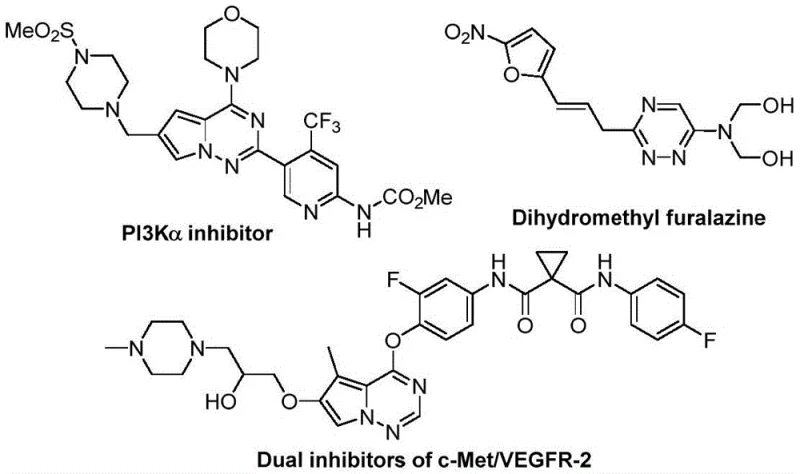
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Intermediates for Pharma
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Intermediates for Pharma
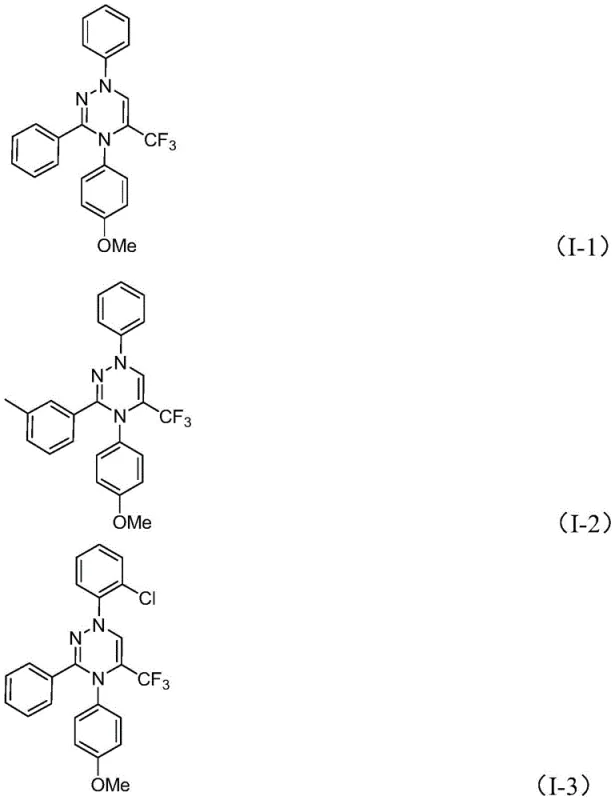
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to access nitrogen-rich heterocyclic scaffolds, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. Patent CN116253692A introduces a groundbreaking preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds, addressing critical bottlenecks in current synthetic workflows. This innovation leverages a synergistic [3+3] cycloaddition strategy between chlorohydrazones and trifluoroacetyl sulfur ylides, facilitated by inexpensive potassium carbonate. Unlike traditional routes that often rely on harsh conditions or precious metal catalysis, this protocol operates under mild, aerobic conditions, offering a streamlined pathway for producing high-value intermediates. The structural versatility of the resulting 1,2,4-triazine core is exemplified by its presence in various bioactive molecules, ranging from kinase inhibitors to antifungal agents, underscoring the immense commercial potential of this synthetic advancement for reliable pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine ring has relied heavily on the condensation reactions of amidrazones with 1,2-diketones or alkynes, as well as multicomponent reactions involving hydrazides and dicarbonyl compounds. While these classical methods have served the community for decades, they are increasingly viewed as inefficient for modern process chemistry due to several inherent drawbacks. Conventional protocols frequently necessitate the pre-synthesis of complex substrates, which adds multiple steps to the overall timeline and significantly increases material costs. Furthermore, many of these reactions suffer from poor atom economy and limited structural diversity, often failing to accommodate sensitive functional groups required in late-stage drug development. The reliance on specific activation modes or harsh thermal conditions can also lead to the formation of difficult-to-remove impurities, complicating the purification process and reducing the overall yield of the desired high-purity OLED material or API precursor.
The Novel Approach
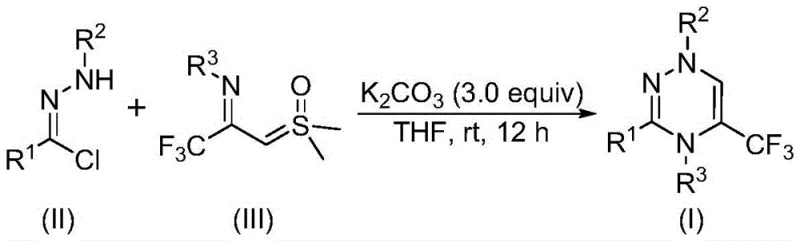
In stark contrast to legacy techniques, the methodology disclosed in CN116253692A represents a paradigm shift towards green and efficient synthesis. By utilizing chlorohydrazones and trifluoroacetyl sulfur ylides as direct building blocks, this novel approach bypasses the need for pre-functionalized diketones. The reaction is driven by potassium carbonate, a benign and cost-effective inorganic base, which promotes the formation of a reactive nitrile imine intermediate in situ. This intermediate then undergoes a rapid [3+3] cycloaddition with the sulfur ylide, followed by the elimination of dimethyl sulfoxide, to furnish the target trifluoromethyl-substituted 1,2,4-triazine. The ability to conduct this transformation at room temperature and under an air atmosphere eliminates the need for inert gas protection and energy-intensive heating, drastically simplifying the operational complexity and enhancing safety profiles for commercial scale-up of complex polymer additives or fine chemicals.
Mechanistic Insights into Potassium Carbonate-Promoted [3+3] Cycloaddition
The mechanistic elegance of this transformation lies in the generation of a 1,3-dipole species without the need for external oxidants or photochemical activation. Upon treatment with potassium carbonate, the chlorohydrazone undergoes dehydrohalogenation to generate a highly reactive nitrile imine intermediate. This species acts as a potent 1,3-dipole that engages the electron-deficient carbon-carbon double bond of the trifluoroacetyl sulfur ylide. The subsequent cycloaddition is a concerted process that constructs the six-membered triazine ring with high regioselectivity. The presence of the trifluoromethyl group on the ylide not only activates the dipolarophile but also ensures the final product possesses the desired lipophilic and electronic properties crucial for biological activity. Following the ring closure, the spontaneous extrusion of dimethyl sulfoxide drives the equilibrium forward, ensuring high conversion rates and minimizing the formation of side products that could compromise the purity of the final API intermediate.
From an impurity control perspective, this mechanism offers distinct advantages over transition-metal catalyzed alternatives. The absence of heavy metals such as palladium, copper, or rhodium means there is no risk of metal leaching into the final product, a critical parameter for regulatory compliance in pharmaceutical manufacturing. The primary byproduct, dimethyl sulfoxide, is highly polar and water-soluble, allowing for its facile removal during the aqueous workup or silica gel filtration steps described in the patent. This clean reaction profile significantly reduces the burden on downstream purification teams, who would otherwise need to employ specialized scavengers or recrystallization techniques to meet stringent residual metal specifications. Consequently, the process delivers a cleaner crude profile, facilitating the isolation of high-purity electronic chemical or pharmaceutical grades with minimal processing.
How to Synthesize Trifluoromethyl 1,2,4-Triazine Efficiently
The experimental procedure outlined in the patent provides a robust framework for laboratory and pilot-scale production. The process begins with the precise weighing of chlorohydrazone and trifluoroacetyl sulfur ylide, typically in a molar ratio favoring the ylide to drive the reaction to completion. These reagents are suspended in an aprotic organic solvent, with tetrahydrofuran (THF) identified as the optimal medium due to its ability to solubilize both organic substrates and the inorganic base effectively. The addition of potassium carbonate serves as the trigger for the reaction, initiating the cascade that leads to ring formation. Detailed standardized synthesis steps for replicating this high-yielding transformation are provided in the guide below, ensuring reproducibility across different manufacturing sites.
- Combine chlorohydrazone, trifluoroacetyl sulfur ylide, and potassium carbonate (3.0 equivalents) in an organic solvent such as THF.
- Stir the reaction mixture at room temperature (20-40°C) under an air atmosphere for 10 to 14 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target triazine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond simple yield metrics. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Chlorohydrazones and trifluoroacetyl sulfur ylides are derived from commodity chemicals such as acyl chlorides, hydrazines, and trifluoroacetic acid, all of which are available in bulk quantities from established global suppliers. This abundance ensures a stable supply line and mitigates the risk of production delays caused by the scarcity of exotic reagents. Furthermore, the elimination of noble metal catalysts removes a significant cost center associated with both the purchase of the catalyst and the subsequent validation of its removal, leading to substantial cost savings in API manufacturing without compromising quality.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound. By replacing expensive transition metal catalysts with inexpensive potassium carbonate, the direct material cost of the reaction is significantly reduced. Additionally, the simplified workup procedure, which involves basic filtration and column chromatography rather than complex metal scavenging or distillation, lowers the operational expenditure related to labor and consumables. The high atom efficiency of the [3+3] cycloaddition minimizes waste generation, further contributing to a leaner and more cost-effective production model that aligns with modern green chemistry principles.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. Since the process tolerates air and moisture and operates at ambient temperatures, it does not require specialized reactor setups such as high-pressure autoclaves or gloveboxes. This flexibility allows for production in a wider range of facilities, reducing the dependency on single-source contract manufacturers with specialized capabilities. The use of common solvents like THF also ensures that solvent availability remains consistent, preventing bottlenecks that often arise when relying on niche or hazardous reagents.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram levels is straightforward due to the exothermic nature of the reaction being manageable at room temperature. The absence of toxic heavy metals simplifies environmental compliance and waste disposal protocols, as the effluent streams do not require specialized treatment for metal contamination. This aligns perfectly with increasingly stringent global environmental regulations, allowing manufacturers to maintain their sustainability certifications while expanding production capacity to meet market demand for these valuable heterocyclic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on reaction parameters and substrate compatibility. Understanding these nuances is essential for process chemists aiming to adapt this methodology for specific target molecules.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the process described in patent CN116253692A utilizes potassium carbonate as a promoter, completely eliminating the need for costly heavy metal catalysts and simplifying downstream purification.
Q: What are the optimal reaction conditions for this cycloaddition?
A: The reaction proceeds efficiently at mild temperatures between 20°C and 40°C under an air atmosphere, typically requiring 10 to 14 hours in tetrahydrofuran (THF) solvent.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the use of inexpensive inorganic bases, ambient pressure, and readily available starting materials makes this route highly amenable to commercial scale-up without specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug discovery and development. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN116253692A and is fully equipped to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are designed to handle complex heterocyclic synthesis with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced trifluoromethylation technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data for our reference standards and to discuss route feasibility assessments that can optimize your supply chain and reduce time-to-market for your critical therapeutic candidates.
