Advanced Photoredox Synthesis of Beta-Nitroketones for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex structural motifs efficiently. A significant breakthrough in this domain is detailed in Chinese Patent CN114213254B, which discloses a novel preparation method for beta-nitroketones and their subsequent applications. This technology leverages visible light photoredox catalysis to couple enol silyl ethers with geminal halogenated nitro hydrocarbons, achieving yields exceeding 95% under remarkably mild conditions. For R&D directors and procurement specialists, this represents a paradigm shift from traditional thermal methods to sustainable, light-driven synthesis. The ability to generate beta-nitroketones, which serve as critical precursors to beta-amino ketones and 1,3-amino alcohols, opens new avenues for synthesizing antidepressants and other bioactive molecules without the limitations of classical chemistry.
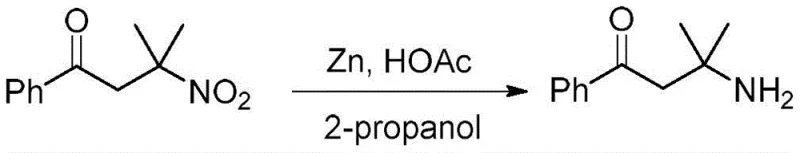
Beta-amino ketones and 1,3-amino alcohols are ubiquitous structural units found in numerous natural products and medicinal compounds. Historically, accessing these motifs has relied heavily on the Mannich reaction, which typically yields ketones with N-modified amino groups at the beta-secondary or beta-tertiary carbon positions. A major limitation of this conventional approach is the difficulty in removing substituents from the nitrogen atom to introduce the desired primary amine functionality. This deprotection often requires harsh reaction conditions that can compromise the integrity of the molecule. In contrast, the technology described in CN114213254B utilizes beta-nitroketones as stable equivalents. The nitro group can be selectively reduced to an unmodified amino group under mild conditions, bypassing the need for difficult deprotection steps and providing direct access to primary beta-amino ketones.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for beta-nitroketones have been plagued by significant inefficiencies and safety concerns. One common method involves the nucleophilic substitution of brominated or iodinated hydrocarbons with silver nitrite or sodium nitrite. However, the ambident nature of the nitrite ion often leads to the competitive formation of nitrite ester byproducts, drastically reducing the yield of the desired nitro compound and complicating purification. Another approach, the Michael addition of nitrous acid to alpha,beta-unsaturated ketones, suffers from a very narrow substrate scope, limited essentially to easily prepared alpha-methylene ketones. Furthermore, existing methods often require harsh acidic or basic conditions, high temperatures, or expensive stoichiometric reagents, making them unsuitable for the cost-sensitive and safety-critical environment of modern pharmaceutical manufacturing.
The Novel Approach
The innovative method disclosed in the patent overcomes these hurdles by employing a visible light-mediated photoredox catalytic system. By utilizing enol silyl ethers and geminal halogenated nitro hydrocarbons as starting materials, the reaction proceeds smoothly at room temperature under neutral conditions. This eliminates the need for external heating or cooling and avoids the use of corrosive acids or bases that could induce side reactions, such as the elimination of nitrous acid from the beta-nitroketone product. The use of cheap and readily available raw materials, combined with the high efficiency of the photoredox catalyst, ensures that the process is not only chemically superior but also economically viable for industrial applications.
Mechanistic Insights into Visible Light Photoredox Coupling
The core of this technology lies in the generation of radical intermediates through single-electron transfer (SET) processes facilitated by the photoredox catalyst. Upon irradiation with visible light (wavelength > 400 nm), typically from blue LED sources, the catalyst enters an excited state capable of oxidizing or reducing the substrates. In this specific transformation, the interaction between the excited catalyst and the geminal halogenated nitro hydrocarbon likely generates a carbon-centered radical adjacent to the nitro group. This radical species then couples with the enol silyl ether, followed by desilylation and oxidation to furnish the beta-nitroketone product. The mildness of this radical pathway is crucial, as it preserves sensitive functional groups that would otherwise decompose under thermal radical initiation conditions.
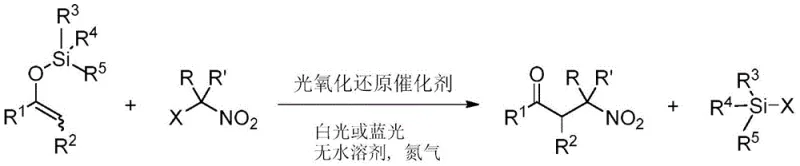
Impurity control is a critical aspect of this mechanism. In traditional thermal syntheses, beta-nitroketones are prone to eliminating nitrous acid under heating or in the presence of acid/base additives, leading to the formation of alpha,beta-unsaturated ketones as major impurities. The photoredox method operates at ambient temperature (around 25°C) and maintains a neutral pH throughout the reaction course. This effectively suppresses the elimination pathway, ensuring high chemical purity of the final product. Additionally, the reaction demonstrates excellent tolerance for various substituents on both the enol silyl ether and the nitroalkane, allowing for the synthesis of diverse structures including those with aromatic, heteroaromatic, and cyclic moieties without significant loss in yield.
The versatility of the resulting beta-nitroketones is further demonstrated by their downstream transformations. As shown in the patent data, the nitro group can be selectively reduced to an amino group using zinc powder or iron powder to yield beta-amino ketones. Alternatively, catalytic hydrogenation using Pd/C can simultaneously reduce both the nitro and carbonyl groups to produce 1,3-amino alcohols. This flexibility makes the initial beta-nitroketone intermediate a powerful building block for generating a wide library of pharmacologically active compounds.
How to Synthesize Beta-Nitroketones Efficiently
The synthesis protocol outlined in the patent is designed for operational simplicity and reproducibility, making it ideal for both laboratory scale-up and industrial implementation. The process begins with the preparation of the enol silyl ether from the corresponding ketone and trimethylchlorosilane (TMSCl), a straightforward transformation that does not require rigorous purification before use. The subsequent coupling reaction is performed in an anhydrous solvent, preferably acetonitrile, under an inert atmosphere to prevent oxygen quenching of the radical intermediates. Detailed standardized synthesis steps are provided in the guide below to ensure consistent results.
- Prepare the reaction mixture by combining enol silyl ether and geminal halogenated nitro hydrocarbon in an anhydrous solvent such as acetonitrile under an inert nitrogen atmosphere.
- Add a photoredox catalyst, such as an Iridium or Ruthenium complex, at a low molar loading ranging from 0.1% to 1.0% relative to the substrate.
- Irradiate the mixture with visible light (blue LED, 420-460nm) at room temperature for approximately 2 hours, then purify the crude product via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photoredox technology offers substantial strategic benefits beyond mere chemical efficiency. The shift from harsh thermal conditions to ambient light-driven synthesis fundamentally alters the cost structure and risk profile of manufacturing these critical intermediates. By eliminating the need for high-energy heating systems and specialized corrosion-resistant reactors required for strong acids or bases, the capital expenditure for production facilities can be significantly optimized. Furthermore, the simplicity of the workup procedure, which often involves basic concentration and silica gel chromatography, reduces the consumption of solvents and processing time, directly contributing to cost reduction in pharmaceutical intermediates manufacturing.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive and commercially available starting materials. Enol silyl ethers can be generated in situ or purchased at low cost, and the geminal halogenated nitro hydrocarbons are stable and easy to handle. Crucially, the photoredox catalysts, such as Iridium or Ruthenium complexes, are used at extremely low loadings (0.1% to 1.0% molar ratio). This minimal catalyst requirement mitigates the cost impact of precious metals and simplifies the removal of metal residues from the final product, avoiding the need for expensive scavenging resins or complex purification trains often associated with transition metal catalysis.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the reaction conditions. Since the reaction proceeds at room temperature and is not sensitive to minor fluctuations in thermal control, the risk of batch failure due to equipment malfunction is minimized. The raw materials are structurally diverse yet chemically stable, ensuring a reliable supply base. Moreover, the compatibility of this chemistry with continuous flow reactors means that production can be scaled linearly by increasing run time rather than vessel size, offering a flexible response to market demand fluctuations and reducing lead time for high-purity beta-nitroketones.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method aligns perfectly with green chemistry principles. The absence of toxic heavy metal reagents (used stoichiometrically) and the avoidance of hazardous acidic or basic waste streams simplify wastewater treatment and disposal. The reaction's suitability for flow chemistry using LED light sources further enhances energy efficiency compared to traditional UV lamps or thermal heaters. This ease of scale-up from gram to multi-ton quantities ensures that the commercial scale-up of complex fine chemical intermediates can be achieved with minimal regulatory hurdles and a reduced environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photoredox synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this method for your specific production needs.
Q: What are the advantages of this photoredox method over traditional Mannich reactions?
A: Unlike traditional Mannich reactions which often produce N-modified amines requiring harsh deprotection steps, this photoredox method directly yields beta-nitroketones. The nitro group serves as a versatile equivalent to the amino group, easily reducible to unmodified primary amines under mild conditions, thereby simplifying the synthetic route and avoiding harsh acidic or basic treatments that can degrade sensitive substrates.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly amenable to scale-up. It operates at room temperature and neutral pH, eliminating the need for expensive heating or cooling infrastructure and reducing corrosion risks. Furthermore, the compatibility with continuous flow chemistry using LED arrays and glass coils allows for safe, efficient, and consistent manufacturing of complex pharmaceutical intermediates on a multi-ton scale.
Q: What types of substrates are compatible with this synthesis method?
A: The method exhibits exceptional substrate scope, tolerating a wide variety of functional groups. Enol silyl ethers derived from aromatic, heteroaromatic, aliphatic, and cyclic ketones all react efficiently. Additionally, the geminal halogenated nitro hydrocarbons can vary from simple alkyl chains to complex cyclic structures, enabling the synthesis of diverse beta-nitroketone scaffolds essential for drug discovery and development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Nitroketone Supplier
The technological potential of visible-light mediated synthesis for beta-nitroketones is immense, offering a cleaner and more efficient route to valuable pharmaceutical building blocks. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team is equipped to translate this patented laboratory methodology into robust industrial processes, ensuring stringent purity specifications and rigorous QC labs are utilized to guarantee the quality of every batch. We understand the critical nature of these intermediates in the drug development pipeline and are committed to delivering consistency and reliability.
We invite you to collaborate with us to leverage this advanced synthetic technology for your projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in photoredox chemistry can enhance your supply chain efficiency and product quality.
