the formation of oligomers and other side products that typically contaminate the crude reaction mixture. The high selectivity observed in the experimental data indicates that the catalyst system effectively suppresses competing reactions such as isomerization of the double bond prior to carbonylation. For quality assurance teams, this means a cleaner crude product profile, which significantly simplifies downstream purification processes. The ability to achieve such high specificity reduces the load on distillation columns and lowers the energy consumption associated with separating close-boiling isomers, directly contributing to a more sustainable and cost-effective manufacturing process for high-purity OLED material or polymer additive precursors.
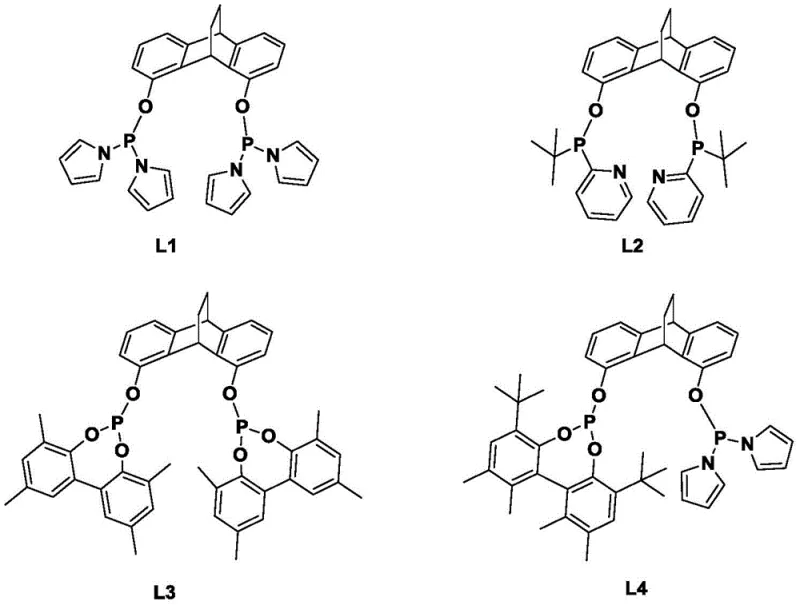
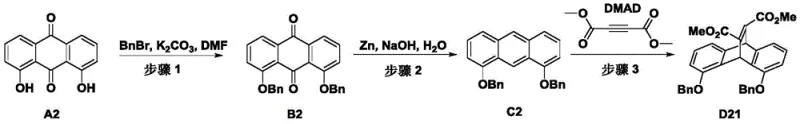
How to Synthesize Bidentate Phosphine Ligand L1 Efficiently
The synthesis of these advanced ligands follows a logical and scalable sequence starting from readily available anthraquinone derivatives. The process begins with the protection of hydroxyl groups and the construction of the bridged aromatic system, followed by functionalization to introduce the reactive sites necessary for phosphination. The final step involves the coupling of the phenolic intermediate with chlorophosphines under basic conditions to form the P-O-C linkage. This robust synthetic route ensures high purity and reproducibility, which are essential for commercial scale-up of complex polymer additives. Detailed standardized synthesis steps are provided in the guide below.
- Protection and Reduction: React 1,8-dihydroxyanthraquinone with benzyl bromide and potassium carbonate, followed by zinc powder reduction to form the dibenzobarrelene backbone.
- Backbone Functionalization: Perform a Diels-Alder reaction with dimethyl butynedioate, followed by hydrolysis and decarboxylation to refine the core structure.
- Phosphination: React the hydroxyl-functionalized intermediate with dipyrrolylphosphinous chloride in the presence of triethylamine to yield the final ligand L1.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers strategic advantages that extend far beyond simple yield improvements. By shifting the feedstock base from specialized cyclic alcohols to bulk petrochemical butadiene, manufacturers can decouple their production costs from the volatility of niche intermediate markets. The elimination of expensive stoichiometric oxidants not only reduces raw material costs but also removes the logistical burden of handling and disposing of hazardous chemical waste. This transition aligns perfectly with global sustainability goals, reducing the carbon footprint of the supply chain while enhancing the overall resilience of the production network against raw material shortages.
- Cost Reduction in Manufacturing: The most significant economic driver is the drastic simplification of the process flow. By replacing multi-step oxidation sequences with a direct catalytic hydroformylation, the number of unit operations is significantly reduced, leading to lower capital expenditure and operating costs. The removal of heavy metal oxidants eliminates the need for complex wastewater treatment facilities dedicated to heavy metal removal, resulting in substantial cost savings in environmental compliance and waste management. Furthermore, the milder reaction pressures reduce the engineering requirements for reactor vessels, allowing for the use of standard high-pressure equipment rather than specialized ultra-high-pressure infrastructure.
- Enhanced Supply Chain Reliability: 1,3-Butadiene is a major commodity chemical produced in vast quantities globally as a byproduct of ethylene production, ensuring a stable and abundant supply source. Unlike specialized precursors that may be subject to supply disruptions or monopolistic pricing, butadiene is available from multiple integrated petrochemical complexes worldwide. This abundance guarantees reducing lead time for high-purity intermediates and provides a buffer against market fluctuations. The robustness of the catalyst system also means longer catalyst life and less frequent replacement, ensuring continuous operation and consistent product availability for downstream customers.
- Scalability and Environmental Compliance: The process operates at temperatures and pressures that are well within the range of standard industrial chemical reactors, facilitating easy commercial scale-up of complex fine chemicals. The absence of toxic oxidants and the use of synthesis gas, which can be derived from renewable sources in the future, position this technology as a future-proof solution for green manufacturing. The high selectivity minimizes the generation of organic waste streams, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations without the need for costly end-of-pipe solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel ligand technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on performance metrics and operational parameters. Understanding these details is crucial for evaluating the feasibility of integrating this catalytic system into existing production lines.
Q: What is the primary advantage of these new ligands over traditional hydroformylation catalysts?
A: The primary advantage is the significant improvement in regioselectivity for adipaldehyde. While early catalysts achieved less than 10% selectivity under harsh pressures (>750 bar), these novel bidentate ligands achieve selectivity approaching 60% at much milder pressures (2-5 MPa).
Q: Can these ligands be used for large-scale industrial production?
A: Yes, the patent explicitly states that the catalyst system lays a foundation for further industrialization. The use of abundant butadiene feedstock and the elimination of expensive oxidants make the process economically viable for scale-up.
Q: What metal precursors are compatible with these phosphine ligands?
A: The ligands are designed to coordinate with Rhodium and Cobalt precursors. Specifically, Rh(CO)2(acac) is highlighted as the preferred metal precursor for achieving optimal catalytic activity and selectivity in the hydroformylation reaction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bidentate Phosphine Ligand Supplier
As the demand for efficient and sustainable chemical processes grows, partnering with an experienced CDMO becomes essential for translating patented innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to this new hydroformylation technology is seamless and risk-free. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ligand delivered meets the exacting standards required for high-performance catalytic applications.
We invite you to engage with our technical procurement team to discuss how this technology can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this butadiene-based route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, taking the first step towards optimizing your supply chain and securing a competitive advantage in the global market for polymer intermediates.
