Advanced Palladium-Catalyzed Carbonylation for Scalable Dihydroquinolone Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Carbonylation for Scalable Dihydroquinolone Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic methodologies to access complex heterocyclic scaffolds that serve as the core structures for potent bioactive molecules. Patent CN112239456B discloses a significant advancement in this domain by presenting a novel preparation method for substituted 2,3-dihydroquinolone compounds. These nitrogen-containing carbonyl heterocycles are not merely academic curiosities; they form the critical backbone of numerous therapeutic agents, including those with documented antitumor and analgesic properties. As illustrated in the structural diversity of known bioactive agents, the ability to efficiently functionalize this scaffold is paramount for drug discovery pipelines. The disclosed technology leverages a transition metal palladium-catalyzed carbonylation reaction, utilizing N-pyridine sulfonyl-o-iodoaniline and olefins as primary building blocks to construct the quinolone core with high precision and efficiency.
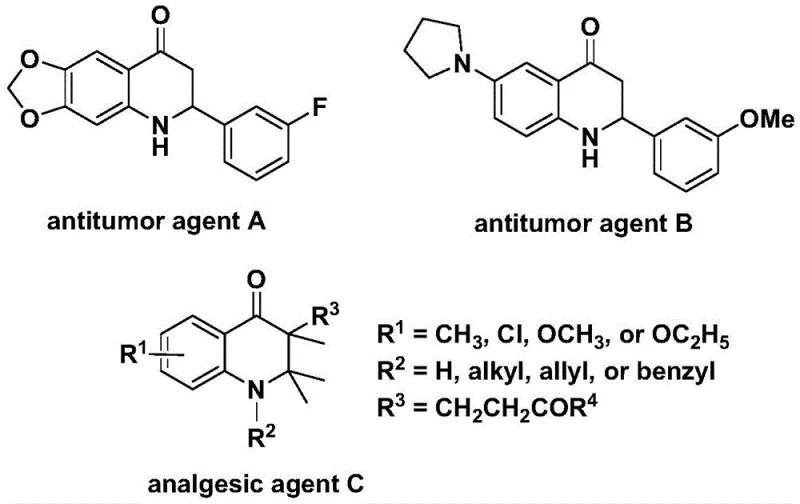
This innovative approach addresses the long-standing challenges associated with synthesizing these valuable intermediates. By employing a solid carbon monoxide surrogate instead of toxic gaseous CO, the process significantly enhances operational safety and ease of handling, which are critical factors for both laboratory research and commercial manufacturing environments. The method boasts simple operational steps, broad substrate compatibility, and high reaction yields, positioning it as a superior alternative for the reliable pharmaceutical intermediate supplier seeking to optimize their production capabilities. Furthermore, the versatility of the reaction allows for the design and synthesis of both 2-aryl and 3-alkyl substituted derivatives, thereby widening the applicability of this methodology across various medicinal chemistry programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2,3-dihydroquinolone skeleton has relied on various synthetic strategies, yet many suffer from significant drawbacks that hinder their widespread adoption in cost reduction in API manufacturing. Traditional carbonylation reactions often necessitate the use of carbon monoxide gas, which presents severe safety hazards due to its toxicity and the requirement for high-pressure equipment, thereby increasing capital expenditure and operational complexity. Moreover, existing methods frequently exhibit limited substrate scope, struggling to accommodate diverse functional groups without compromising yield or selectivity. The reliance on harsh reaction conditions or expensive, air-sensitive catalysts further exacerbates the economic burden, making these conventional routes less attractive for large-scale production. Additionally, the purification of products from complex reaction mixtures generated by older methods can be tedious, leading to lower overall throughput and increased waste generation, which conflicts with modern green chemistry principles.
The Novel Approach
In stark contrast, the novel methodology described in the patent introduces a streamlined and safer pathway that overcomes these traditional bottlenecks through the strategic use of a solid CO surrogate, specifically 1,3,5-trimesic acid phenol ester. This substitution eliminates the need for specialized high-pressure gas handling infrastructure, allowing the reaction to proceed under standard atmospheric pressure conditions while maintaining high efficiency. The process utilizes a palladium catalyst system comprising bis(acetylacetone)palladium and a dppp ligand, which facilitates the smooth insertion of the carbonyl group and subsequent cyclization. As depicted in the general reaction scheme, this approach enables the direct coupling of readily available N-pyridine sulfonyl-o-iodoaniline with a wide array of olefins. The result is a versatile synthetic route that not only simplifies the operational workflow but also ensures high conversion rates and excellent yields, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
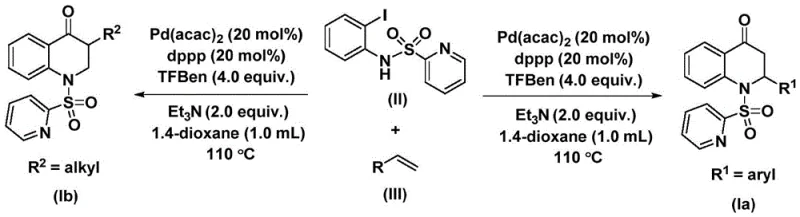
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The success of this synthetic strategy lies in the intricate dance of organometallic steps orchestrated by the palladium catalyst. The mechanism initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This step is crucial as it activates the aromatic ring for subsequent transformations. Following this activation, the carbon monoxide molecule, released in situ from the thermal decomposition of the 1,3,5-trimesic acid phenol ester, inserts into the palladium-carbon bond. This insertion forms an acyl-palladium intermediate, effectively installing the carbonyl functionality required for the quinolone structure. The precision of this CO insertion step is vital for ensuring the correct regiochemistry of the final product, avoiding the formation of unwanted isomers that could complicate downstream purification processes.
Subsequently, the olefin substrate coordinates to the palladium center and undergoes migratory insertion into the acyl-palladium bond, creating a new carbon-carbon bond and extending the molecular framework. This alkene insertion step is highly dependent on the electronic and steric properties of the olefin, yet the catalyst system demonstrates remarkable tolerance, accommodating both electron-rich and electron-deficient aryl olefins as well as aliphatic variants. The cycle concludes with a reductive elimination step, which releases the substituted 2,3-dihydroquinolone product and regenerates the active palladium(0) catalyst to enter another turnover cycle. This efficient catalytic cycle minimizes the amount of precious metal required, contributing to the overall economic viability of the process. Furthermore, the presence of the pyridine sulfonyl group acts as an internal directing group or stabilizer, facilitating the intramolecular cyclization that closes the six-membered ring, ensuring high structural fidelity of the target heterocycle.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
Implementing this synthesis requires careful attention to reagent ratios and reaction conditions to maximize yield and purity. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO surrogate, and substrates in an aprotic organic solvent such as dioxane. The detailed standardized synthesis steps see the guide below.
- Combine palladium bis(acetylacetonate), dppp ligand, triethylamine, 1,3,5-trimesic acid phenol ester, N-pyridine sulfonyl-o-iodoaniline, and olefin in an organic solvent such as dioxane.
- Heat the reaction mixture to 110 °C and stir for 24 to 48 hours to facilitate the carbonylation and cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 2,3-dihydroquinolone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible benefits that extend beyond mere chemical elegance. The shift from gaseous CO to a solid surrogate fundamentally alters the risk profile of the manufacturing process, reducing the need for specialized safety protocols and expensive containment systems. This simplification translates directly into lower operational overheads and a more resilient supply chain, as the reliance on hazardous gas cylinders is eliminated. Moreover, the use of commercially available starting materials, such as simple olefins and iodoanilines, ensures a stable and continuous supply of raw materials, mitigating the risk of production delays caused by sourcing bottlenecks. The robustness of the reaction conditions also implies a higher success rate in batch production, reducing the incidence of failed batches and the associated costs of reprocessing or disposal.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and hazardous gas handling infrastructure leads to significant capital expenditure savings. Additionally, the high catalytic efficiency and use of a solid CO surrogate reduce material waste and simplify the reaction setup, resulting in substantial cost savings in pharmaceutical intermediate manufacturing. The straightforward workup procedure, involving simple filtration and chromatography, further lowers labor and processing costs compared to complex multi-step syntheses.
- Enhanced Supply Chain Reliability: By utilizing widely available and stable reagents like 1,3,5-trimesic acid phenol ester and common olefins, the process reduces dependency on specialized or hard-to-source chemicals. This accessibility ensures a more reliable pharmaceutical intermediate supplier capability, minimizing lead times and enhancing the ability to respond quickly to market demands. The stability of the reagents also allows for longer storage periods without degradation, providing greater flexibility in inventory management.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic gas emissions make this process inherently safer and more environmentally friendly, aligning with strict regulatory standards for industrial chemical production. The scalability is evidenced by the successful gram-level synthesis reported, suggesting a clear path to kilogram and ton-scale production without fundamental changes to the process chemistry. This ease of scale-up supports the commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality and supply for downstream drug development.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this palladium-catalyzed carbonylation method. Understanding these details is essential for R&D teams planning to integrate this technology into their existing workflows.
Q: What is the advantage of using 1,3,5-trimesic acid phenol ester in this synthesis?
A: Using 1,3,5-trimesic acid phenol ester serves as a safe and convenient solid carbon monoxide surrogate, eliminating the need for handling hazardous high-pressure CO gas while maintaining high reaction efficiency.
Q: What types of substituents are tolerated on the olefin substrate?
A: The process demonstrates excellent substrate compatibility, tolerating various aryl groups with methyl, tert-butyl, methoxy, fluorine, or chlorine substituents, as well as linear or branched alkyl groups and silyl groups.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method utilizes commercially available starting materials and simple post-treatment procedures like filtration and column chromatography, making it highly practical for scaling up to industrial levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the acceleration of drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into full-scale manufacturing. We are committed to delivering high-purity 2,3-dihydroquinolone derivatives that meet stringent purity specifications, supported by our rigorous QC labs which employ advanced analytical techniques to verify identity and purity. Our expertise in palladium-catalyzed reactions allows us to optimize this specific carbonylation route for maximum efficiency and cost-effectiveness, providing our partners with a competitive edge in the marketplace.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your supply chain goals and drive innovation in your pharmaceutical pipeline.
