Advanced Manufacturing of Bidentate Phosphine Ligands via Double Grignard Reagents
The landscape of organometallic catalysis is undergoing a significant transformation driven by the urgent need for safer, more sustainable, and economically viable synthetic routes. Patent CN113912646A introduces a groundbreaking preparation method for bidentate phosphine ligands, specifically targeting the critical demands of the olefin hydroformylation industry. This technology represents a paradigm shift away from the traditionally hazardous and energy-intensive protocols that have long dominated the sector. By leveraging a novel double Grignard reagent strategy, the invention successfully circumvents the reliance on n-butyllithium, a reagent notorious for its pyrophoric nature and stringent handling requirements. For R&D directors and process engineers, this development signals a new era of accessibility for high-performance ligands like BISBI and its analogues, promising not only enhanced safety profiles but also streamlined purification workflows that are essential for modern reliable catalyst supplier operations. The core innovation lies in the ability to generate the necessary organometallic intermediates under mild reflux conditions, effectively removing the barrier of cryogenic processing that has historically constrained production capacity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bidentate phosphine ligands for hydroformylation has been plagued by severe operational hazards and complex process engineering challenges. Traditional methodologies, such as those described in earlier patents like U.S. Pat. No. 4,090,909, rely heavily on the use of n-butyllithium to generate lithiated intermediates. This approach necessitates maintaining reaction temperatures as low as -70°C to prevent thermal decomposition and uncontrolled side reactions, imposing a massive energy burden on the manufacturing facility. Furthermore, n-butyllithium is extremely sensitive to moisture and oxygen, requiring rigorous anhydrous and anaerobic conditions that complicate reactor design and increase the risk of catastrophic safety incidents. The quenching and workup procedures associated with organolithium chemistry often generate substantial volumes of hazardous waste, creating significant environmental compliance hurdles. For procurement managers, these factors translate into inflated raw material costs due to the premium pricing of specialized cryogenic reagents and the high capital expenditure required for specialized low-temperature reactor infrastructure. The inherent instability of these intermediates also limits the batch size, creating bottlenecks in cost reduction in specialty chemical manufacturing initiatives.
The Novel Approach
In stark contrast, the methodology disclosed in CN113912646A utilizes a robust double Grignard reagent pathway that fundamentally alters the thermodynamic and kinetic profile of the synthesis. By starting with a biaryl compound and employing N-bromosuccinimide (NBS) for radical bromination, the process generates a stable halogenated precursor that can be smoothly converted into a bis-Grignard species using magnesium powder. This transformation occurs under standard reflux conditions in solvents like tetrahydrofuran or toluene, completely eliminating the need for ultra-low temperature control. The subsequent reaction with trivalent phosphine compounds, such as chlorodiphenylphosphine, is facilitated by the addition of TMEDA, which enhances the reactivity of the Grignard species without compromising safety. This approach not only simplifies the operational protocol but also significantly improves the overall yield by minimizing the formation of by-products typically associated with harsh lithiation conditions. The result is a streamlined two-step process that is inherently safer, more energy-efficient, and far more amenable to commercial scale-up of complex ligands, offering a distinct competitive advantage in the global supply chain.
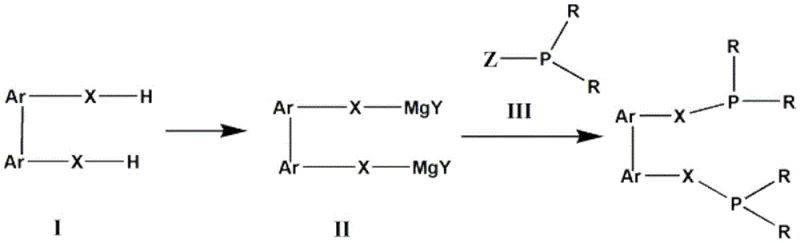
Mechanistic Insights into Double Grignard Reagent Formation
The mechanistic elegance of this synthesis lies in the controlled generation of the bis-Grignard intermediate, which serves as the pivotal nucleophile in the construction of the phosphine-carbon bonds. The process initiates with a radical bromination where NBS, activated by AIBN, selectively functionalizes the benzylic positions of the biaryl substrate. This step is crucial as it installs the leaving groups necessary for the subsequent oxidative insertion of magnesium. Unlike lithiation, which can suffer from issues regarding regioselectivity and metal-halogen exchange equilibria, the formation of the Grignard reagent here is driven by the direct reaction of the alkyl bromide with magnesium metal. The presence of iodine acts as an initiator, cleaning the magnesium surface and facilitating the electron transfer required to break the carbon-bromine bond. Once formed, the bis-Grignard species exhibits remarkable stability compared to its organolithium counterparts, allowing it to persist in solution at elevated temperatures without significant degradation. This stability is paramount for ensuring high purity in the final product, as it prevents the formation of homocoupling by-products or elimination species that often contaminate ligand batches produced via older methods.
Furthermore, the inclusion of TMEDA (N,N,N',N'-tetramethylethylenediamine) plays a sophisticated role in modulating the reactivity of the Grignard center. TMEDA acts as a bidentate Lewis base that coordinates to the magnesium atom, breaking up Schlenk equilibrium aggregates and generating a more reactive, monomeric or dimeric species. This activation allows the nucleophilic attack on the phosphorus center of the chlorophosphine to proceed efficiently even at moderate temperatures (e.g., 70°C). From an impurity control perspective, this mechanism is superior because the milder conditions suppress competing elimination reactions that could lead to olefinic impurities difficult to remove downstream. The result is a cleaner reaction profile where the primary challenge shifts from managing hazardous exotherms to optimizing stoichiometry and mixing efficiency. For quality control teams, this translates to a much narrower impurity spectrum, facilitating easier crystallization and purification steps to achieve the high-purity bidentate phosphine ligand specifications required for sensitive catalytic applications.
How to Synthesize BISBI Efficiently
The synthesis of 2,2'-bis(diphenylphosphinomethylene)-1,1'-biphenyl (BISBI) serves as the prime exemplar of this technology's practical utility. The procedure begins with the radical bromination of 2,2'-dimethylbiphenyl using NBS and AIBN in dichloromethane, yielding the dibromide intermediate in high purity after simple recrystallization. This solid intermediate is then subjected to Grignard formation in dry THF with magnesium and iodine, followed by the addition of TMEDA and chlorodiphenylphosphine. The reaction mixture is heated to reflux to ensure complete conversion, after which standard aqueous workup and recrystallization afford the target ligand. This robust protocol demonstrates how the theoretical advantages of the double Grignard route translate into tangible laboratory and plant-floor successes, providing a clear roadmap for reducing lead time for high-purity intermediates. The detailed standardized synthesis steps for this specific transformation are outlined below.
- Radical Bromination: React 2,2'-dimethylbiphenyl with N-bromosuccinimide (NBS) and AIBN in dichloromethane under reflux to generate 2,2'-bromomethyl biphenyl.
- Grignard Formation & Phosphination: Treat the brominated intermediate with magnesium powder and iodine in THF to form the bis-Grignard reagent, then react with chlorodiphenylphosphine in the presence of TMEDA.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain leaders, the adoption of this Grignard-based synthesis offers profound economic and logistical benefits that extend far beyond the laboratory bench. The elimination of n-butyllithium removes a major cost driver, as this reagent is not only expensive to purchase but also costly to store and handle due to its classification as a dangerous good. By replacing cryogenic operations with standard reflux conditions, facilities can utilize existing general-purpose reactor trains without needing specialized low-temperature jackets or extensive insulation, drastically lowering capital expenditure barriers for new production lines. The simplified two-step nature of the process reduces the number of unit operations, which directly correlates to lower labor costs, reduced solvent consumption, and decreased utility usage per kilogram of product. These efficiencies compound to create a significantly leaner manufacturing model that enhances margin potential while maintaining rigorous quality standards. Moreover, the improved stability of the intermediates allows for more flexible scheduling and larger batch sizes, mitigating the risks of supply disruptions that often plague complex fine chemical supply chains.
- Cost Reduction in Manufacturing: The transition away from ultra-low temperature processing results in substantial energy savings, as the massive refrigeration loads required for -70°C operations are entirely eliminated. Additionally, the use of magnesium powder and NBS represents a shift towards more commodity-grade raw materials that are widely available and price-stable compared to specialized organolithium reagents. The higher overall yields reported in the patent examples mean that less raw material is wasted per unit of output, further driving down the cost of goods sold. This economic efficiency is compounded by the simplified workup procedures, which reduce the volume of wastewater and hazardous waste requiring treatment, thereby lowering environmental compliance costs.
- Enhanced Supply Chain Reliability: By utilizing a synthetic route that relies on stable intermediates and common solvents like toluene and THF, the supply chain becomes more resilient to external shocks. The avoidance of pyrophoric reagents simplifies logistics and transportation, reducing the regulatory burden and insurance costs associated with moving dangerous chemicals. The robustness of the Grignard method allows for production in a wider range of geographic locations, diversifying the supplier base and reducing dependency on single-source vendors who possess niche cryogenic capabilities. This flexibility ensures consistent availability of critical ligands for downstream customers, safeguarding their own production schedules against raw material shortages.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of highly toxic by-products make this process inherently scalable from pilot plant to multi-ton commercial production. The reduced generation of hazardous waste aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing asset against tightening emission standards. The ability to run reactions at reflux rather than cryogenic temperatures also improves heat transfer efficiency in large vessels, ensuring uniform reaction progress and consistent product quality across large batches. This scalability is essential for meeting the growing demand for hydroformylation catalysts in the petrochemical and polymer industries without compromising on safety or sustainability metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel bidentate phosphine ligand synthesis. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the operational benefits and chemical rationale behind the technology. Understanding these nuances is critical for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the double Grignard route superior to traditional n-butyllithium methods?
A: The double Grignard route eliminates the need for hazardous n-butyllithium and cryogenic conditions (-70°C), significantly improving operational safety and reducing energy costs associated with cooling.
Q: What is the role of TMEDA in this synthesis?
A: N,N,N',N'-tetramethylethylenediamine (TMEDA) acts as a chelating ligand that stabilizes the Grignard reagent and enhances its nucleophilicity, facilitating the substitution reaction with the trivalent phosphine compound.
Q: Can this process be scaled for industrial production?
A: Yes, the process uses mild reflux conditions and avoids ultra-low temperatures, making it highly suitable for large-scale industrial manufacturing using standard reactor equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bidentate Phosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer and more efficient synthetic methodologies is not just a technical upgrade but a strategic imperative for the modern chemical industry. Our team of expert process chemists has extensively analyzed the Grignard-based route described in CN113912646A and validated its potential for delivering high-quality ligands at a competitive cost structure. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this technology are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reflux conditions required for this synthesis, backed by rigorous QC labs that guarantee stringent purity specifications for every batch released. We are committed to being a partner that not only supplies materials but also drives value through continuous process optimization and technical support.
We invite you to collaborate with us to leverage this advanced manufacturing capability for your specific catalytic applications. Whether you require custom synthesis of novel ligand derivatives or bulk supply of established products like BISBI, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to see how our implementation of this patent technology can enhance your supply chain resilience and product performance.
