Advanced Synthesis of Tetraazido Alkanes for Next-Generation Energetic Material Applications
Advanced Synthesis of Tetraazido Alkanes for Next-Generation Energetic Material Applications
The development of high-energy density materials is a critical frontier in the aerospace and defense sectors, driving the demand for novel energetic plasticizers that can enhance propellant performance without compromising stability. Patent CN102603560A introduces a groundbreaking methodology for the synthesis of a specific tetraazido alkane, identified as 2,6-diazido-1,7-diazidoheptane, which addresses the longstanding limitations of existing azide-based compounds. This innovation leverages a robust four-step organic synthesis pathway to construct a carbon backbone densely functionalized with azide groups, each contributing substantial formation enthalpy to the final energetic matrix. For R&D directors and procurement specialists in the energetic materials industry, this patent represents a significant opportunity to access a reliable energetic material intermediate supplier capable of delivering compounds with superior burning characteristics. The technical disclosure outlines a reproducible process that transitions from simple diethyl malonate precursors to a complex, high-nitrogen content molecule, promising to redefine the performance benchmarks for solid propellant formulations.
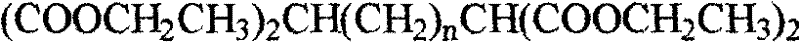
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the formulation of solid propellants has relied heavily on azide derivatives of pentaerythritol, which, while providing four azide groups for energy enhancement, have been plagued by inherent kinetic limitations. The primary drawback of these conventional pentaerythritol-based plasticizers is their relatively slow burning speed, which restricts their utility in high-performance propulsion systems where rapid energy release is paramount. Furthermore, the synthesis of highly branched azide compounds often involves complex stereochemical controls and harsh reaction conditions that can lead to unpredictable impurity profiles, posing risks to the long-term storage stability of the munition. These traditional methods frequently struggle to balance the trade-off between high nitrogen content and favorable rheological properties, often resulting in formulations that are either too viscous for processing or too unstable for safe handling. Consequently, the industry has been searching for alternative molecular architectures that can decouple energy density from burning rate limitations, a challenge that previous synthetic routes have failed to adequately resolve.
The Novel Approach
The methodology disclosed in CN102603560A offers a transformative solution by utilizing a linear alkane backbone functionalized with four terminal azide groups, specifically designed to optimize both energy output and combustion kinetics. This novel approach employs a sequential synthetic strategy that begins with the construction of a stable carbon skeleton via the alkylation of diethyl malonate, followed by a exhaustive reduction to generate a tetra-alcohol intermediate. Unlike the rigid cage structure of pentaerythritol derivatives, the flexible heptane chain allows for better compatibility with polymeric binders like HMX and RDX, enhancing the mechanical properties of the cured propellant grain. The process is characterized by its operational simplicity and the use of standard laboratory reagents, which significantly lowers the barrier for cost reduction in energetic chemical manufacturing. By shifting from a branched polyol core to a linear dialkylated malonate derivative, the inventors have created a molecule that burns faster and more completely, directly addressing the critical performance gap identified in prior art.
Mechanistic Insights into Multi-Step Azide Functionalization
The core of this synthetic innovation lies in the precise execution of nucleophilic substitution reactions to install the high-energy azide functionalities onto a pre-formed carbon scaffold. The process initiates with the formation of a tetra-ester intermediate, which serves as the foundational block for the entire molecule. This intermediate is then subjected to a vigorous reduction using lithium aluminum hydride (LiAlH4) in anhydrous ether, a reaction that quantitatively converts the four ester moieties into primary hydroxyl groups. This reduction step is critical as it generates the necessary leaving group handles for the subsequent functionalization. The resulting tetra-alcohol, 2,6-dimethylol-1,7-heptanediol, is a versatile synthon that allows for the controlled introduction of leaving groups, typically via tosylation, preparing the molecule for the final, energy-defining transformation.
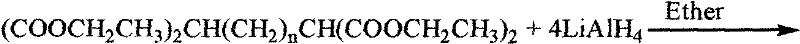
The final and most crucial stage of the synthesis involves the displacement of tosylate groups with azide ions through an SN2 mechanism in a polar aprotic solvent like DMF. This reaction is conducted at elevated temperatures (80°C) to overcome the activation energy barrier associated with substituting four sterically demanding groups on the same molecule. The use of sodium azide in excess ensures that the reaction proceeds to completion, minimizing the presence of partially substituted intermediates which could act as instability points in the final energetic material. The mechanistic efficiency of this step is evidenced by the high isolated yield of 85%, indicating a clean conversion with minimal side reactions such as elimination or rearrangement. This high fidelity in chemical transformation is essential for producing high-purity energetic intermediates that meet the stringent safety specifications required for military and aerospace applications, ensuring that the final propellant behaves predictably under extreme conditions.
How to Synthesize 2,6-Diazido-1,7-Diazidoheptane Efficiently
The synthesis of this high-energy tetraazido alkane is achieved through a logical sequence of organic transformations that prioritize yield and purity at every stage. The process begins with the generation of a sodium ethoxide solution, which acts as a strong base to deprotonate diethyl malonate, facilitating its nucleophilic attack on 1,3-dibromopropane to form the central carbon chain. Following the isolation of the tetra-ester, the protocol moves to a reduction phase using lithium aluminum hydride, requiring strict moisture control to prevent hazardous hydrogen evolution. The subsequent activation of the hydroxyl groups via tosylation creates excellent leaving groups, setting the stage for the final azidation. For a detailed breakdown of the specific molar ratios, temperature profiles, and workup procedures required to replicate this synthesis safely and effectively, please refer to the standardized guide below.
- Synthesize the tetraethyl ester backbone via alkylation of diethyl malonate with 1,3-dibromopropane under nitrogen protection.
- Perform a four-fold reduction using lithium aluminum hydride in anhydrous ether to convert ester groups to primary hydroxyl groups.
- Execute nucleophilic substitution using sodium azide in DMF at 80°C to replace tosylate groups with azide functionalities.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial strategic benefits for organizations looking to optimize their supply chain for energetic materials. The reliance on commodity chemicals such as diethyl malonate, sodium azide, and common solvents like ethanol and ether means that the raw material supply is robust and less susceptible to the geopolitical volatility often associated with specialized precursors. This accessibility translates directly into enhanced supply chain reliability, as manufacturers are not dependent on single-source suppliers for exotic starting materials. Furthermore, the process avoids the use of expensive transition metal catalysts or complex chiral ligands, which simplifies the procurement landscape and reduces the overall cost of goods sold. The ability to source inputs from multiple vendors ensures continuity of supply, a critical factor for long-term defense contracts where production interruptions are not an option.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy of the alkylation steps and the use of inexpensive inorganic salts for the functionalization stages. By eliminating the need for precious metal catalysts and reducing the number of purification steps required compared to older methods, the overall manufacturing overhead is significantly lowered. The high yields reported in the patent, particularly in the final azidation step, minimize waste generation and maximize the output per batch, leading to substantial cost savings in large-scale production environments. Additionally, the recycling of solvents like ether and ethanol further contributes to the economic efficiency of the process, making it a financially attractive option for commercial scale-up.
- Enhanced Supply Chain Reliability: The synthetic pathway is designed with resilience in mind, utilizing reagents that are widely available in the global chemical market. This diversification of the supply base mitigates the risk of bottlenecks that can occur when relying on niche intermediates. The robustness of the reaction conditions, which tolerate standard industrial equipment and do not require cryogenic temperatures or ultra-high pressures, ensures that production can be maintained consistently across different manufacturing sites. This flexibility allows for a more agile supply chain capable of responding to fluctuating demand signals without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial quantities is facilitated by the straightforward nature of the unit operations involved, such as reflux, distillation, and crystallization. The waste streams generated are primarily aqueous salts and organic solvents, which can be managed through established treatment protocols, ensuring compliance with environmental regulations. The absence of heavy metal residues simplifies the waste disposal process and reduces the environmental footprint of the manufacturing facility. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile of the manufacturer, a growing priority for government and commercial buyers alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this tetraazido alkane, based on the detailed disclosures within the patent literature. Understanding these aspects is vital for stakeholders evaluating the feasibility of integrating this material into their existing product lines. The answers provided reflect the specific experimental data and comparative advantages highlighted in the invention summary.
Q: What are the key advantages of this tetraazido alkane over pentaerythritol derivatives?
A: Unlike pentaerythritol derivatives which suffer from slow burning speeds limiting their application, this novel tetraazido alkane exhibits a significantly faster combustion rate while maintaining excellent plasticizing properties and compatibility with HMX and RDX.
Q: How does the synthesis process ensure high purity for energetic applications?
A: The process utilizes rigorous purification steps including vacuum distillation, recrystallization from methanol and toluene, and column chromatography, ensuring the removal of unreacted precursors and by-products critical for stable energetic formulations.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the method employs standard organic transformations like ester alkylation and nucleophilic substitution with readily available reagents like sodium azide and LiAlH4, demonstrating high yields (up to 85% in the final step) suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetraazido Alkane Supplier
As the global demand for high-performance energetic materials continues to rise, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides a strategic advantage in bringing complex molecules like 2,6-diazido-1,7-diazidoheptane to market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and quality of every batch, guaranteeing that the energetic performance matches the theoretical predictions. Our commitment to safety and quality makes us the preferred partner for defense contractors and aerospace firms seeking dependable sources of critical intermediates.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how optimizing this supply chain can benefit your bottom line. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven expertise. Let us help you accelerate your development timeline with our reliable supply of next-generation energetic material precursors.
