Advanced Sulfonylation Strategy for High-Purity Blonanserin Manufacturing
Advanced Sulfonylation Strategy for High-Purity Blonanserin Manufacturing
The pharmaceutical landscape for antipsychotic agents continues to evolve, driven by the demand for safer, more efficient synthetic routes that guarantee exceptional purity profiles. Patent CN102887856A introduces a transformative methodology for the synthesis of Blonanserin, a potent atypical antipsychotic, shifting the paradigm from hazardous chlorination to a refined sulfonylation strategy. This technical breakthrough addresses long-standing challenges in heterocyclic chemistry, specifically regarding regioselectivity and impurity control in complex fused ring systems. By replacing aggressive chlorinating agents with substituted sulfonyl chlorides, the process achieves a remarkable purity of 99.8%, meeting the stringent standards required for active pharmaceutical ingredients (APIs). For R&D directors and procurement specialists alike, this innovation represents a significant leap forward in cost reduction in pharmaceutical intermediate manufacturing, offering a pathway that is not only chemically elegant but also industrially robust and environmentally sustainable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Blonanserin has relied heavily on the activation of the pyridone ketone moiety using phosphorus oxychloride (POCl3) or phenylphosphonyl dichloride. While these reagents are effective at converting the hydroxyl group into a leaving group, they introduce severe operational and chemical drawbacks that complicate large-scale production. The use of POCl3 is associated with high toxicity, corrosiveness, and the generation of substantial acidic waste, posing significant environmental and safety burdens on manufacturing facilities. More critically from a chemical perspective, the resulting 2-chloro intermediate exhibits poor reaction selectivity during the subsequent nucleophilic substitution with N-ethylpiperazine.
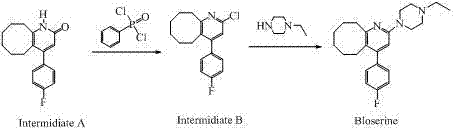
As illustrated in the conventional pathway, the presence of both a chloropyridine and a fluorobenzene structure within the same molecule creates a competitive reactivity scenario. Since fluorine and chlorine belong to the same halogen group, the nucleophile can inadvertently attack the fluorine position on the phenyl ring, leading to the formation of "Impurity A". This bis-substituted byproduct is structurally similar to the target molecule and is notoriously difficult to remove during purification, often requiring multiple recrystallizations that erode overall yield. Furthermore, the harsh conditions required for POCl3 activation can degrade sensitive functional groups, limiting the scalability and reliability of the supply chain for this critical pharmaceutical intermediate.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a sulfonylation strategy that fundamentally alters the reactivity profile of the intermediate. By reacting the starting ketone, 4-(4-fluorophenyl)-5,6,7,8,9,10-hexahydrocyclooctano[b]pyridin-2(1H)-one, with various substituted sulfonyl chlorides, the process generates a sulfonate ester intermediate (Intermediate II). This transformation can be conducted under mild conditions, often at room temperature, using a wide range of benign solvents such as dichloromethane, chloroform, or even greener alternatives like ethyl acetate.
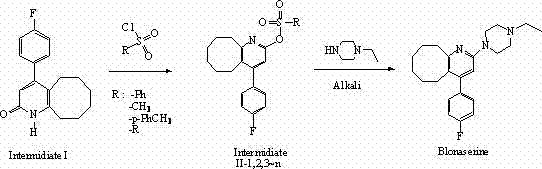
The resulting sulfonate group serves as an excellent leaving group that is highly susceptible to nucleophilic attack by N-ethylpiperazine, yet it does so with exceptional regioselectivity. Unlike the chloro-intermediate, the sulfonate intermediate directs the substitution exclusively to the pyridine 2-position, effectively suppressing the unwanted displacement of the fluorine atom on the aromatic ring. This selectivity is the cornerstone of the invention, enabling the direct production of Blonanserin with minimal byproduct formation. The operational simplicity, combined with the ability to use low-toxicity solvents and catalysts like triethylamine or pyridine, makes this route ideally suited for commercial scale-up of complex pharmaceutical intermediates, ensuring a consistent and reliable supply for downstream API manufacturers.
Mechanistic Insights into Sulfonylation and Impurity Control
To fully appreciate the value of this synthetic route, one must delve into the mechanistic nuances that govern the reaction selectivity. The core challenge in Blonanserin synthesis lies in the differential reactivity of the two potential electrophilic sites: the C-2 position of the pyridine ring and the C-4 position of the fluorophenyl ring. In traditional chlorination methods, the electron-withdrawing nature of the pyridine nitrogen activates the C-2 chlorine, but the C-F bond remains sufficiently labile under the high temperatures and strong basic conditions often required for the piperazine coupling.
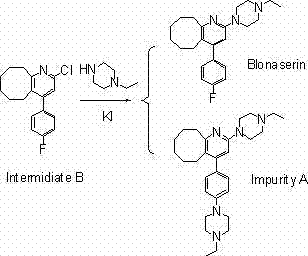
The formation of Impurity A, as depicted in the mechanistic analysis, occurs when the N-ethylpiperazine acts as a nucleophile against the electron-deficient fluorophenyl ring. This side reaction is exacerbated by the harsh conditions and the comparable leaving group ability of chloride in certain contexts. However, the sulfonylation method mitigates this risk through electronic and steric modulation. The sulfonate ester formed at the C-2 position is a much softer and more reactive leaving group compared to the aryl fluoride. Consequently, the nucleophilic substitution proceeds rapidly at the pyridine ring under milder thermal conditions, kinetically favoring the desired product before the thermodynamic conditions necessary for C-F bond cleavage can be reached. This kinetic control is vital for maintaining the integrity of the fluorophenyl moiety, ensuring that the final product profile is dominated by the target API rather than structural analogs.
Furthermore, the versatility of the sulfonyl chloride reagent allows for fine-tuning of the leaving group ability. The patent explores various substituents (R groups) on the sulfonyl chloride, such as methyl, tosyl, and phenyl groups. Each variant offers slightly different electronic properties, allowing process chemists to optimize the reaction rate and solubility profile of the intermediate. For instance, the tosylate intermediate (Intermediate II-1) provides a balance of stability and reactivity that facilitates easy isolation and handling, while still undergoing smooth substitution with N-ethylpiperazine. This level of control over the reaction mechanism translates directly into a cleaner crude product, reducing the burden on purification units and enhancing the overall efficiency of the manufacturing process.
How to Synthesize Blonanserin Efficiently
The practical implementation of this synthesis involves a straightforward two-step sequence that is amenable to standard reactor setups found in most fine chemical plants. The first step focuses on the activation of the ketone precursor, while the second step executes the crucial carbon-nitrogen bond formation. The protocol emphasizes the importance of solvent selection and stoichiometry to maximize conversion while minimizing waste. Detailed operational parameters, including specific molar ratios and temperature ranges, are critical for reproducing the high yields reported in the patent data.
- React 4-(4-fluorophenyl)-5,6,7,8,9,10-hexahydrocyclooctano[b]pyridin-2(1H)-one with a substituted sulfonyl chloride in the presence of a base catalyst to form the sulfonate intermediate.
- Condense the resulting sulfonate intermediate with N-ethylpiperazine under reflux conditions to substitute the sulfonate group.
- Purify the crude product through acid-base extraction and recrystallization to achieve pharmaceutical grade purity exceeding 99.8%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this sulfonylation-based process offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain risk profile. By eliminating the need for phosphorus oxychloride, a highly regulated and hazardous material, manufacturers can reduce the regulatory burden and insurance costs associated with storage and transport. The availability of substituted sulfonyl chlorides is broad, with many variants being commodity chemicals, which ensures a stable and reliable pharmaceutical intermediate supplier network without the bottlenecks often seen with specialized chlorinating agents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive purification steps. Since the selectivity is inherently higher, the formation of Impurity A is suppressed, meaning less material is lost during recrystallization and chromatography. Additionally, the ability to run the first step at room temperature significantly reduces energy consumption compared to the high-temperature reflux often required for chlorination. The use of recoverable solvents like dichloromethane or ethyl acetate further contributes to cost reduction in API manufacturing by lowering raw material expenses and waste disposal fees.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, a critical factor for maintaining long-term supply contracts. The tolerance for a wide range of solvents and bases means that production is not held hostage by the shortage of a single specific reagent. This flexibility allows for agile responses to market fluctuations, ensuring that the reducing lead time for high-purity pharmaceutical intermediates becomes a reality rather than just a goal. The simplified work-up procedures also mean faster cycle times, increasing the throughput of existing manufacturing assets.
- Scalability and Environmental Compliance: As global regulations on chemical emissions tighten, the environmental footprint of a synthesis route becomes a key decision factor. This method generates significantly less acidic waste and avoids the release of toxic phosphorus byproducts. The "green" credentials of the process facilitate easier permitting for plant expansions and align with the sustainability goals of major pharmaceutical buyers. The scalability is proven by the successful demonstration of the reaction in various solvent systems, indicating that moving from pilot scale to multi-ton production can be achieved with minimal process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the feasibility and advantages of the technology.
Q: Why is the sulfonylation method superior to traditional chlorination for Blonanserin?
A: Traditional methods using phosphorus oxychloride often lead to poor selectivity and the formation of difficult-to-remove impurities where the fluorine atom is displaced. The sulfonylation route offers a better leaving group that reacts selectively at the pyridine 2-position without affecting the fluorophenyl moiety, ensuring higher purity.
Q: What represents the critical quality attribute in this synthesis?
A: The critical quality attribute is the control of 'Impurity A', a bis-substituted byproduct formed when the fluorine atom on the phenyl ring reacts. The new method minimizes this risk, allowing the final API to meet the strict requirement of not less than 99.8% purity with single impurities below 0.1%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes low-toxicity solvents and avoids hazardous chlorinating agents like POCl3. The reaction conditions are mild (often room temperature for the first step) and operationally simple, making it highly scalable and environmentally compliant for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Blonanserin Supplier
The synthesis of Blonanserin via sulfonylation represents a mature and highly optimized technology that is ready for immediate industrial deployment. At NINGBO INNO PHARMCHEM, we specialize in translating such innovative patent methodologies into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this route are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced HPLC and NMR capabilities to guarantee that every batch meets the 99.8% purity threshold required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic route for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in high-purity pharmaceutical intermediates can drive value and efficiency for your organization.
