Advanced Iridium-Catalyzed Synthesis of Allyl Alcohol Derivatives for Commercial Scale-Up
Advanced Iridium-Catalyzed Synthesis of Allyl Alcohol Derivatives for Commercial Scale-Up
The chemical industry continuously seeks robust methodologies for constructing complex molecular architectures, particularly for high-value sectors like liquid crystal displays and pharmaceutical active ingredients. Patent CN111072450B introduces a transformative synthesis method for allyl alcohol derivatives that addresses long-standing inefficiencies in traditional manufacturing routes. By leveraging a novel iridium-complex catalytic system combined with alkali metal bicarbonates, this technology enables the selective conversion of stable 3-chloropropyl derivatives into valuable allyl alcohol intermediates with exceptional precision. This breakthrough is particularly significant for R&D directors and procurement specialists looking to optimize the production of trans-alkene precursors, which are critical building blocks in modern material science and drug discovery. The process not only simplifies the synthetic pathway but also drastically improves the economic feasibility of producing these specialized chemicals on a commercial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of allyl alcohol derivatives has relied heavily on the reaction of aldehydes with vinyl magnesium chloride Grignard reagents, a pathway fraught with significant logistical and chemical challenges. As illustrated in the reaction scheme below, this traditional approach requires aldehyde starting materials that are often difficult to source directly from the market, necessitating their prior synthesis from acids or acyl chlorides.
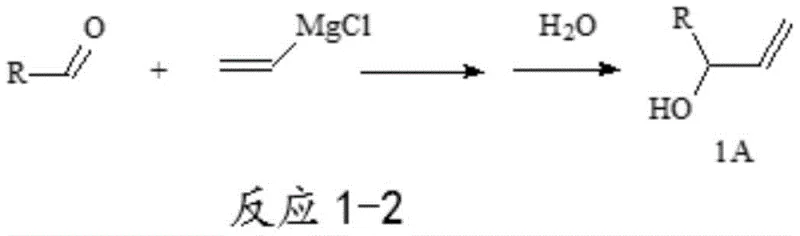
Furthermore, the industrial production of these specific aldehyde compounds frequently suffers from low yields, generates substantial environmental pollution due to harsh oxidation or reduction steps, and relies on expensive reagents that inflate the overall cost of goods. An alternative conventional method involves the selective reduction of alpha-carbonylpropene compounds; however, these substrates are chemically unstable, prone to self-polymerization upon heating or prolonged storage, and are significantly more costly to produce than the corresponding aldehydes. These inherent drawbacks create a bottleneck for supply chain managers who require consistent, high-volume availability of raw materials without the risk of degradation or exorbitant pricing.
The Novel Approach
In stark contrast to these legacy methods, the patented technology utilizes readily available organic acyl chlorides and ethylene to generate stable 3-chloropropyl derivatives, which serve as the pivotal intermediates for the final transformation. The core innovation lies in the subsequent reaction where these 3-chloropropyl derivatives are treated with dialkyl dihydro-silanes under the influence of an iridium complex catalyst and an alkali metal bicarbonate promoter. This specific combination allows for a highly selective reduction that preserves the terminal double bond while installing the hydroxyl group, effectively bypassing the instability issues associated with alpha-carbonyl alkenes.
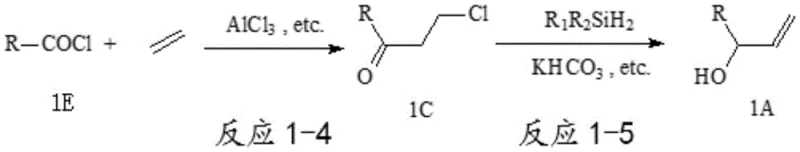
The overall synthetic route, depicted above, demonstrates a streamlined two-step process that begins with a Friedel-Crafts type addition of ethylene to the acyl chloride, followed by the catalytic reduction. This approach leverages the abundance and low cost of ethylene and acyl chlorides, providing a reliable allyl alcohol derivative supplier with a distinct competitive advantage. By replacing the unstable and expensive alpha-carbonyl alkene compounds with robust 3-chloropropyl derivatives, the method ensures that the yield of both the preparation reaction and the subsequent reduction can consistently exceed 90%, making it ideally suited for large-scale industrial production.
Mechanistic Insights into Iridium-Catalyzed Selective Reduction
The success of this synthesis hinges on the precise interplay between the iridium complex catalyst and the alkali metal bicarbonate additive, a mechanistic nuance that was critical to the invention's development. Without the bicarbonate promoter, the reaction of the 3-chloropropyl derivative with dialkyldihydrosilane would proceed via a non-selective pathway, reducing the carbonyl group to a saturated alcohol (compound 1D) rather than the desired allyl alcohol (compound 1A). The inventors discovered that the introduction of the bicarbonate acts as a crucial reaction auxiliary agent, facilitating a selective dehydrohalogenation concurrent with the reduction to ensure the formation of the terminal alkene functionality.
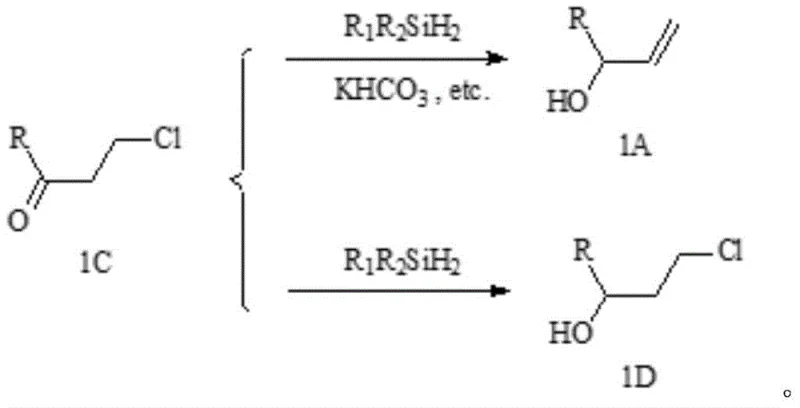
This selectivity mechanism is vital for maintaining high purity specifications, as the formation of the saturated byproduct would complicate downstream purification and reduce the overall efficiency of the process. The iridium catalyst, selected from dimers such as cyclooctene iridium chloride or hydroxide variants, operates effectively at mild temperatures ranging from 20 to 60°C, minimizing energy consumption and thermal stress on the reactants. For R&D teams, understanding this catalytic cycle is essential for troubleshooting and optimizing reaction conditions, as the specific ratio of silane to substrate (0.6 to 1.0 molar equivalents) and the choice of solvent (alcohols or ethers) are tuned to maximize the solubility of the bicarbonate while preventing excessive viscosity.
Furthermore, the impurity profile of this reaction is remarkably clean due to the stability of the 3-chloropropyl intermediate under the non-alkaline reaction conditions employed in the first step. The subsequent hydrolysis step, performed at controlled temperatures of 20-40°C with pH adjustment to 3-6, ensures that any remaining silyl intermediates are quenched efficiently without promoting side reactions. This rigorous control over the reaction environment allows for the production of high-purity allyl alcohol derivatives with gas chromatography purities reaching approximately 99% after simple crystallization, thereby reducing the burden on quality control laboratories and ensuring consistent batch-to-batch reproducibility.
How to Synthesize Allyl Alcohol Derivatives Efficiently
The practical implementation of this synthesis involves a carefully orchestrated sequence of addition, temperature control, and workup procedures designed to maximize safety and yield. The process begins with the preparation of the 3-chloropropyl derivative via the reaction of an acyl chloride with ethylene in the presence of a Lewis acid, followed by the critical iridium-catalyzed reduction step where the bicarbonate promoter is introduced to drive selectivity. Detailed operational parameters, including specific solvent choices like tetrahydrofuran or methanol and precise temperature ramps, are essential for replicating the high yields reported in the patent examples.
- Prepare 3-chloropropyl derivative (1C) by reacting organic acyl chloride with ethylene using a Lewis acid catalyst like AlCl3 at low temperatures (-10 to 20°C).
- React compound 1C with dialkyldihydrosilane in the presence of an iridium complex catalyst and alkali metal bicarbonate promoter at 20-60°C.
- Perform hydrolysis and workup by filtering the reaction mixture, adding water, adjusting pH to 3-6, and extracting to isolate the high-purity allyl alcohol derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the fundamental shift of raw material sourcing; by utilizing organic acyl chlorides and ethylene instead of specialized aldehydes or unstable alpha-carbonyl alkenes, manufacturers can tap into a much broader and more stable global supply base. Acyl chlorides are commodity chemicals produced in vast quantities for various industries, ensuring that the cost of raw materials is significantly lower and less susceptible to the volatility often seen with niche fine chemical intermediates. This substitution effectively decouples the production of high-value allyl alcohol derivatives from the bottlenecks associated with complex aldehyde synthesis.
- Cost Reduction in Manufacturing: The elimination of expensive and unstable starting materials directly translates to a substantial decrease in the cost of goods sold. Since the process avoids the need for pre-synthesizing difficult aldehydes or handling polymerization-prone alkenes, the operational expenditure related to raw material inventory and waste disposal is drastically simplified. Furthermore, the high yields exceeding 90% per step mean that less feedstock is required to produce the same amount of final product, enhancing the overall atom economy and reducing the financial impact of material loss during production.
- Enhanced Supply Chain Reliability: The stability of the 3-chloropropyl intermediate under non-alkaline conditions allows for flexible manufacturing schedules and potential stockpiling of intermediates without the risk of degradation. This robustness mitigates the risk of production delays caused by raw material spoilage, a common issue with the alpha-carbonyl alkene precursors used in conventional methods. Consequently, suppliers can offer more reliable lead times and guarantee continuous supply continuity even during periods of high market demand, which is critical for downstream clients in the pharmaceutical and electronics sectors.
- Scalability and Environmental Compliance: The reaction conditions are mild, operating at temperatures between 20 and 60°C, which reduces the energy load on manufacturing facilities and simplifies the engineering requirements for reactor cooling and heating systems. The use of common solvents like alcohols and ethers, combined with the absence of heavy metal contaminants often associated with other catalytic systems, streamlines the wastewater treatment process. This environmental friendliness facilitates easier regulatory compliance and permits faster scale-up from pilot plant to multi-ton commercial production without the need for extensive new infrastructure investments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iridium-catalyzed synthesis method. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on the operational nuances and strategic benefits of adopting this technology for industrial applications.
Q: Why is alkali metal bicarbonate critical in this synthesis?
A: The patent identifies that without the bicarbonate promoter, the dialkyldihydrosilane reduces the 3-chloropropyl derivative directly to the saturated alcohol (1D) rather than the desired allyl alcohol (1A). The bicarbonate ensures selective dehydrohalogenation and reduction to form the double bond.
Q: What are the advantages of using acyl chlorides over aldehydes?
A: Acyl chlorides are generally more stable and commercially available at lower costs compared to the specific aldehyde precursors required in traditional Grignard routes. Additionally, the intermediate 3-chloropropyl derivatives exhibit superior stability under non-alkaline conditions, facilitating storage and handling.
Q: What yields can be expected from this iridium-catalyzed process?
A: According to the patent data, both the preparation of the 3-chloropropyl intermediate and the subsequent selective reduction step achieve yields exceeding 90%, resulting in a robust total yield suitable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Alcohol Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the fast-paced world of fine chemical manufacturing. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of allyl alcohol derivative performs consistently in your downstream applications, whether for liquid crystal alignment layers or complex pharmaceutical syntheses.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce manufacturing costs by integrating this superior synthesis method into your production portfolio today.
