Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Scalable Pharmaceutical Manufacturing
Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Scalable Pharmaceutical Manufacturing
The integration of trifluoromethyl groups into heterocyclic scaffolds represents a cornerstone strategy in modern medicinal chemistry, driven by the profound impact of fluorine on the pharmacokinetic profiles of drug candidates. Patent CN110467579B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in the synthesis of these high-value pharmaceutical intermediates. The 1,2,4-triazole motif is ubiquitous in bioactive molecules, serving as a key structural element in drugs ranging from antifungal agents to kinase inhibitors, as illustrated by the diverse biological applications shown below. This patent introduces a novel, iodine-promoted cyclization strategy that bypasses the limitations of traditional trifluoromethylation techniques, offering a robust pathway for the commercial scale-up of complex heterocyclic intermediates.
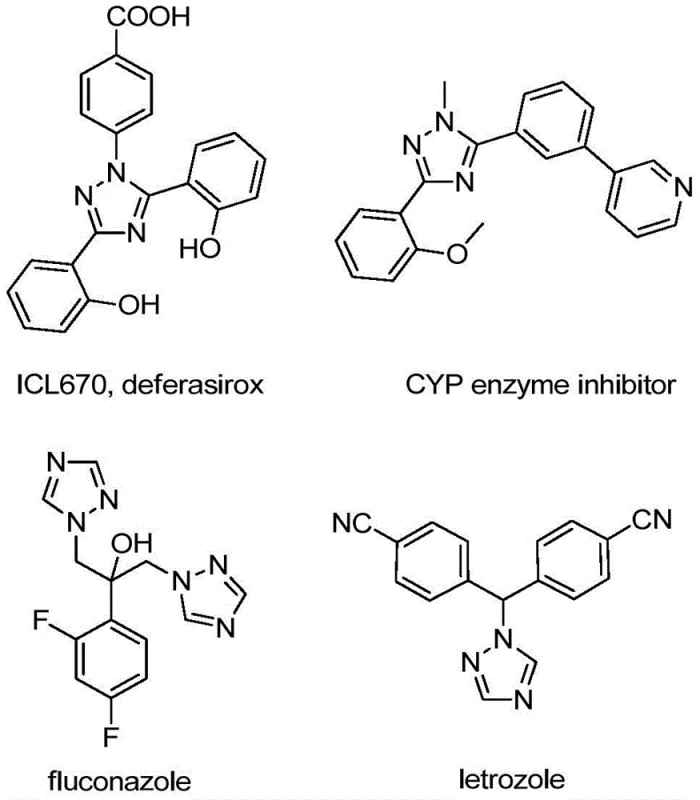
The significance of this technology extends beyond mere academic interest; it provides a tangible solution for cost reduction in API manufacturing by utilizing readily available starting materials and eliminating the need for stringent reaction conditions. For R&D directors and procurement specialists alike, the ability to access diverse 5-trifluoromethyl-1,2,4-triazole derivatives through a unified, efficient protocol is a game-changer. The method described in CN110467579B leverages a unique combination of trifluoroethylimidoyl chloride and hydrazones, facilitated by elemental iodine, to construct the triazole ring with high atom economy and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a trifluoromethyl group into a nitrogen-containing heterocycle has been fraught with synthetic challenges that hinder efficient production. Conventional literature reports primarily describe two approaches: the direct trifluoromethylation of pre-synthesized heterocycles or the cycloaddition of trifluoromethyl-bearing synthons. The former often relies on specialized and expensive trifluoromethylating reagents that can be hazardous to handle on a large scale, while the latter frequently employs unstable precursors like trifluorodiazoethane. These traditional pathways often necessitate rigorous anhydrous and anaerobic conditions, requiring specialized equipment and inert gas manifolds that drive up capital expenditure and operational complexity. Furthermore, the use of transition metal catalysts in some of these protocols introduces the risk of heavy metal contamination, necessitating costly and time-consuming purification steps to meet the stringent purity specifications required for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN110467579B offers a streamlined alternative that fundamentally reshapes the synthetic landscape for these compounds. By employing trifluoroethylimidoyl chloride and hydrazones as the primary building blocks, the process utilizes stable, commercially available feedstocks that are significantly cheaper and safer to transport and store. The reaction proceeds efficiently in common organic solvents like dichloroethane at moderate temperatures, completely obviating the need for moisture-free or oxygen-free environments. As demonstrated by the specific examples in the patent, this approach exhibits remarkable substrate tolerance, accommodating a wide array of substituents on both the aryl and heteroaryl moieties. The versatility of this method allows for the rapid generation of diverse chemical libraries, facilitating the discovery of new drug candidates while ensuring a reliable supply chain for established APIs.
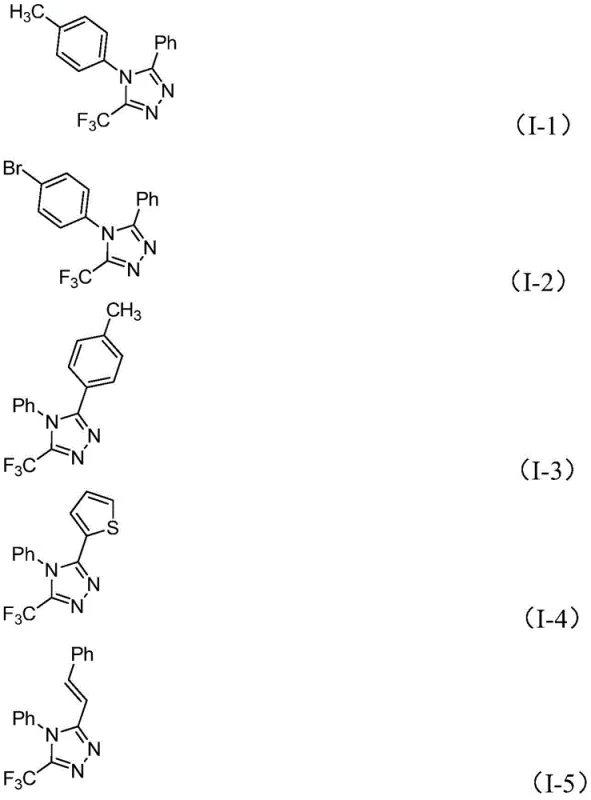
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic elegance of this transformation lies in the dual role of the reagents and the promoter. The reaction initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and the hydrazone, generating a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization, setting the stage for the critical cyclization event. The addition of elemental iodine acts as a mild oxidant and promoter, facilitating an oxidative iodination that generates a reactive iodine-containing intermediate. This intermediate then undergoes an intramolecular electrophilic substitution followed by aromatization to yield the final 5-trifluoromethyl-1,2,4-triazole core. The general reaction scheme below highlights the simplicity of the stoichiometry and the clean conversion of starting materials to the desired heterocycle.
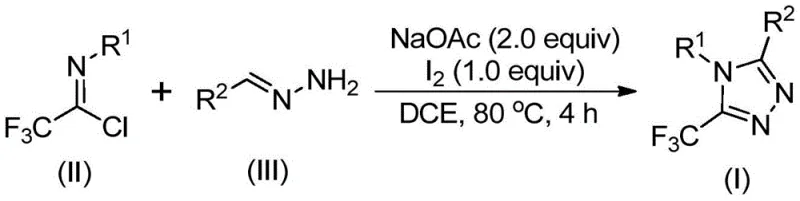
From an impurity control perspective, this mechanism is particularly advantageous for industrial applications. The absence of transition metals eliminates a major class of difficult-to-remove impurities, thereby simplifying the downstream purification process. The mild reaction conditions minimize the formation of decomposition byproducts often associated with harsh acidic or basic environments. Furthermore, the use of sodium acetate as a base provides a buffered environment that prevents the degradation of sensitive functional groups on the substrate, ensuring high fidelity in the final product structure. This level of control is essential for maintaining the integrity of complex molecular architectures found in modern therapeutics.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of execution, making it accessible for both laboratory-scale optimization and pilot-plant operations. The protocol involves a straightforward one-pot procedure where reagents are mixed and heated, followed by a simple addition of iodine to drive the reaction to completion. Detailed standardized operating procedures regarding specific molar ratios, solvent volumes, and workup techniques are critical for maximizing yield and purity. For a comprehensive guide on executing this synthesis with precision, please refer to the technical steps outlined below.
- Mix sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane.
- Heat the reaction mixture to 80°C and maintain for 2 to 4 hours to allow initial condensation.
- Add elemental iodine to the system and continue heating for an additional 1 to 2 hours to promote cyclization.
- Perform post-treatment including filtration and silica gel chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The shift away from exotic reagents and complex reaction conditions translates into a more resilient and cost-effective supply chain. By leveraging commodity chemicals as starting materials, manufacturers can mitigate the risks associated with the volatility of specialized reagent markets. The operational simplicity of the process also reduces the burden on manufacturing facilities, allowing for higher throughput and better utilization of existing infrastructure.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of inexpensive elemental iodine significantly lower the raw material costs per kilogram of product. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary materials and labor hours. This cumulative effect results in substantial cost savings that can be passed down the supply chain, enhancing the competitiveness of the final API.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable starting materials such as hydrazones and imidoyl chlorides ensures a consistent supply of inputs, reducing the risk of production delays caused by reagent degradation or scarcity. The robustness of the reaction conditions means that production is less susceptible to variations in environmental factors, leading to more predictable batch cycles and improved on-time delivery performance for customers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from gram scales upwards, which facilitates a smooth transition from R&D to commercial production. Furthermore, the avoidance of toxic heavy metals aligns with increasingly stringent environmental regulations and green chemistry principles, reducing the costs and complexities associated with waste disposal and environmental compliance auditing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential partners.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method utilizes elemental iodine as a promoter instead of toxic or expensive heavy metal catalysts, significantly simplifying purification and reducing environmental impact.
Q: What are the advantages of using trifluoroethylimidoyl chloride over other synthons?
A: Trifluoroethylimidoyl chloride is more stable and easier to handle compared to alternatives like trifluorodiazoethane, and it allows for direct construction of the heterocyclic core under mild conditions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates under standard atmospheric conditions without strict anhydrous or anaerobic requirements, making it highly amenable to commercial scale-up and continuous manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies described in CN110467579B for the production of high-value pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the bench to the plant. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,4-triazole delivered meets the highest industry standards for quality and consistency.
We invite you to collaborate with us to leverage this advanced chemistry for your next drug development program. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your path to market.
