Advanced Copper-Catalyzed Coupling for Scalable Diyne and Analogues Production
Advanced Copper-Catalyzed Coupling for Scalable Diyne and Analogues Production
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more sustainable, cost-effective, and scalable methodologies for constructing complex molecular architectures. A pivotal advancement in this domain is detailed in patent CN111662147B, which discloses a robust process for preparing various diynes and their analogues. This technology represents a significant leap forward for the production of high-purity pharmaceutical intermediates and functional materials. Unlike traditional methods that often rely on harsh conditions or expensive noble metal catalysts, this invention utilizes a monovalent copper catalytic system operating under remarkably mild conditions. Specifically, the process employs propargyl bromide compounds and terminal alkynes as substrates, reacting them in the presence of potassium phosphate as a base within an N,N-dimethylacetamide (DMA) solvent system at a low temperature of 40°C. This approach not only simplifies the operational complexity but also opens new avenues for the commercial scale-up of complex fine chemicals, addressing critical pain points for R&D directors and supply chain managers alike who seek reliable diyne intermediate suppliers capable of delivering consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,3-diyne and 1,4-diyne scaffolds has been fraught with significant chemical and economic challenges that hinder efficient manufacturing. The conventional Glaser-Hay coupling reaction, while foundational, typically requires a great excess of alkyne substrates to suppress competitive homocoupling side reactions, leading to poor atom economy and difficult downstream purification processes. Furthermore, the Cadiot-Chodkiewicz coupling, another staple in alkyne chemistry, necessitates the pre-functionalization of terminal alkynes into alkynyl bromides, which are notoriously unstable and hazardous to handle on a large industrial scale. Recent developments involving gold catalysts have offered improved efficiency but at the prohibitive cost of expensive noble metals and specialized ligands, making them economically unviable for bulk commodity production. Additionally, older methods often relied on strongly basic organometallic reagents like alkynyl lithium or Grignard reagents, which demand stringent anhydrous conditions and cryogenic temperatures, thereby escalating energy consumption and safety risks in a production environment.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in CN111662147B introduces a streamlined catalytic cycle that bypasses the need for unstable intermediates and expensive catalysts. By utilizing readily available propargyl bromides and terminal alkynes directly, the process achieves high selectivity for the desired cross-coupled products. The use of inexpensive monovalent copper salts, such as CuBr or CuI, combined with a mild inorganic base like potassium phosphate, drastically reduces the raw material costs associated with the synthesis. The reaction proceeds smoothly in polar aprotic solvents like DMA at a温和 temperature of 40°C, eliminating the need for energy-intensive heating or cooling systems. This technological shift allows for the construction of diverse diyne structures, including asymmetric 1,3-diynes and aryl-substituted 1,4-diynes, with moderate to excellent yields ranging significantly across different substrate combinations. The versatility of this method means it can serve as a universal platform for cost reduction in pharmaceutical intermediate manufacturing, enabling the rapid generation of diverse libraries for drug discovery without the baggage of complex waste streams.
Mechanistic Insights into Cu-Catalyzed Cross-Coupling
The efficacy of this synthetic route lies in the precise modulation of the copper catalytic cycle, which facilitates the activation of the terminal alkyne C-H bond under mild basic conditions. In this mechanism, the monovalent copper species interacts with the terminal alkyne to form a reactive copper-acetylide intermediate in situ. This nucleophilic species then attacks the electrophilic propargyl bromide substrate, likely proceeding through an SN2 or SN2' displacement pathway depending on the specific steric and electronic nature of the substituents. The presence of potassium phosphate is crucial as it acts as a proton scavenger to regenerate the active catalyst and drive the equilibrium forward without inducing elimination side reactions that are common with stronger bases. The choice of DMA as a solvent is particularly strategic; its high polarity stabilizes the charged transition states and ensures the solubility of the inorganic base, creating a homogeneous reaction environment that maximizes collision frequency between reactants. This mechanistic understanding is vital for R&D teams aiming to optimize the process further, as it highlights the delicate balance between catalyst loading, base strength, and solvent polarity required to maintain high turnover numbers.
From an impurity control perspective, this method offers distinct advantages over oxidative coupling strategies. Traditional oxidative couplings often generate symmetrical homocoupling byproducts (e.g., 1,3-butadiynes from two terminal alkynes) which are structurally similar to the target product and difficult to separate. By employing a cross-coupling strategy between a distinct electrophile (propargyl bromide) and a nucleophile (terminal alkyne), the formation of symmetrical byproducts is inherently suppressed. Furthermore, the mild reaction temperature of 40°C minimizes thermal degradation of sensitive functional groups, such as halogens or heterocycles, which might otherwise decompose under the harsher conditions required by classical methods. The patent data indicates that careful control of the molar ratio of alkyne to bromide (typically 1.2 to 2.0 equivalents) further suppresses residual starting materials, ensuring a cleaner crude profile. This inherent selectivity translates directly to reduced purification burdens, allowing for simpler workup procedures involving standard aqueous extraction and silica gel chromatography, which is a critical factor for maintaining high purity specifications in GMP environments.
How to Synthesize Diynes Efficiently
To implement this technology effectively, manufacturers must adhere to a standardized protocol that ensures reproducibility and safety. The general procedure involves the precise weighing of propargyl bromide derivatives and terminal alkynes, followed by the addition of the copper catalyst and base in an inert atmosphere. The reaction mixture is then subjected to mild heating with continuous stirring to ensure mass transfer efficiency. Detailed standard operating procedures regarding quenching, extraction, and crystallization are essential for translating this laboratory-scale success into a robust commercial process. For a comprehensive breakdown of the specific reagent quantities, reaction times, and purification techniques validated in the patent examples, please refer to the technical guide below.
- Charge a reaction vessel with propargyl bromide derivative, terminal alkyne (1.2-2.0 equiv), CuBr catalyst (10-20 mol%), and potassium phosphate base in DMA solvent.
- Maintain the reaction mixture under a nitrogen atmosphere and stir at a mild temperature of 40°C for approximately 16 hours to ensure complete conversion.
- Quench with saturated ammonium chloride, extract with ethyl acetate, dry over magnesium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology presents a compelling value proposition centered around cost stability and operational resilience. The shift away from noble metal catalysts like gold or palladium to abundant copper salts significantly de-risks the supply chain from volatile precious metal markets. Moreover, the ability to run reactions at near-ambient temperatures (40°C) drastically reduces the energy footprint of the manufacturing process, contributing to lower utility costs and a smaller carbon footprint. The use of stable, commercially available starting materials eliminates the need for complex, multi-step precursor synthesis, thereby shortening the overall lead time for high-purity diynes. This streamlined approach enhances supply chain reliability by reducing the number of potential failure points in the production sequence, ensuring a steady flow of critical intermediates for downstream API synthesis.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and specialized ligands results in substantial cost savings on raw materials. By replacing cryogenic conditions with mild heating, the process also lowers energy consumption and equipment maintenance costs associated with extreme temperature control. The high atom economy and reduced formation of difficult-to-remove byproducts minimize solvent usage and waste disposal fees, further driving down the total cost of ownership for the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on stable propargyl bromides and terminal alkynes, which are widely available from bulk chemical suppliers, mitigates the risk of raw material shortages. The robustness of the reaction conditions allows for flexible scheduling and batch processing without the need for highly specialized reactor configurations. This flexibility ensures that production timelines can be met consistently, even in the face of fluctuating market demands, providing a secure source of supply for long-term projects.
- Scalability and Environmental Compliance: The mild nature of the reaction conditions facilitates straightforward scale-up from kilogram to tonnage production without encountering the heat transfer or mixing limitations often seen in exothermic oxidative couplings. The reduced use of hazardous reagents and the generation of less toxic waste streams align with increasingly stringent environmental regulations, simplifying the permitting process for new production lines. This green chemistry profile not only ensures compliance but also enhances the corporate sustainability metrics of the final product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diyne synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a transparent view of the process capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary limitations of traditional Glaser-Hay coupling that this new method addresses?
A: Traditional Glaser-Hay coupling often suffers from homocoupling side reactions requiring a large excess of alkyne, while Cadiot-Chodkiewicz coupling necessitates unstable alkynyl bromide precursors. This novel method utilizes stable propargyl bromides and avoids excessive metal salt usage.
Q: Why is the choice of N,N-dimethylacetamide (DMA) critical for this reaction?
A: DMA serves as a polar aprotic solvent that effectively solubilizes both the organic substrates and the inorganic base (K3PO4), facilitating the nucleophilic attack under mild thermal conditions (40°C) without requiring high-energy inputs.
Q: Can this process be adapted for asymmetric 1,3-diyne synthesis?
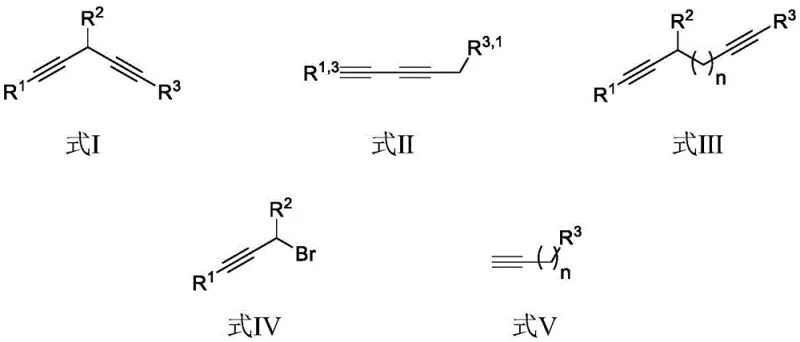
A: Yes, the method demonstrates excellent universality. By selecting specific propargyl bromides (Formula IV) and terminal alkynes (Formula V), both symmetric 1,4-diynes and asymmetric 1,3-diynes can be constructed with high selectivity and moderate to excellent yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diyne Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the copper-catalyzed coupling technology described in CN111662147B for the production of advanced pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of diyne compounds meets the exacting standards required for drug substance synthesis. We are committed to leveraging our technical expertise to optimize this green and efficient route for your specific application needs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this copper-catalyzed process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in delivering high-quality fine chemical solutions.
