Advanced Rhodium-Catalyzed Synthesis of Trifluoromethyl Dihydrophenanthrene Intermediates for Commercial Production
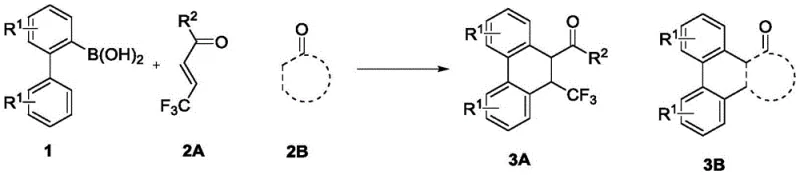
The pharmaceutical and fine chemical industries are constantly seeking efficient pathways to access complex polycyclic scaffolds that serve as critical cores for bioactive molecules. Patent CN113511966A introduces a groundbreaking synthetic method for trifluoromethyl substituted dihydrophenanthrene compounds, utilizing advanced transition metal catalysis to overcome historical bottlenecks in organic synthesis. The introduction of the trifluoromethyl group is particularly strategic, as this fluorine-containing moiety possesses strong electron-withdrawing properties and lipophilicity, which often enhances the medicinal activity, metabolic stability, and bioavailability of drug candidates. This patented technology leverages a rhodium-catalyzed system to achieve a direct [4+2] cyclization, offering a streamlined alternative to traditional multi-step sequences that often suffer from low overall throughput and excessive waste generation.
By employing 2-biphenylboronic acid compounds and alpha,beta-unsaturated ketone compounds as readily accessible starting materials, this method achieves high-yield synthesis under remarkably mild conditions. The ability to construct the dihydrophenanthrene skeleton in a single operational step represents a significant leap forward for process chemistry teams aiming to accelerate lead optimization campaigns. Furthermore, the protocol's compatibility with air and use of common organic solvents underscores its potential for seamless integration into existing manufacturing infrastructures without requiring specialized anaerobic reactors. This innovation not only addresses the scientific challenge of regioselective cyclization but also aligns with modern green chemistry principles by minimizing auxiliary reagents and simplifying downstream processing workflows for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of dihydrophenanthrene derivatives has relied on classical cyclization strategies that often impose severe constraints on process scalability and operational safety. Traditional routes frequently necessitate the use of harsh Lewis acids, stoichiometric amounts of toxic reagents, or rigorous exclusion of moisture and oxygen, which dramatically increases the cost of goods sold and complicates facility maintenance. Many prior art methods struggle with poor regioselectivity, leading to complex mixtures of 1,4-addition by-products that are difficult to separate, thereby reducing the overall isolated yield and purity of the final active pharmaceutical ingredient. Additionally, the requirement for multiple protection and deprotection steps in conventional syntheses extends the production timeline and generates substantial chemical waste, creating environmental compliance burdens for manufacturers striving to meet stringent regulatory standards.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated rhodium catalytic system that fundamentally alters the reaction trajectory to favor the desired [4+2] cyclization pathway exclusively. By leveraging the unique electronic and steric properties of the catalyst, this method bypasses the formation of unwanted addition by-products, ensuring a cleaner reaction profile that simplifies purification protocols significantly. The process operates effectively in environmentally benign solvents like ethyl acetate and tolerates the presence of air, eliminating the need for expensive inert gas purging systems and specialized glovebox equipment. This shift from sensitive, multi-step protocols to a robust, one-pot catalytic transformation represents a paradigm shift in how these valuable intermediates are produced, offering a reliable pharmaceutical intermediates supplier the ability to deliver high-quality materials with greater consistency and reduced operational risk.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Cyclization
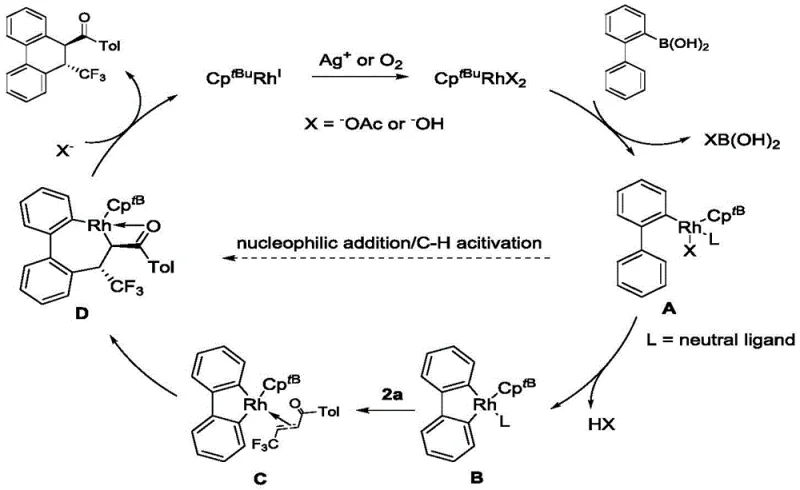
The success of this synthetic methodology hinges on the intricate interplay between the rhodium center and the bulky cyclopentadienyl ligand, which orchestrates a precise sequence of organometallic transformations. The catalytic cycle initiates with the activation of the rhodium precursor by the silver salt oxidant, generating the active cationic species capable of engaging the substrate. A critical C-H activation step occurs on the biphenylboronic acid framework, forming a stable rhodacycle intermediate that positions the metal center for subsequent migratory insertion. The steric bulk of the CptBu ligand plays a decisive role here, as it physically blocks alternative coordination modes that would lead to simple 1,4-addition, thereby enforcing the geometry required for the [4+2] annulation. This ligand-accelerated catalysis ensures that the reductive elimination step proceeds efficiently to release the cyclized product and regenerate the active catalyst, maintaining high turnover numbers throughout the reaction duration.
Furthermore, the mechanism elucidates how impurity profiles are tightly controlled through the specific design of the catalytic environment. The isolation of bridged ring compounds during mechanistic studies confirms that the reaction proceeds through a two-step Michael addition sequence that is carefully managed by the metal center. This level of mechanistic understanding allows process chemists to predict and mitigate potential side reactions, ensuring that the final high-purity API intermediate meets the rigorous specifications required for clinical development. By avoiding the use of transition metals that are difficult to remove or leave toxic residues, this rhodium-based system also facilitates easier downstream processing, reducing the burden on quality control laboratories to perform extensive heavy metal scavenging procedures before the material can be released for further synthesis.
How to Synthesize Trifluoromethyl Substituted Dihydrophenanthrene Efficiently
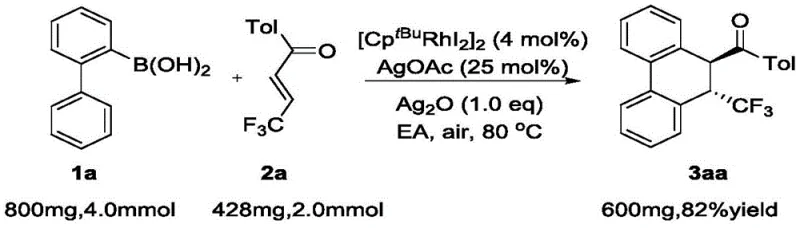
Implementing this synthesis route in a practical setting involves a straightforward protocol that balances reaction efficiency with operational simplicity. The process begins with the precise weighing of 2-biphenylboronic acid and the trifluoromethyl-substituted enone, ensuring the molar ratios align with the optimized conditions to maximize conversion. These starting materials are combined in a reaction vessel with the rhodium catalyst and silver oxidant, followed by the addition of ethyl acetate as the reaction medium. The mixture is then heated to the specified temperature range under ambient air conditions, where the transformation proceeds to completion as monitored by standard analytical techniques. Detailed standardized synthesis steps see the guide below.
- Prepare 2-biphenylboronic acid compounds and alpha,beta-unsaturated ketone compounds as starting materials in the specified molar ratios.
- Conduct the heating reaction in an organic solvent such as ethyl acetate using a rhodium catalyst and silver salt oxidant under air atmosphere.
- Quench the reaction with water, extract with organic solvent, and purify the crude product via silica gel column chromatography to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented technology offers substantial cost savings and supply chain resilience by fundamentally simplifying the manufacturing process. The elimination of strict inert atmosphere requirements means that production can be conducted in standard glass-lined reactors without the need for nitrogen or argon blanketing systems, drastically reducing utility costs and equipment complexity. Moreover, the use of ethyl acetate, a solvent with a favorable safety and environmental profile, minimizes the costs associated with solvent recovery and hazardous waste disposal, contributing to a more sustainable and economically viable production model. These factors collectively enhance the reliability of supply, as the process is less susceptible to disruptions caused by equipment failure or the unavailability of specialized gases, ensuring consistent delivery schedules for downstream partners.
- Cost Reduction in Manufacturing: The streamlined nature of this catalytic process directly translates to lower operational expenditures by removing the need for expensive anhydrous conditions and complex purification trains. By achieving high selectivity for the [4+2] cyclized product, the method minimizes the loss of valuable starting materials to by-products, thereby improving the overall mass balance and reducing the raw material cost per kilogram of finished goods. The avoidance of transition metal catalysts that require costly removal steps further optimizes the cost structure, allowing for competitive pricing in the global market for fine chemical intermediates without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures that production timelines are predictable and less prone to delays caused by sensitive operational parameters. Since the starting materials are commercially available and the reaction tolerates air, the risk of batch failure due to environmental contamination is significantly mitigated, providing procurement managers with greater confidence in supply continuity. This stability is crucial for long-term project planning, as it reduces the lead time for high-purity pharmaceutical intermediates and allows for more agile responses to fluctuations in market demand or urgent clinical trial material requirements.
- Scalability and Environmental Compliance: The demonstrated feasibility of scaling this reaction, as evidenced by successful gram-scale experiments, indicates a clear path toward commercial scale-up of complex pharmaceutical intermediates. The process generates fewer hazardous by-products and utilizes greener solvents, aligning with increasingly stringent environmental regulations and corporate sustainability goals. This compliance reduces the regulatory burden on manufacturing sites and facilitates smoother audits, ensuring that the supply chain remains uninterrupted by environmental enforcement actions while supporting the industry's shift towards more eco-friendly chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent's experimental data and beneficial effects. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing pipelines. The answers reflect the specific advantages related to catalyst performance, operational conditions, and substrate versatility described in the intellectual property documentation.
Q: Does this synthesis method require strict inert gas protection?
A: No, one of the significant advantages of this patented method is its resistance to air conditions. The reaction can be directly carried out in the air without the protection of inert gas, which simplifies operational requirements and reduces equipment costs for large-scale manufacturing.
Q: What is the role of the bulky cyclopentadienyl ligand in the catalyst?
A: The bulky cyclopentadienyl ligand (CptBu) in the rhodium catalyst effectively promotes the reduction and elimination process before proton dissociation. This steric bulk ensures the formation of the desired [4+2] cyclized product rather than the simple 1,4-addition by-product, significantly enhancing selectivity.
Q: Is the substrate scope suitable for diverse pharmaceutical applications?
A: Yes, the method demonstrates a wide application range of substrates. It tolerates various substituents on the biphenylboronic acid and the unsaturated ketone, including halogens, alkyl groups, and electron-withdrawing groups, making it highly versatile for synthesizing diverse drug lead compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Dihydrophenanthrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the pharmaceutical landscape. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like this rhodium-catalyzed method can be successfully translated into industrial reality. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of trifluoromethyl substituted dihydrophenanthrene meets the highest international standards for identity, strength, and quality, providing our clients with the assurance they need for their regulatory filings.
We invite you to collaborate with us to leverage this innovative chemistry for your next drug development program. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your journey from concept to commercial success with a reliable partner committed to excellence in fine chemical manufacturing.
