Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Pharma
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Pharma
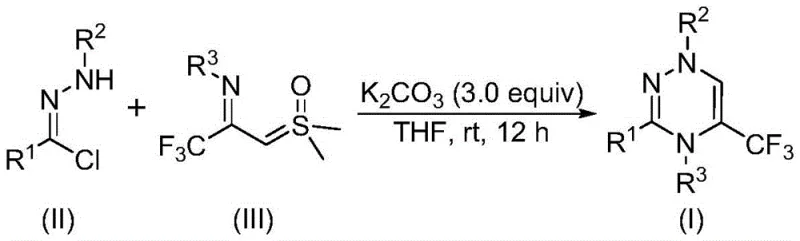
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that possess significant biological potential. A recent breakthrough detailed in patent CN116253692A introduces a highly practical preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds, a class of six-membered nitrogen-containing heterocycles known for their diverse bioactivities including anticancer, antifungal, and antihypertensive properties. The incorporation of the trifluoromethyl group is particularly strategic, as it enhances the lipophilicity, metabolic stability, and bioavailability of the parent molecule, making these intermediates invaluable for modern drug discovery programs. This novel methodology distinguishes itself by operating under exceptionally mild conditions, utilizing air as the atmosphere and room temperature as the thermal input, thereby eliminating the energy-intensive requirements often associated with heterocycle synthesis.
For R&D directors and process chemists, the significance of this patent lies in its operational simplicity and the avoidance of complex catalytic systems. Traditional syntheses of 1,2,4-triazines often rely on the condensation of amidrazones with 1,2-diketones or multicomponent reactions that can suffer from poor atom economy and limited structural diversity. In contrast, the disclosed method leverages a synergistic [3+3] cycloaddition strategy between readily accessible chlorohydrazones and trifluoroacetyl sulfur ylides. This approach not only streamlines the synthetic pathway but also ensures high purity profiles by minimizing the formation of difficult-to-remove byproducts, a critical factor for regulatory compliance in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine core has been achieved through several classical pathways, such as the cyclization of hydrazines with oxazolones or the reaction of hydrazides with dicarbonyl compounds. However, these conventional methods frequently encounter significant bottlenecks when applied to the synthesis of trifluoromethyl-substituted derivatives. Many existing protocols require the pre-synthesis of specialized substrates that are unstable or expensive to procure, leading to increased overall production costs and extended lead times. Furthermore, traditional routes often necessitate the use of strong acids or bases, high temperatures, and strictly anhydrous conditions, which can degrade sensitive functional groups and limit the scope of compatible substituents. The reliance on transition metal catalysts in some modern variations also introduces the risk of heavy metal contamination, necessitating costly and time-consuming purification steps to meet stringent pharmaceutical standards.
The Novel Approach
The innovative strategy presented in the patent data overcomes these historical challenges by employing a direct cycloaddition between chlorohydrazones and trifluoroacetyl sulfur ylides promoted by potassium carbonate. This method operates efficiently in common organic solvents like tetrahydrofuran (THF) at temperatures ranging from 20°C to 40°C, effectively removing the need for cryogenic cooling or high-energy heating. The use of potassium carbonate, an inexpensive and non-toxic inorganic salt, serves as a mild base that facilitates the generation of the reactive nitrile imine intermediate without promoting excessive decomposition. By conducting the reaction in an open air atmosphere, the process eliminates the logistical burden of maintaining inert gas lines, making it inherently safer and more adaptable to large-scale reactor environments. This streamlined workflow allows for the rapid assembly of complex trifluoromethyl-triazine architectures with high yields, as demonstrated by the successful synthesis of various derivatives.
The versatility of this synthetic route is further evidenced by its tolerance to a wide array of functional groups. As illustrated in the reaction scope, substituents such as methoxy, methyl, chloro, bromo, and even bulky naphthyl groups are well-accommodated on the aromatic rings of the starting materials. This structural flexibility is paramount for medicinal chemists who need to explore structure-activity relationships (SAR) rapidly. The ability to introduce electron-withdrawing and electron-donating groups at ortho, meta, and para positions without compromising reaction efficiency underscores the robustness of this chemistry. Consequently, this method provides a reliable platform for generating diverse libraries of trifluoromethyl-substituted 1,2,4-triazines, accelerating the hit-to-lead optimization phase in drug development projects.
Mechanistic Insights into [3+3] Cycloaddition via Nitrile Imine Intermediate
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing reaction parameters and ensuring reproducibility at scale. The reaction initiates with the deprotonation of the chlorohydrazone by potassium carbonate, which triggers the elimination of hydrogen chloride to generate a highly reactive nitrile imine intermediate in situ. This 1,3-dipole species is then poised to undergo a concerted [3+3] cycloaddition with the trifluoroacetyl sulfur ylide, which acts as the dipolarophile. The synergy between these two components drives the formation of the six-membered triazine ring, followed by the elimination of dimethyl sulfoxide (DMSO) to restore aromaticity and yield the final product. Alternatively, the mechanism may proceed through a stepwise pathway involving intermolecular nucleophilic addition followed by intramolecular nucleophilic substitution, both of which converge to the same thermodynamic product.
From an impurity control perspective, the mild basicity of potassium carbonate plays a pivotal role in maintaining a clean reaction profile. Stronger bases might induce side reactions such as hydrolysis of the imine bond or polymerization of the reactive intermediates, leading to complex impurity spectra that are difficult to separate. By carefully tuning the stoichiometry—typically using a molar ratio of chlorohydrazone to sulfur ylide to potassium carbonate of 1:2:3—the process maximizes conversion while minimizing the accumulation of unreacted starting materials or degradation products. The use of THF as the preferred solvent ensures that all reagents remain in solution, facilitating homogeneous reaction kinetics and consistent heat transfer, which are essential factors for maintaining batch-to-batch consistency in commercial manufacturing settings.
How to Synthesize Trifluoromethyl Substituted 1,2,4-Triazine Efficiently
To implement this synthesis effectively, precise control over reagent quality and reaction timing is essential. The protocol dictates dissolving the chlorohydrazone and trifluoroacetyl sulfur ylide in dry THF, followed by the addition of powdered potassium carbonate. The mixture is then stirred at ambient temperature for a duration of 10 to 14 hours, allowing sufficient time for the cycloaddition to reach completion. Monitoring the reaction progress via TLC or HPLC is recommended to determine the exact endpoint, although the patent data suggests that 12 hours is generally sufficient for high conversion. Upon completion, the solid inorganic salts are removed by filtration, and the filtrate is concentrated. The crude residue is typically purified by flash column chromatography on silica gel to afford the pure trifluoromethyl-substituted 1,2,4-triazine compound. For detailed standard operating procedures and specific stoichiometric ratios for different substrates, please refer to the guide below.
- Prepare the reaction mixture by adding potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide into an organic solvent such as tetrahydrofuran (THF).
- Stir the reaction mixture at room temperature (20-40°C) under an air atmosphere for 10 to 14 hours to allow the [3+3] cycloaddition to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target trifluoromethyl substituted 1,2,4-triazine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers compelling economic and logistical benefits. The primary driver for cost reduction is the elimination of expensive transition metal catalysts, such as palladium or copper complexes, which are not only costly to purchase but also require specialized ligands and rigorous removal processes to meet residual metal specifications. By replacing these with commodity-grade potassium carbonate, the raw material costs are drastically reduced, and the downstream purification burden is significantly lightened. Furthermore, the reaction's ability to proceed in air removes the capital expenditure associated with nitrogen generation systems and the operational cost of consuming large volumes of inert gas, directly contributing to lower overheads in the manufacturing facility.
- Cost Reduction in Manufacturing: The economic advantage of this process is multifaceted, stemming from both raw material savings and operational efficiencies. Since the starting materials—chlorohydrazones and sulfur ylides—can be synthesized from widely available acyl chlorides, hydrazines, and trifluoroacetic acid derivatives, the supply chain is less vulnerable to bottlenecks associated with exotic reagents. The absence of heavy metals means that manufacturers can avoid the substantial costs linked to metal scavenging resins and the extensive analytical testing required to certify low metal content. Additionally, the high yields reported in the patent data, reaching up to 87% for certain substrates, imply a more efficient use of raw materials, reducing waste disposal costs and improving the overall mass balance of the production process.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness and simplicity of the reaction conditions. Because the synthesis does not require strictly anhydrous solvents or oxygen-free environments, it is less susceptible to failures caused by equipment leaks or utility interruptions. The starting materials are stable and can be stockpiled without special storage requirements, ensuring that production schedules can be maintained even during periods of market volatility. The scalability of the process, which has been demonstrated from gram to multi-gram levels without loss of efficiency, assures supply chain heads that the technology can be seamlessly transferred from pilot plants to full-scale commercial reactors, guaranteeing a steady flow of high-purity intermediates to downstream customers.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this method aligns well with green chemistry principles. The use of potassium carbonate generates benign byproducts, and the avoidance of toxic heavy metals reduces the environmental footprint of the waste stream. The reaction operates at near-ambient temperatures, significantly lowering the energy consumption compared to processes requiring reflux or cryogenic cooling. This energy efficiency not only reduces utility bills but also supports corporate sustainability goals. The simplified workup procedure, involving basic filtration and chromatography, minimizes the use of hazardous quenching agents and reduces the volume of aqueous waste generated, making the process easier to permit and operate within strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of trifluoromethyl-substituted 1,2,4-triazines using this patented methodology. These answers are derived directly from the experimental data and technical disclosures found in the source patent, providing a transparent view of the technology's capabilities and limitations. Understanding these details helps stakeholders make informed decisions regarding process adoption and integration into existing manufacturing workflows.
Q: What are the key advantages of this synthesis method over traditional routes?
A: Unlike conventional methods that often require harsh conditions or expensive transition metal catalysts, this novel approach utilizes inexpensive potassium carbonate as a promoter and operates under mild conditions (room temperature, air atmosphere), significantly simplifying post-treatment and reducing heavy metal contamination risks.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process is highly scalable. It avoids the need for inert gas protection (nitrogen/argon) and uses readily available, stable starting materials. The simple workup involving filtration and column chromatography facilitates easy expansion from gram to kilogram scales.
Q: What is the structural diversity achievable with this method?
A: The method exhibits excellent substrate tolerance. Various substituents such as alkyl, alkoxy, halogens, and trifluoromethyl groups on the phenyl rings of the starting chlorohydrazones and sulfur ylides are well-tolerated, allowing for the synthesis of a wide library of diverse 1,2,4-triazine derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our team of expert process chemists has extensively evaluated the synthetic route described in CN116253692A and confirmed its viability for commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed impurity profiling and residual solvent analysis, to guarantee that every batch meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your drug development programs. Whether you require custom synthesis of specific trifluoromethyl-triazine derivatives or scale-up services for clinical trial materials, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can accelerate your timeline and reduce your overall cost of goods sold.
