Advanced Manufacturing of Fosfomycin Intermediates for Global Pharmaceutical Supply Chains
Advanced Manufacturing of Fosfomycin Intermediates for Global Pharmaceutical Supply Chains
Introduction to Patent CN115677771A Technology
The pharmaceutical industry continuously seeks robust synthetic routes for critical antibiotic intermediates, and the technology disclosed in patent CN115677771A represents a significant leap forward in the manufacturing of fosfomycin intermediates. This specific innovation addresses the longstanding challenges associated with the synthesis of left phosphorus and right amine salt monohydrate, a pivotal building block in the production of broad-spectrum antibacterial agents. By re-engineering the traditional synthetic pathway, this method offers a solution that mitigates the high costs and procedural complexities that have historically hindered efficient industrial production. The core breakthrough lies in the strategic sequencing of salt formation and epoxidation, which fundamentally alters the impurity profile and processing requirements. For technical directors and procurement specialists, understanding the nuances of this patent is essential for evaluating potential supply chain partners who can leverage such advanced methodologies to ensure consistent quality and availability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fosfomycin intermediates has been plagued by inefficient processes that struggle to meet the rigorous demands of modern Good Manufacturing Practices. Prior art, such as the methods described in earlier patents, often relies on starting materials that necessitate extensive downstream purification to achieve acceptable stereochemical purity. A common drawback involves the use of racemic mixtures that require energy-intensive resolution steps post-epoxidation, leading to substantial material loss and increased waste generation. Furthermore, conventional routes frequently involve multiple solvent exchange operations and distillation steps, such as the removal of ethanol and petroleum ether, which not only escalate operational costs but also introduce significant safety risks due to prolonged exposure to volatile organic compounds. These inefficiencies create bottlenecks in production schedules and compromise the economic viability of large-scale manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in this patent utilizes propynyl alcohol as a cost-effective starting material, initiating a streamlined sequence that maximizes atom economy and minimizes waste. The process begins with an esterification reaction with phosphorus trichloride to generate allenylphosphoryl chloride, which is subsequently hydrolyzed to allenephosphoric acid. 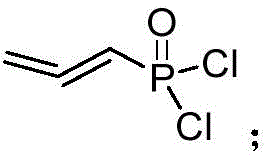 . A critical innovation is the selective hydrogenation using palladium carbon to produce cis-acrylophosphoric acid, followed by a one-pot salt formation with R-phenylethylamine. This sequence allows for the epoxidation to occur on the pre-formed salt, thereby bypassing the need for difficult chiral resolution later in the process. The result is a cleaner reaction profile with fewer by-products, making it inherently more suitable for continuous commercial production environments.
. A critical innovation is the selective hydrogenation using palladium carbon to produce cis-acrylophosphoric acid, followed by a one-pot salt formation with R-phenylethylamine. This sequence allows for the epoxidation to occur on the pre-formed salt, thereby bypassing the need for difficult chiral resolution later in the process. The result is a cleaner reaction profile with fewer by-products, making it inherently more suitable for continuous commercial production environments.
Mechanistic Insights into Sodium Tungstate-Catalyzed Epoxidation
The heart of this synthetic route lies in the epoxidation step, where the precise control of reaction conditions dictates the stereochemical outcome and overall yield. The utilization of sodium tungstate as a catalyst in conjunction with hydrogen peroxide facilitates a highly selective oxidation of the double bond in the presence of the chiral amine salt. This catalytic system operates effectively under mild pH conditions, specifically adjusted to between 5.5 and 6, which is crucial for maintaining the stability of the phosphonic acid moiety while promoting the formation of the oxirane ring. The addition of EDTA-2Na serves as a stabilizing agent, sequestering trace metal ions that could otherwise catalyze the decomposition of hydrogen peroxide or lead to unwanted side reactions. 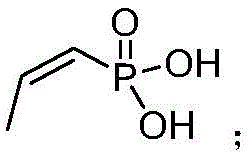 . By maintaining the temperature within a controlled range of 40-55°C during the oxidant addition, the process ensures a steady reaction rate that prevents thermal runaway while achieving high conversion efficiency.
. By maintaining the temperature within a controlled range of 40-55°C during the oxidant addition, the process ensures a steady reaction rate that prevents thermal runaway while achieving high conversion efficiency.
Impurity control is meticulously managed through the physical properties of the intermediates and the specific workup procedures employed. The initial esterification step utilizes toluene, which acts as an effective extraction solvent to remove unreacted phosphorus species and allenyl phosphate impurities before they can propagate through the synthesis. Following the hydrogenation step, the filtration of the palladium carbon catalyst is performed directly without intermediate solvent distillation, which preserves the integrity of the sensitive cis-acrylophosphoric acid. The final recrystallization involves dissolving the crude salt in ethanol, treating with activated carbon to adsorb colored impurities, and then inducing crystallization through the controlled addition of warm water. This gradient cooling and anti-solvent addition strategy ensures that the resulting crystals possess a uniform lattice structure with minimal solvent inclusion, thereby guaranteeing the high purity required for downstream antibiotic synthesis.
How to Synthesize Fosfomycin Intermediate Efficiently
Implementing this synthesis route requires strict adherence to the specified molar ratios and thermal profiles to replicate the high yields reported in the patent data. The process is designed to be telescoped where possible, reducing the number of isolation steps and minimizing the handling of hazardous intermediates. Operators must ensure precise pH control during the amine addition phase, as deviations can affect the solubility of the salt and the subsequent epoxidation kinetics. The detailed standardized synthesis steps见下方的指南。
- Perform esterification of propynyl alcohol with phosphorus trichloride in toluene to generate allenylphosphoryl chloride.
- Hydrolyze the chloride to allenephosphoric acid followed by selective palladium-catalyzed hydrogenation to form cis-acrylophosphoric acid.
- Conduct one-pot salt formation with R-phenylethylamine and sodium tungstate-catalyzed epoxidation to yield the crude salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible operational improvements that extend beyond mere technical specifications. The elimination of complex resolution steps and the reduction in solvent usage directly correlate to a simplified manufacturing workflow, which enhances the reliability of supply timelines. By avoiding energy-intensive distillation processes, the production facility can significantly lower its utility consumption, contributing to a more sustainable and cost-effective operation. This efficiency gain allows manufacturers to offer more competitive pricing structures without compromising on the stringent quality standards demanded by the pharmaceutical sector. Furthermore, the robustness of the process under mild conditions reduces the risk of batch failures, ensuring a consistent flow of materials to meet global demand.
- Cost Reduction in Manufacturing: The streamlined process architecture eliminates the need for expensive chiral resolution columns and reduces the volume of organic solvents required for purification. By integrating the salt formation and epoxidation into a cohesive one-pot sequence, labor hours and equipment occupancy time are drastically reduced. The removal of ethanol distillation steps further cuts down on energy costs associated with heating and vacuum systems. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, offering a distinct economic advantage over suppliers relying on legacy synthetic routes.
- Enhanced Supply Chain Reliability: The use of readily available starting materials like propynyl alcohol and common reagents such as hydrogen peroxide ensures that raw material sourcing remains stable and unaffected by niche market fluctuations. The simplified process flow reduces the number of critical control points where production delays could occur, thereby enhancing the predictability of lead times. Additionally, the high stability of the intermediates allows for flexible scheduling and inventory management, providing a buffer against unexpected demand surges. This reliability is crucial for maintaining uninterrupted production lines for finished antibiotic formulations.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor configurations and avoiding extreme pressure or temperature conditions that require specialized equipment. The reduction in waste generation, particularly through the minimized use of petroleum ether and ethanol, aligns with increasingly stringent environmental regulations and corporate sustainability goals. Lower waste volumes translate to reduced disposal costs and a smaller environmental footprint. This compliance readiness ensures long-term operational viability and reduces the regulatory burden on manufacturing partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and quality assurance of this fosfomycin intermediate. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits for potential partners. Understanding these details helps stakeholders assess the feasibility of integrating this material into their existing supply networks.
Q: How does this method improve upon the traditional Glamkowski synthesis route?
A: Unlike the Glamkowski method which requires complex resolution steps after epoxidation, this patented process utilizes R-phenylethylamine for direct salt formation prior to epoxidation. This strategic sequence eliminates the need for separating corresponding isomers post-reaction, significantly simplifying the workflow and enhancing overall process efficiency for industrial applications.
Q: What specific measures ensure the high purity of the final intermediate?
A: Purity is rigorously maintained through a multi-stage purification strategy. Initially, toluene extraction effectively removes allenyl phosphate impurities generated during esterification. Subsequently, the process avoids energy-intensive ethanol distillation by filtering directly before epoxidation. Finally, a specialized recrystallization step using activated carbon and controlled water addition ensures the removal of trace organic impurities and color bodies.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process is explicitly designed for scalability. It operates under relatively mild conditions, primarily utilizing low-temperature controls which enhance operational safety. Furthermore, the reduction in solvent consumption and the elimination of complex separation units lower the barrier for scaling from pilot batches to multi-ton annual production capacities without compromising product consistency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fosfomycin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate complex patent methodologies like CN115677771A into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial volume is seamless and compliant. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify every batch against the highest international standards. Our commitment to quality ensures that the fosfomycin intermediates we supply meet the exacting requirements of global pharmaceutical manufacturers.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through advanced chemical solutions. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals. Let us be your partner in delivering high-quality chemical intermediates with unmatched reliability and efficiency.
