Advanced Synthesis of Indolizine-Containing Diarylmethane Derivatives for Pharmaceutical Applications
The pharmaceutical industry is constantly seeking novel heterocyclic scaffolds that offer enhanced biological activity and improved synthetic accessibility. Patent CN114163436A introduces a groundbreaking class of diarylmethane derivatives containing an indolizine core, addressing the critical need for diverse chemical libraries in drug discovery. These molecules are not merely academic curiosities; they represent a strategic evolution in medicinal chemistry, combining the pharmacophore of indolizine with the structural versatility of diarylmethanes. As illustrated in the background art, related indolizine structures have demonstrated potent inhibitory activities against targets such as secreted phospholipase sPLA2 and renin, alongside significant anticancer properties. 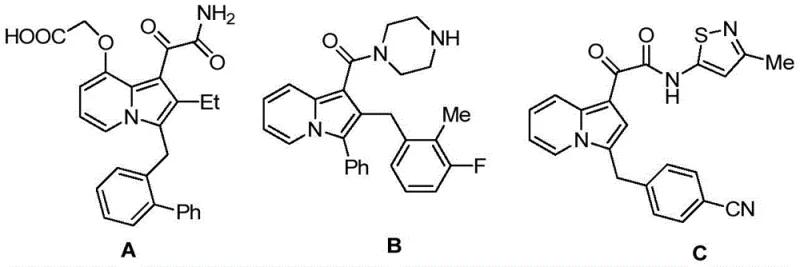 . The disclosed invention expands this chemical space by providing a robust library of derivatives defined by general formulas (I) and (II), where substituents can be finely tuned to optimize pharmacokinetic profiles.
. The disclosed invention expands this chemical space by providing a robust library of derivatives defined by general formulas (I) and (II), where substituents can be finely tuned to optimize pharmacokinetic profiles. 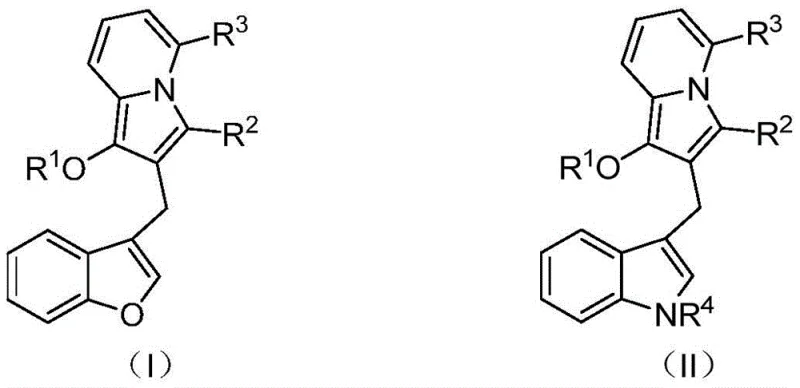 . For R&D directors and procurement specialists, this patent signifies a reliable source of high-purity pharmaceutical intermediates that can accelerate the development of next-generation therapeutics.
. For R&D directors and procurement specialists, this patent signifies a reliable source of high-purity pharmaceutical intermediates that can accelerate the development of next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of complex indolizine-diarylmethane hybrids has been fraught with synthetic challenges that hinder rapid scale-up and cost-effective production. Traditional routes often rely on multi-step sequences involving the separate preparation of the indolizine core followed by cumbersome coupling reactions to attach the diarylmethane moiety. These legacy methods frequently necessitate the use of harsh Lewis acids, extreme temperatures, or sensitive organometallic reagents that are incompatible with diverse functional groups. Furthermore, the atom economy of such stepwise approaches is often poor, generating substantial chemical waste and requiring extensive purification efforts to remove toxic metal residues. For supply chain managers, these inefficiencies translate into longer lead times, higher raw material costs, and increased regulatory burdens associated with impurity control. The inability to construct the core skeleton in a single operation has long been a bottleneck in the commercial manufacturing of these valuable bioactive intermediates.
The Novel Approach
The methodology described in patent CN114163436A represents a paradigm shift by employing a palladium-catalyzed tandem cyclization strategy that constructs the entire molecular architecture in a single pot. This innovative approach utilizes readily accessible 2-iodophenyl allene compounds and propargyl pyridine derivatives as building blocks, which undergo a seamless cascade reaction to form the target indolizine-diarylmethane scaffold. By eliminating the need for pre-functionalized heterocyclic intermediates, this novel route drastically reduces the number of unit operations required. The reaction proceeds under relatively mild thermal conditions, typically between 60°C and 90°C, which preserves sensitive functional groups and minimizes energy consumption. This streamlined synthesis not only enhances the overall yield but also simplifies the downstream processing, offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing. The versatility of this method allows for the easy incorporation of various substituents, enabling the rapid generation of analog libraries for structure-activity relationship (SAR) studies.
Mechanistic Insights into Pd-Catalyzed Tandem Cyclization
The core of this technological breakthrough lies in the elegant palladium-catalyzed mechanism that drives the formation of the indolizine ring system concurrently with the establishment of the diarylmethane linkage. The reaction initiates with the oxidative addition of the palladium(0) catalyst to the carbon-iodine bond of the 2-iodophenyl allene substrate, generating a reactive aryl-palladium species. This intermediate then undergoes a migratory insertion into the alkyne moiety of the propargyl pyridine component, a critical step that sets the stereochemistry and connectivity of the final product. Following this insertion, an intramolecular nucleophilic attack by the pyridine nitrogen onto the activated allene system triggers the cyclization event, closing the five-membered ring characteristic of the indolizine core. 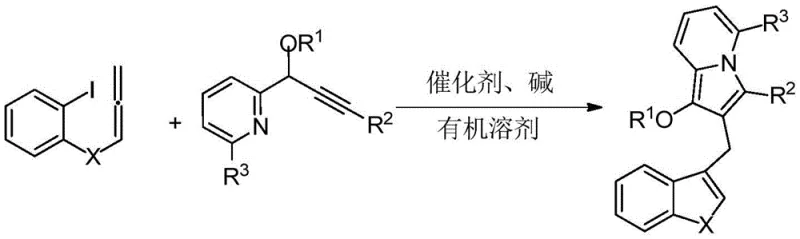 . The presence of a base, such as potassium carbonate or cesium carbonate, is essential to neutralize the acidic byproducts and regenerate the active catalytic species, ensuring the cycle continues efficiently. This mechanistic pathway is highly selective, minimizing the formation of regioisomers and side products that often plague traditional heterocycle synthesis.
. The presence of a base, such as potassium carbonate or cesium carbonate, is essential to neutralize the acidic byproducts and regenerate the active catalytic species, ensuring the cycle continues efficiently. This mechanistic pathway is highly selective, minimizing the formation of regioisomers and side products that often plague traditional heterocycle synthesis.
From an impurity control perspective, the mild reaction conditions and the specific choice of ligands on the palladium catalyst play a pivotal role in maintaining high product purity. The use of polar aprotic solvents like N,N-dimethylformamide (DMF) facilitates the solubility of both organic substrates and inorganic bases, promoting homogeneous reaction kinetics that reduce the likelihood of localized hot spots which can lead to decomposition. Furthermore, the tandem nature of the reaction means that reactive intermediates are consumed immediately upon formation, preventing their accumulation and subsequent degradation into hard-to-remove impurities. For quality assurance teams, this inherent selectivity translates to a cleaner crude product profile, reducing the burden on purification steps such as column chromatography. The ability to fine-tune the electronic properties of the substrates through substituent variation (R1, R2, R3) allows chemists to further optimize the reaction rate and selectivity, ensuring consistent batch-to-batch reproducibility essential for GMP manufacturing environments.
How to Synthesize Indolizine-Containing Diarylmethane Derivatives Efficiently
The practical implementation of this synthesis protocol is designed for operational simplicity while maintaining rigorous control over reaction parameters to ensure optimal yields. The process begins with the precise weighing of the 2-iodophenyl allene and propargyl pyridine precursors, typically in a molar ratio ranging from 1:1 to 1:2, to drive the reaction to completion. These substrates are combined with a palladium catalyst, such as tetrakis(triphenylphosphine)palladium, and an inorganic base in an anhydrous organic solvent within a sealed reaction vessel purged with inert gas. The detailed standardized synthesis steps, including specific stoichiometric ratios, temperature ramping profiles, and workup procedures, are outlined in the guide below.
- Mix 2-iodophenyl allene compound, propargyl pyridine compound, palladium catalyst (e.g., Pd(PPh3)4), alkali (e.g., K2CO3), and organic solvent (e.g., DMF) in a reaction vessel.
- Stir and react the mixture for 4-12 hours at a temperature range of 60-90°C under an inert nitrogen atmosphere to facilitate the cyclization.
- Perform post-treatment including extraction with ethyl acetate, drying over anhydrous sodium sulfate, filtration, solvent removal, and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic advantages that directly impact the bottom line and operational resilience. The primary driver of value is the significant simplification of the supply chain for raw materials; the key starting materials, 2-iodophenyl allenes and propargyl pyridines, are derived from commodity chemicals that are widely available from multiple global suppliers. This diversification of the supply base mitigates the risk of shortages and price volatility often associated with exotic or proprietary reagents. Moreover, the elimination of multiple synthetic steps reduces the total volume of solvents and reagents consumed per kilogram of product, leading to substantial cost savings in waste disposal and raw material procurement. The robustness of the reaction conditions also means that the process can be transferred to manufacturing sites with standard equipment capabilities, avoiding the need for specialized high-pressure or cryogenic infrastructure.
- Cost Reduction in Manufacturing: The one-pot nature of this tandem cyclization fundamentally alters the cost structure of producing these complex intermediates. By consolidating what would traditionally be a multi-step sequence into a single reaction vessel, manufacturers can drastically reduce labor costs, utility consumption, and equipment occupancy time. The avoidance of expensive transition metal catalysts beyond the standard palladium loading, and the use of inexpensive bases like potassium carbonate, further lowers the direct material costs. Additionally, the high atom efficiency of the cyclization minimizes the generation of chemical waste, reducing the environmental compliance costs associated with effluent treatment. These cumulative efficiencies result in a highly competitive cost position for the final API intermediate, allowing for better margin management in downstream drug production.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable starting materials ensures a consistent and predictable supply flow. Unlike processes that require freshly prepared unstable intermediates or sensitive organometallic reagents, this method utilizes components that can be stockpiled without significant degradation, providing a buffer against supply chain disruptions. The reaction's tolerance to a variety of functional groups means that the same core process can be adapted to produce a wide range of derivatives without needing to requalify entirely new synthetic routes. This flexibility allows supply chain planners to respond rapidly to changing demand signals for different analogs, enhancing the overall agility of the manufacturing operation. The simplified workup procedure, involving standard extraction and crystallization or chromatography, further ensures that production throughput is not bottlenecked by complex purification stages.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous exotherms or extreme operating conditions. The reaction can be safely conducted in standard stainless steel reactors under a nitrogen blanket, adhering to established safety protocols for organic synthesis. The use of DMF, while requiring careful handling, is a well-understood solvent in the industry with established recovery and recycling protocols, supporting sustainability goals. The reduced step count inherently lowers the E-factor (mass of waste per mass of product), aligning with green chemistry principles and facilitating easier regulatory approval for the manufacturing process. This scalability ensures that the technology can meet the demands of clinical trial material production as well as full-scale commercial manufacturing without the need for process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these indolizine derivatives, based on the detailed disclosures in the patent literature. Understanding these aspects is crucial for technical teams evaluating the feasibility of integrating this technology into their existing pipelines. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property, ensuring accuracy and relevance for decision-makers.
Q: What are the key advantages of this Pd-catalyzed method over traditional synthesis?
A: This method utilizes a one-pot tandem cyclization strategy that significantly simplifies the synthetic route compared to multi-step traditional approaches. It operates under relatively mild conditions (60-90°C) and uses readily available starting materials like 2-iodophenyl allenes and propargyl pyridines, reducing both cost and complexity.
Q: What is the biological potential of these indolizine diarylmethane derivatives?
A: The synthesized compounds combine indolizine and benzofuran or indole moieties, structures known for significant biological activities. According to the patent data, these derivatives exhibit promising antibacterial and anticancer activities, making them valuable candidates for drug discovery and development.
Q: Is this process scalable for commercial production of API intermediates?
A: Yes, the process is highly amenable to scale-up. It employs standard industrial solvents like DMF and common bases like potassium carbonate. The reaction conditions are robust, and the workup involves conventional extraction and chromatography techniques, ensuring feasibility for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolizine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in patent CN114163436A for advancing pharmaceutical research and development. As a premier CDMO partner, we possess the technical expertise and infrastructure to seamlessly translate this innovative laboratory methodology into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We operate stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of indolizine-containing diarylmethane derivatives meets the highest quality standards required for drug substance manufacturing.
We invite you to collaborate with us to leverage this advanced synthetic route for your next-generation therapeutic programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in turning complex chemical innovation into commercial reality.
