Advanced Photoredox Synthesis of Beta-Nitroketones for Scalable Pharmaceutical Manufacturing
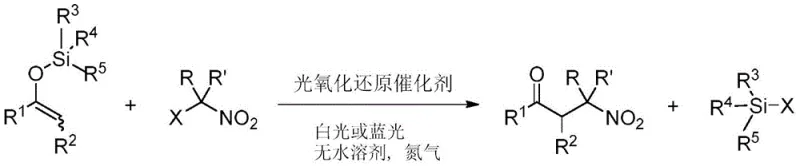
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access critical structural motifs like beta-aminoketones and 1,3-aminoalcohols, which are ubiquitous in bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN114213254B, which discloses a novel preparation method for beta-nitroketones using visible light photoredox catalysis. This technology represents a paradigm shift from traditional thermal methods, offering a robust route to synthesize these valuable intermediates under neutral, room-temperature conditions. By leveraging enol silyl ethers and geminal halogenated nitro hydrocarbons, this process achieves exceptional yields exceeding 95% in many cases, providing a reliable foundation for the subsequent production of beta-aminoketones. For R&D directors and procurement specialists, understanding this patented methodology is crucial for optimizing supply chains and reducing the environmental footprint of complex organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-aminoketones has relied heavily on the Mannich reaction, which typically yields ketones with N-modified amino groups at the beta-secondary or beta-tertiary carbon positions. A major drawback of this conventional approach is the difficulty in accessing beta-nitroketones containing primary amine or alpha-tertiary alkylamine structures directly. Furthermore, removing substituents from the nitrogen atom in Mannich products often requires harsh reaction conditions that are significantly more difficult than introducing substituents to an unmodified amine. Alternative prior art methods, such as the nucleophilic substitution of brominated hydrocarbons with silver nitrite, suffer from competitive formation of nitrite ester byproducts due to the ambident nature of the nitrite ion, leading to low yields and arduous purification processes. Similarly, Michael addition reactions of alpha,beta-unsaturated ketones with nitrous acid are restricted to a very narrow range of easily prepared alpha-methylene ketones, limiting their utility in diverse drug discovery programs.
The Novel Approach
In stark contrast to these legacy techniques, the method described in patent CN114213254B utilizes a visible light-driven photoredox catalytic system to couple enol silyl ethers with geminal halogenated nitro hydrocarbons. This innovative approach operates at room temperature under neutral conditions, effectively preventing the elimination of nitrous acid that often plagues acid or base-induced reactions. The use of inexpensive and readily available raw materials, combined with the ability to achieve yields up to 100%, marks a substantial improvement in process efficiency. Moreover, the resulting beta-nitroketones feature a nitro group located on a beta-quaternary carbon, which serves as a versatile handle for further functionalization. This method not only simplifies the synthetic route but also enhances the structural diversity accessible to medicinal chemists, allowing for the rapid generation of libraries containing unmodified beta-aminoketones.
Mechanistic Insights into Visible Light Photoredox Coupling
The core of this technological advancement lies in the activation of geminal halogenated nitro hydrocarbons via single-electron transfer (SET) mediated by a photoredox catalyst. Upon irradiation with visible light, typically in the blue spectrum range of 420-460nm, catalysts such as fac-Ir(ppy)3 or Ru(bpy)3Cl2 enter an excited state capable of reducing the carbon-halogen bond. This generates a reactive nitroalkyl radical species which subsequently couples with the electron-rich enol silyl ether. The neutrality of the reaction medium is a critical mechanistic feature; it ensures that the sensitive beta-nitroketone product does not undergo retro-Mannich type elimination or decomposition, which are common side reactions in acidic or basic environments. This mechanistic elegance allows for the tolerance of a wide array of functional groups, including halogens, esters, and heterocycles, without the need for protecting groups.
From an impurity control perspective, the absence of strong acids or bases significantly reduces the formation of degradation products. Traditional methods often struggle with the stability of the nitro group under thermal stress or pH extremes, leading to complex impurity profiles that are difficult to separate. In this photoredox system, the mild conditions preserve the integrity of the nitro functionality, resulting in a cleaner crude reaction mixture. The patent data indicates that simple purification via silica gel column chromatography is sufficient to isolate the target compounds with high purity. This streamlined workup process is particularly advantageous for scale-up, as it minimizes solvent consumption and waste generation, aligning with green chemistry principles that are increasingly demanded by regulatory bodies and corporate sustainability goals.
How to Synthesize Beta-Nitroketones Efficiently
The practical implementation of this synthesis involves dissolving the enol silyl ether and the geminal halogenated nitro hydrocarbon in an anhydrous solvent such as acetonitrile, followed by the addition of a catalytic amount of the photoredox complex. The reaction vessel is degassed to remove oxygen, which can quench the radical intermediates, and then irradiated with LEDs while maintaining a temperature of approximately 25°C. Monitoring via TLC typically shows completion within 2 hours. The detailed standardized synthesis steps for replicating this high-yielding transformation are outlined in the guide below.
- Prepare the reaction mixture by combining enol silyl ether and geminal halogenated nitro hydrocarbon in an anhydrous solvent such as acetonitrile under a nitrogen atmosphere.
- Add a photoredox catalyst, such as fac-Ir(ppy)3 or Ru(bpy)3Cl2, to the mixture at a molar ratio ranging from 0.1% to 1.0%.
- Irradiate the reaction with visible light (420-460nm blue LED) at room temperature for approximately 2 hours, then purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photoredox technology offers tangible strategic benefits beyond mere chemical novelty. The shift from expensive silver salts or specialized alpha,beta-unsaturated precursors to commodity enol silyl ethers and simple nitroalkanes drives significant cost reduction in pharmaceutical intermediate manufacturing. The elimination of precious metal scavengers and the reduction in energy consumption due to room temperature operation further contribute to a lower cost of goods sold (COGS). Additionally, the robustness of the reaction conditions means that raw material specifications can be slightly more relaxed without compromising yield, providing greater flexibility in sourcing and inventory management.
- Cost Reduction in Manufacturing: The replacement of stoichiometric silver nitrite with catalytic amounts of earth-abundant or recyclable photoredox catalysts drastically lowers reagent costs. Furthermore, the ability to recover solvents like acetonitrile via atmospheric distillation, as opposed to high-boiling solvents like DMSO or DMF, reduces waste disposal fees and solvent replenishment expenses. The high atom economy and minimal byproduct formation mean that less material is wasted during purification, directly enhancing the overall process efficiency and profitability.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable starting materials such as enol silyl ethers and gem-halo nitro compounds mitigates the risk of supply disruptions associated with unstable or highly regulated reagents. The mild reaction conditions reduce the wear and tear on reactor equipment, extending asset life and minimizing unplanned maintenance downtime. This reliability ensures consistent delivery schedules for downstream customers, which is critical for maintaining just-in-time manufacturing workflows in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: The patent explicitly highlights the suitability of this method for continuous flow chemistry, utilizing glass coils and LED arrays instead of traditional batch kettles. This adaptability facilitates seamless scale-up from gram to ton quantities without the heat transfer limitations typical of exothermic batch reactions. Moreover, the neutral aqueous workup and reduced solvent burden simplify wastewater treatment, ensuring compliance with increasingly stringent environmental regulations regarding volatile organic compounds (VOCs) and heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this photoredox synthesis platform. These answers are derived directly from the experimental data and embodiments provided in the patent documentation to ensure accuracy and relevance for technical stakeholders.
Q: What are the advantages of this photoredox method over traditional Mannich reactions?
A: Unlike traditional Mannich reactions which often produce N-modified amino groups requiring harsh deprotection, this method produces beta-nitroketones that can be easily reduced to unmodified primary amines under mild conditions, avoiding complex protection-deprotection sequences.
Q: Can this synthesis be scaled for industrial production?
A: Yes, the patent explicitly mentions compatibility with continuous flow reactors using glass coils and LED arrays, eliminating the need for traditional batch kettle reactors and facilitating safe, large-scale manufacturing.
Q: What types of substituents are tolerated in this reaction?
A: The method exhibits broad substrate scope, tolerating various groups including alkyl, aryl, heteroaryl, and halogen substituents on both the enol silyl ether and the nitro hydrocarbon components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Nitroketone Supplier
As the demand for complex pharmaceutical intermediates grows, partnering with a manufacturer who understands both the chemistry and the commercial implications is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our rigorous QC labs and adherence to stringent purity specifications guarantee that every batch of beta-nitroketone meets the highest industry standards, ready for immediate conversion into valuable beta-aminoketones or amino alcohols.
We invite you to contact our technical procurement team to discuss how this advanced photoredox technology can be integrated into your supply chain. Request a Customized Cost-Saving Analysis today to evaluate the potential economic impact of switching to this greener, more efficient synthesis route. We are prepared to provide specific COA data and route feasibility assessments to support your R&D and production planning efforts, ensuring a smooth transition to this next-generation manufacturing process.
