Scaling High-Energy Tetraazido Alkanes for Next-Generation Propellant Systems
Scaling High-Energy Tetraazido Alkanes for Next-Generation Propellant Systems
The development of advanced energetic materials requires a delicate balance between high energy density, thermal stability, and processability, a challenge that is effectively addressed by the novel synthesis methodology disclosed in patent CN102603560A. This intellectual property outlines a robust pathway for producing 2,6-diazido-1,7-diazidoheptane, a specialized tetraazido alkane that serves as a superior plasticizer and binder in solid propellant formulations. By integrating four high-energy azide groups into a flexible alkane backbone, this compound delivers a substantial increase in formation enthalpy, specifically contributing approximately 356kJ/mol per azide group, without compromising the carbon-hydrogen ratio essential for clean combustion. For R&D directors and procurement specialists in the defense and aerospace sectors, understanding the nuances of this synthesis is critical for securing a reliable energetic material intermediate supplier capable of meeting stringent performance specifications. The following analysis dissects the technical merits and commercial viability of this route, highlighting its potential to redefine cost reduction in propellant manufacturing through efficient, scalable chemistry.
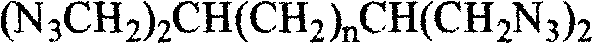
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the formulation of high-energy propellants has relied heavily on azide derivatives of pentaerythritol, which, while capable of providing significant energy due to their four azide groups, suffer from inherent kinetic limitations during combustion. These conventional derivatives often exhibit slow burning rates, which restricts their utility in applications requiring rapid thrust generation or specific impulse modulation, thereby limiting the widespread adoption of these preparation technologies in modern weapon systems. Furthermore, the synthesis of traditional azido compounds often involves complex purification challenges where residual unreacted azides or unstable byproducts can compromise the safety profile of the final energetic material. The incompatibility of certain older azide structures with common oxidizers like ammonium perchlorate or binders like HTPB can also lead to phase separation and mechanical instability in the cured propellant grain. Consequently, the industry has faced a persistent bottleneck in finding azido compounds that offer both the high nitrogen content required for increased exhaust volume and the rapid burn rates necessary for advanced propulsion without elevating flame temperatures to damaging levels.
The Novel Approach
The methodology presented in the patent data introduces a groundbreaking structural alternative, 2,6-diazido-1,7-diazidoheptane, which overcomes the burning rate deficiencies of pentaerythritol derivatives while maintaining excellent plasticizing performance. This novel approach utilizes a linear alkane backbone with strategically placed branching points that allow for the attachment of four azide groups, resulting in a molecule that is fully compatible with standard explosive fillers like HMX and RDX. The synthesis route is characterized by its simplicity and stability, avoiding the use of highly sensitive intermediates that plague other azide syntheses, thus enhancing the overall safety of the manufacturing process. By employing a stepwise construction starting from commodity chemicals like diethyl malonate and 1,3-dibromopropane, the process ensures a high degree of reproducibility and yield, which is essential for consistent batch-to-batch quality in industrial settings. This strategic shift not only improves the ballistic performance of the final propellant but also simplifies the supply chain by relying on readily available raw materials rather than specialized, hard-to-source precursors.
Mechanistic Insights into Multi-Step Azide Installation
The core of this synthesis lies in a carefully orchestrated four-step sequence that transforms simple esters into a high-energy tetraazide, beginning with the alkylation of diethyl malonate to form a tetraester intermediate. Under strict nitrogen protection to prevent moisture ingress, sodium ethoxide acts as a base to deprotonate the malonate, facilitating a double alkylation with 1,3-dibromopropane to create the carbon skeleton of 1,1,5,5-pentanetetracarboxylate tetraethyl ester. This step is critical for establishing the symmetry of the molecule, which is later exploited to ensure uniform functionalization of all four terminal positions. Following the formation of the ester, the process moves to a vigorous reduction phase where lithium aluminum hydride (LiAlH4) in anhydrous ether serves as the reducing agent to convert the four ester groups into primary hydroxyl groups. This reduction must be conducted at controlled low temperatures, specifically around -5°C in an ice bath, to manage the exothermic nature of the reaction and prevent the decomposition of the sensitive hydride reagent, ultimately yielding 2,6-dimethylol-1,7-heptanediol with high fidelity.
The subsequent activation of these hydroxyl groups is achieved through tosylation, where p-toluenesulfonyl chloride reacts with the tetraol in a pyridine solution to install excellent leaving groups at all four reactive sites. This transformation is pivotal because it converts the relatively inert alcohol functionalities into highly reactive tosylates, priming the molecule for the final nucleophilic substitution. The final and most critical step involves the displacement of these tosylate groups with azide ions using sodium azide in a polar aprotic solvent like DMF at elevated temperatures of 80°C. 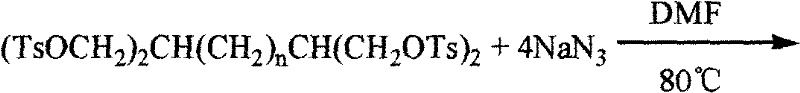 This SN2 reaction mechanism ensures the inversion of configuration at the carbon centers, although given the primary nature of the carbons involved, stereochemical issues are minimized, leading to the clean formation of the target 2,6-diazido-1,7-diazidoheptane. The use of DMF as a solvent is particularly advantageous here as it solvates the sodium cation effectively, increasing the nucleophilicity of the azide anion and driving the reaction to completion within a reasonable timeframe of 12 to 15 hours.
This SN2 reaction mechanism ensures the inversion of configuration at the carbon centers, although given the primary nature of the carbons involved, stereochemical issues are minimized, leading to the clean formation of the target 2,6-diazido-1,7-diazidoheptane. The use of DMF as a solvent is particularly advantageous here as it solvates the sodium cation effectively, increasing the nucleophilicity of the azide anion and driving the reaction to completion within a reasonable timeframe of 12 to 15 hours.
How to Synthesize 2,6-Diazido-1,7-diazidoheptane Efficiently
Executing this synthesis requires strict adherence to the patented conditions to maximize yield and ensure operator safety, particularly given the energetic nature of the final product. The process begins with the preparation of the tetraester backbone, followed by reduction, activation, and finally azidation, with each step requiring specific workup procedures such as vacuum distillation and recrystallization to remove impurities. Detailed standard operating procedures regarding stoichiometry, temperature ramps, and quenching protocols are essential for reproducing the reported yields of 60% for the ester, 78% for the alcohol, 80% for the tosylate, and 85% for the final azide. For a comprehensive breakdown of the exact operational parameters and safety measures required for each stage, please refer to the standardized guide below.
- Perform alkylation of diethyl malonate with 1,3-dibromopropane under nitrogen protection to form tetraethyl 1,1,5,5-pentanetetracarboxylate.
- Reduce the tetraester intermediate using lithium aluminum hydride in anhydrous ether at low temperature (-5°C) to obtain the tetraol.
- Activate the hydroxyl groups by reacting the tetraol with p-toluenesulfonyl chloride in pyridine to form the tetratosylate derivative.
- Execute nucleophilic substitution using sodium azide in DMF at 80°C to install the four azide groups, yielding the final energetic product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers distinct advantages that directly address the pain points of procurement managers and supply chain heads in the energetic materials sector. By utilizing a linear synthetic pathway that avoids the need for precious metal catalysts or exotic reagents, the process significantly reduces the raw material costs associated with producing high-energy plasticizers. The reliance on commodity chemicals such as diethyl malonate, 1,3-dibromopropane, and sodium azide ensures that the supply chain remains resilient against market fluctuations that often affect specialized fine chemical intermediates. Furthermore, the robustness of the reaction conditions, which operate at atmospheric pressure and moderate temperatures, lowers the capital expenditure required for reactor infrastructure, making it easier to scale production from pilot plants to full commercial volumes without extensive re-engineering.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of stoichiometric reagents like lithium aluminum hydride and sodium azide, which are industrially abundant, leads to substantial cost savings in the overall bill of materials. The high yields reported at each stage, particularly the 85% yield in the final azidation step, minimize waste generation and reduce the cost per kilogram of the active pharmaceutical ingredient equivalent in energetic terms. Additionally, the purification methods described, such as simple extraction and recrystallization, are far less expensive than chromatographic separations often required for more complex chiral molecules, further driving down the operational expenses.
- Enhanced Supply Chain Reliability: Because the starting materials are bulk chemicals produced by numerous global suppliers, the risk of supply disruption is drastically reduced compared to routes dependent on single-source proprietary intermediates. The synthesis does not require cryogenic conditions below -20°C or ultra-high vacuum systems, meaning that production can be maintained in standard chemical manufacturing facilities with existing utility setups. This accessibility ensures that lead times for high-purity energetic intermediates can be kept short, allowing propellant manufacturers to respond quickly to changing demand schedules without being bottlenecked by raw material availability.
- Scalability and Environmental Compliance: The process generates waste streams that are well-understood and manageable, such as aqueous salts from the neutralization steps and organic solvents that can be recovered and recycled through distillation. The absence of heavy metals in the final product simplifies the environmental compliance burden, as there is no need for expensive heavy metal scavenging or disposal protocols that are mandatory for palladium or platinum-catalyzed reactions. This environmental friendliness, combined with the straightforward scale-up potential of batch reactors, makes the technology highly attractive for long-term industrial adoption and regulatory approval in strict jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this tetraazido alkane, based on the specific data points and benefits outlined in the patent literature. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their existing formulation pipelines. The answers provided reflect the rigorous standards and performance metrics established by the inventors to ensure product efficacy.
Q: What are the primary advantages of this tetraazido alkane over pentaerythritol derivatives?
A: Unlike pentaerythritol-based azides which suffer from slow burning rates, this specific tetraazido alkane structure offers significantly faster combustion speeds while maintaining excellent plasticizing properties and compatibility with HMX and RDX.
Q: How does the synthesis route ensure high purity for energetic applications?
A: The process utilizes rigorous purification steps including vacuum distillation for the ester intermediate, multiple extractions and recrystallizations for the alcohol and tosylate stages, and careful phase separation for the final azide to minimize hazardous impurities.
Q: Is this synthesis route scalable for industrial propellant manufacturing?
A: Yes, the route relies on commodity chemicals like diethyl malonate and standard unit operations such as reflux, filtration, and extraction, making it highly amenable to commercial scale-up without requiring exotic catalysts or extreme pressures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Diazido-1,7-diazidoheptane Supplier
As the global demand for high-performance energetic materials continues to rise, partnering with a manufacturer that possesses deep technical expertise in complex azide chemistry is paramount for success. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of 2,6-diazido-1,7-diazidoheptane meets the exacting standards required for solid propellant and explosive applications. We understand the critical nature of supply continuity in the defense sector and have optimized our logistics to guarantee timely delivery without compromising on safety or quality.
We invite you to engage with our technical team to discuss how this advanced synthesis route can be tailored to your specific formulation needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how switching to this intermediate can optimize your overall manufacturing economics. We encourage potential partners to contact our technical procurement team directly to索取 specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain reliability.
