Advanced Manufacturing of Indolizine Derivatives for High-Purity Pharmaceutical Intermediates
The pharmaceutical landscape continuously demands more efficient pathways for synthesizing complex heterocyclic scaffolds, particularly those serving as key intermediates for hormonal regulation therapies. Patent CN1130909A introduces a robust and novel process for the preparation of indolizine derivatives, a class of compounds exhibiting significant pharmacological activity as testosterone 5α-reductase inhibitors. This technological breakthrough addresses the critical need for scalable manufacturing routes that can support the global demand for treatments targeting prostatic hypertrophy, prostate cancer, and androgenetic alopecia. By leveraging a direct condensation strategy between specific pyridine derivatives and functionalized alkyl chains, this method offers a distinct advantage over traditional multi-step syntheses. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials. The process outlined herein not only simplifies the synthetic route but also enhances the overall economic viability of producing these vital active pharmaceutical ingredient (API) precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indolizine core has often relied on cumbersome methodologies involving multiple protection and deprotection steps, which inherently increase production costs and extend lead times. Conventional routes frequently utilize harsh reaction conditions or expensive transition metal catalysts that pose significant challenges for impurity control and environmental compliance. These traditional methods often suffer from poor regioselectivity, leading to complex mixtures that require extensive and costly purification processes such as preparative HPLC. Furthermore, the use of unstable intermediates in older synthetic pathways can result in batch-to-batch variability, creating substantial risks for supply chain continuity. For large-scale manufacturing, the accumulation of toxic byproducts and the difficulty in recycling solvents from these inefficient processes present major hurdles. Consequently, there is a pressing industry need for a streamlined approach that minimizes waste generation while maximizing yield and purity standards.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by employing a direct reaction between a compound of formula (II) and a compound of formula (III) to generate the target indolizine structure. This innovative route bypasses many of the tedious steps associated with classical synthesis, utilizing readily available starting materials that are commercially accessible on a multi-ton scale. The reaction proceeds efficiently in common organic solvents such as alcohols, tetrahydrofuran, or dichloromethane, facilitating easier solvent recovery and waste management. By avoiding the need for exotic reagents, this process significantly lowers the barrier to entry for commercial scale-up of complex pharmaceutical intermediates. The versatility of the reaction allows for the introduction of diverse substituents at various positions on the indolizine ring, enabling the rapid generation of analog libraries for structure-activity relationship studies. 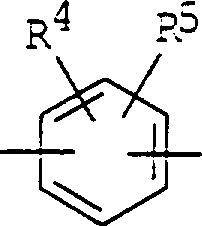
Mechanistic Insights into Base-Promoted Cyclization
The core of this synthetic strategy lies in the base-promoted nucleophilic attack and subsequent cyclization that constructs the fused bicyclic system. The reaction mechanism typically involves the deprotonation of the acidic methylene group adjacent to the carbonyl in the formula (III) precursor by an inorganic or organic base. This generates a reactive enolate species which then attacks the electrophilic center of the pyridine derivative (formula II), initiating the ring-closing sequence. The choice of base is critical, with options ranging from mild alkali metal carbonates to stronger alkoxides or amines like triethylamine, allowing fine-tuning of the reaction kinetics to minimize side reactions. This mechanistic clarity provides R&D teams with the confidence to optimize reaction parameters such as temperature and stoichiometry to achieve optimal conversion rates. Understanding this pathway is crucial for predicting potential impurities and designing effective purification protocols that ensure the final product meets stringent regulatory specifications.
Furthermore, the structural flexibility inherent in the general formulas allows for precise modulation of the physicochemical properties of the final drug substance. As illustrated in the detailed structural definitions, the R groups can be varied to include aryl, heterocyclic, or alkyl moieties, which directly influence the solubility and metabolic stability of the molecule. 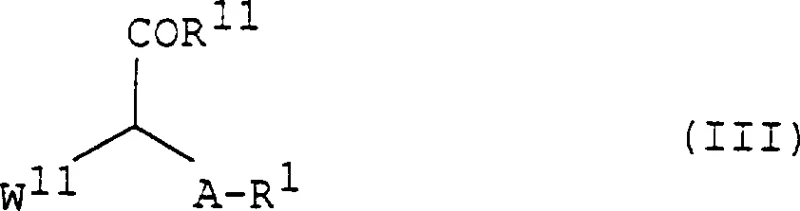
This modularity is particularly valuable for medicinal chemists aiming to improve the bioavailability of the final API. The presence of ester or protected carboxyl groups in the side chains offers convenient handles for further derivatization or salt formation, which is often necessary for formulation development. By controlling the nature of these substituents, manufacturers can tailor the impurity profile to exclude genotoxic alerts or persistent metabolites. This level of control over the molecular architecture ensures that the resulting high-purity pharmaceutical intermediates are perfectly suited for downstream processing into finished dosage forms.
How to Synthesize Indolizine Derivatives Efficiently
Implementing this synthesis requires careful attention to reaction conditions to maximize efficiency and safety. The process generally begins with the dissolution of the reactants in a suitable solvent system, followed by the controlled addition of a base to initiate the cyclization. Temperature control is maintained throughout the reaction, which can proceed from ambient conditions to moderate heating depending on the specific reactivity of the substrates. Upon completion, the target compound is isolated using standard workup procedures such as aqueous extraction and crystallization, ensuring high recovery rates. For detailed operational parameters and specific embodiment examples, please refer to the standardized guide below.
- Prepare the reaction mixture by dissolving the compound of formula (II) and the compound of formula (III) in a suitable solvent such as an alcohol, dichloromethane, or tetrahydrofuran.
- Add an inorganic or organic base, such as sodium hydroxide, potassium carbonate, or triethylamine, to facilitate the condensation and cyclization reaction.
- Stir the mixture at a controlled temperature ranging from cooling to heating, then isolate the target indolizine compound through extraction, crystallization, or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling advantages that directly address the pain points of modern pharmaceutical supply chains. The elimination of complex catalytic systems and the use of commodity chemicals translate into significant cost reduction in API manufacturing. By simplifying the synthetic route, the process reduces the total number of unit operations, thereby lowering labor costs and energy consumption associated with heating and cooling cycles. This efficiency gain is critical for maintaining competitive pricing in the generic drug market while preserving healthy margins for innovator companies. Additionally, the robustness of the reaction conditions ensures consistent quality output, which is a key metric for supply chain reliability.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing inexpensive and widely available starting materials such as substituted pyridines and adipic acid derivatives. Unlike methods requiring precious metal catalysts, this base-mediated approach eliminates the need for expensive scavenging steps to remove trace metals, which is a mandatory requirement for parenteral drugs. The simplified workflow reduces the consumption of solvents and reagents per kilogram of product, directly lowering the variable cost of goods sold. Furthermore, the high atom economy of the condensation reaction minimizes waste disposal fees, contributing to a more sustainable and economically favorable production model.
- Enhanced Supply Chain Reliability: Securing a steady supply of critical intermediates is paramount for avoiding production stoppages, and this method excels in原料 availability. The key building blocks are produced by numerous chemical suppliers globally, reducing the risk of single-source dependency. The reaction's tolerance to minor variations in raw material quality ensures that production schedules remain uninterrupted even when sourcing from different vendors. This resilience is vital for reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond quickly to market fluctuations. The ability to scale the process from laboratory benchtop to industrial reactors without fundamental changes in chemistry further guarantees long-term supply continuity.
- Scalability and Environmental Compliance: The use of standard industrial solvents like ethanol, methanol, and ethyl acetate facilitates easy technology transfer to large-scale manufacturing facilities. These solvents are well-understood in terms of safety handling and waste treatment, ensuring full compliance with increasingly strict environmental regulations. The process generates minimal hazardous byproducts, simplifying the effluent treatment process and reducing the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturing partner. Such scalability ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly to meet global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these indolizine derivatives. These insights are derived directly from the patent specifications and practical manufacturing experience. Understanding these details helps stakeholders make informed decisions regarding process adoption and vendor selection. The answers reflect the balance between technical feasibility and commercial viability inherent in this novel synthetic route.
Q: What are the primary therapeutic applications of these indolizine derivatives?
A: These compounds act as potent testosterone 5α-reductase inhibitors, making them critical intermediates for treating conditions such as prostatic hypertrophy, prostate cancer, alopecia, and acne.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the described process utilizes common inorganic or organic bases like alkali metal hydroxides or amines, significantly reducing raw material costs and eliminating heavy metal contamination risks.
Q: How does this process impact supply chain stability for API manufacturers?
A: By using readily available starting materials and standard industrial solvents, the method ensures consistent production capabilities and reduces reliance on specialized reagents that often cause supply bottlenecks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolizine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes in the competitive pharmaceutical landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to market. We are committed to delivering indolizine derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in process optimization allows us to identify and mitigate potential scale-up risks early, guaranteeing a consistent supply of high-quality intermediates for your critical drug development programs.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency. Let us be your partner in driving innovation and cost-effectiveness in the production of life-saving medications.
