Scalable One-Step Synthesis of 1,3,5-Trisubstituted-1,2,4-Triazoles for Pharmaceutical Applications
The pharmaceutical and agrochemical industries continuously demand efficient, scalable, and environmentally benign synthetic routes for constructing nitrogen-containing heterocycles. Patent CN111471026A introduces a groundbreaking one-step method for preparing 1,3,5-trisubstituted-1,2,4-triazole derivatives, a class of compounds pivotal in medicinal chemistry for treating conditions ranging from schizophrenia to cardiovascular diseases. This innovation addresses critical bottlenecks in existing synthesis technologies by utilizing simple nitrile and hydrazine compounds as starting materials, promoted by inexpensive bases rather than harsh Lewis acids. The significance of this patent lies in its ability to transform readily available feedstocks into high-value fine chemicals through a direct cyclization reaction that operates under remarkably mild conditions. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediates supplier, this technology represents a paradigm shift towards greener and more cost-effective manufacturing processes that do not compromise on yield or purity.
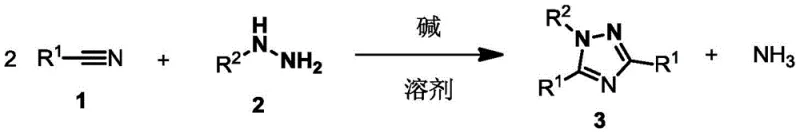
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,3,5-trisubstituted-1,2,4-triazoles has relied heavily on methods employing complex active reagents or stoichiometric amounts of strong Lewis acids like aluminum chloride (AlCl3). These traditional pathways, such as the hydrazide or hydrazone cyclization reactions, often suffer from severe limitations including harsh reaction conditions requiring temperatures between 130-170°C and the necessity for chemical equivalents of corrosive catalysts. The use of AlCl3 not only poses significant safety hazards due to its corrosive nature and the generation of hydrogen chloride gas but also creates substantial downstream processing challenges related to waste disposal and equipment maintenance. Furthermore, these older methods frequently exhibit poor functional group tolerance, limiting the substrate scope and resulting in lower yields when attempting to synthesize complex polysubstituted derivatives. The inability to use catalytic amounts of promoters in these legacy processes leads to inflated raw material costs and a larger environmental footprint, making them increasingly unsustainable for modern cost reduction in fine chemical manufacturing.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a base-promoted cyclization strategy that dramatically simplifies the synthetic workflow while enhancing efficiency. By employing bases such as potassium tert-butoxide (t-BuOK), potassium hydride (KH), or potassium hexamethyldisilazide (KHMDS), the reaction proceeds smoothly at temperatures ranging from 25-120°C, significantly lower than the thermal energy required by AlCl3-mediated processes. This method allows for the direct condensation of nitriles and hydrazines in a single step, eliminating the need for pre-functionalized intermediates and reducing the overall number of unit operations. The versatility of this system is evidenced by its compatibility with a vast array of solvents including 1,4-dioxane, toluene, and THF, providing process engineers with flexibility in optimizing reaction kinetics. Most importantly, the high atom economy and the ability to achieve yields up to 99% in optimized conditions demonstrate that this route is not merely a laboratory curiosity but a robust platform for the commercial scale-up of complex heterocyclic compounds.
Mechanistic Insights into Base-Promoted Cyclization
The core of this technological advancement lies in the mechanistic pathway where the base activates the hydrazine nucleophile, facilitating its attack on the electrophilic carbon of the nitrile group. Unlike transition metal-catalyzed methods that require expensive ligands and rigorous exclusion of air and moisture, this base-mediated cycle relies on fundamental organic reactivity principles that are easier to control on a large scale. The reaction initiates with the formation of an amidrazone intermediate, which subsequently undergoes intramolecular cyclization followed by the elimination of ammonia to furnish the stable 1,2,4-triazole ring system. This mechanism is particularly advantageous because it avoids the formation of toxic heavy metal residues, thereby simplifying the purification process and ensuring the final high-purity 1,2,4-triazole derivatives meet stringent regulatory standards for pharmaceutical applications. The absence of transition metals also removes the need for costly scavenging steps, directly contributing to process economics.
Furthermore, the reaction exhibits exceptional impurity control mechanisms due to the mildness of the basic conditions. Harsh acidic environments often lead to side reactions such as polymerization or decomposition of sensitive functional groups, but the basic medium preserves the integrity of substituents like halogens, ethers, and even amino groups. For instance, substrates containing electron-withdrawing groups like trifluoromethyl or electron-donating groups like methoxy are well-tolerated, allowing for the synthesis of diverse analogues without significant byproduct formation. The kinetic profile of the reaction, which can be completed in as little as 10 minutes for activated substrates, minimizes the residence time of intermediates, further reducing the opportunity for degradation. This level of control is essential for maintaining consistent quality in reducing lead time for high-purity pharmaceutical intermediates, ensuring that batch-to-batch variability is kept to a minimum.

How to Synthesize 1,3,5-Trisubstituted-1,2,4-Triazoles Efficiently
Implementing this synthesis route in a production environment requires careful attention to reagent quality and reaction parameters, although the procedure itself is straightforward. The patent outlines a standardized protocol where nitrile and hydrazine compounds are mixed with a base promoter in a suitable solvent under an inert atmosphere, typically within a glove box or using standard Schlenk techniques to prevent moisture interference. The reaction mixture is stirred at room temperature to ensure homogeneity before being heated to the target temperature, which is optimized based on the specific electronic nature of the substrates involved. Following the reaction completion, indicated by TLC or GC analysis, the workup involves a simple quenching step with water followed by solvent removal, after which the crude product can be purified via column chromatography. Detailed standardized synthesis steps see the guide below.
- Mix nitrile compound, hydrazine compound, base promoter (e.g., t-BuOK), and solvent in a reactor under inert atmosphere.
- Stir the mixture at room temperature then heat to 25-120°C for 1 minute to 5 hours depending on substrate reactivity.
- Quench the reaction with water, remove solvent, and purify the crude product via column chromatography to obtain the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this base-promoted synthesis offers tangible strategic benefits that extend beyond mere chemical yield. The primary advantage is the drastic simplification of the supply chain for raw materials; nitriles and hydrazines are commodity chemicals available from multiple global sources, reducing dependency on single suppliers and mitigating supply risk. Additionally, the replacement of stoichiometric aluminum chloride with catalytic or sub-stoichiometric amounts of common bases like t-BuOK significantly lowers the cost of goods sold (COGS) by reducing both reagent costs and waste disposal fees. The mild reaction conditions also translate to lower energy consumption, as the process does not require sustained high-temperature heating, aligning with corporate sustainability goals and reducing the overall carbon footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and corrosive Lewis acids removes the need for specialized corrosion-resistant reactors and complex metal removal protocols. This simplification of the infrastructure requirements leads to substantial cost savings in both capital expenditure (CapEx) and operational expenditure (OpEx). Furthermore, the high yields observed across a broad substrate scope mean that less raw material is wasted, maximizing the output per batch and improving the overall material efficiency of the plant.
- Enhanced Supply Chain Reliability: Since the method relies on stable, shelf-stable reagents that are not subject to the same supply constraints as specialized organometallic catalysts, production schedules become more predictable and resilient. The robustness of the reaction against variations in substrate electronics ensures that even if alternative grades of starting materials are sourced, the process remains viable, thereby securing the continuity of supply for critical downstream drug substances.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of benign salts and ammonia, which are easier to treat than aluminum-containing sludge. This ease of waste management facilitates smoother regulatory approvals and reduces the environmental compliance burden. The simplicity of the workup procedure, involving basic extraction and crystallization or chromatography, makes the technology highly amenable to scaling from kilogram to multi-ton production without encountering the mixing or heat transfer issues common in more complex heterogeneous catalytic systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational feasibility and scope of the method. Understanding these details is crucial for technical teams evaluating the integration of this process into existing manufacturing lines.
Q: What are the primary advantages of this base-promoted method over traditional AlCl3 methods?
A: The base-promoted method operates under significantly milder conditions (25-120°C vs 130-170°C) and avoids the use of stoichiometric amounts of corrosive aluminum chloride. This results in reduced equipment corrosion, simpler workup procedures, and eliminates the generation of hazardous aluminum waste, leading to substantial environmental and cost benefits.
Q: What is the substrate scope for this triazole synthesis?
A: The method demonstrates excellent functional group tolerance, accommodating a wide range of nitriles including aryl, heteroaryl (pyridine, furan, thiophene), and alkyl nitriles. It also tolerates various substituents such as halogens, ethers, amines, and trifluoromethyl groups, making it highly versatile for synthesizing complex pharmaceutical intermediates.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It utilizes inexpensive and readily available raw materials and bases, features short reaction times (as low as 10 minutes for some substrates), and achieves high yields (up to 99%). The simple workup involving solvent removal and chromatography facilitates efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the base-promoted cyclization technology described in CN111471026A for the production of high-value heterocyclic building blocks. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1,2,4-triazole derivatives meets the exacting standards required by the global pharmaceutical industry. We are committed to leveraging this innovative chemistry to deliver superior products that enhance your drug development pipeline.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this greener methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your supply chain resilience and your bottom line.
