Advanced FeCl3-Catalyzed Synthesis of 5-Trifluoromethyl-1,2,4-Triazole Derivatives for Commercial Scale-Up
The pharmaceutical and agrochemical industries continuously seek robust synthetic methodologies for constructing nitrogen-containing heterocycles, particularly 1,2,4-triazole derivatives, due to their prevalence in bioactive molecules. Patent CN111978265B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole derivatives that addresses critical limitations in existing literature. This technology leverages a dual-promoter system involving sodium bicarbonate and ferric chloride to facilitate a tandem cyclization reaction under remarkably mild conditions. The significance of this innovation is underscored by the structural presence of the 1,2,4-triazole core in major commercial drugs such as Maraviroc, Triazolam, Sitagliptin, and Deferasirox, as illustrated in the following diagram.
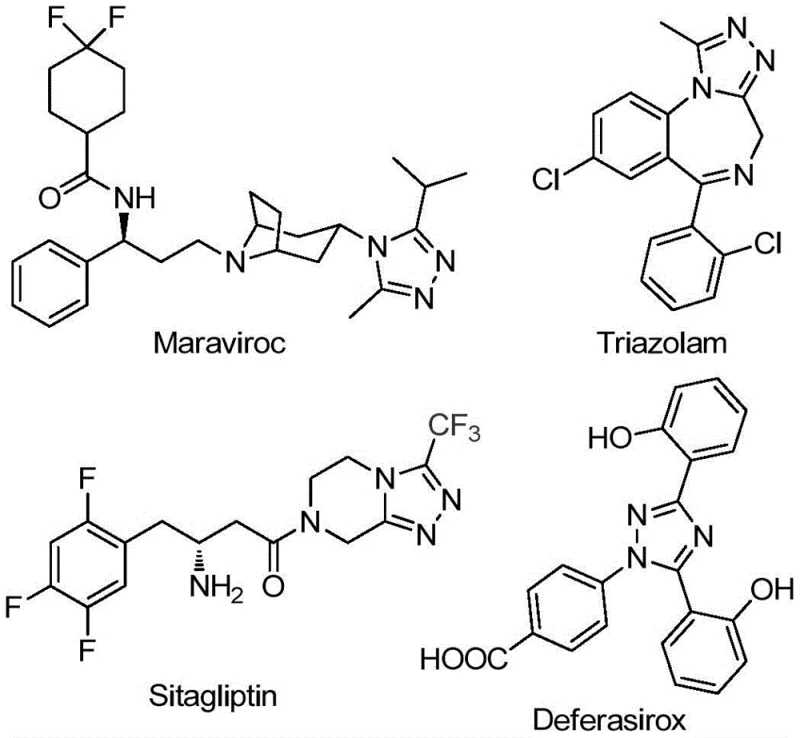
Furthermore, the introduction of the trifluoromethyl group into these heterocyclic frameworks significantly enhances electronegativity, metabolic stability, and lipophilicity, which are paramount parameters in modern drug design. By providing a pathway that avoids the stringent requirements of previous methods, this patent offers a viable solution for the reliable pharmaceutical intermediate supplier seeking to optimize their production pipelines for high-value fluorinated compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been plagued by significant operational challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional routes often rely on the condensation of 3,5-ditrifluoromethyl-1,3,4-oxadiazoles with primary amines or the cyclization of trifluoromethyl hydrazides with amidines, processes that frequently demand harsh reaction conditions and extended reaction times. Moreover, earlier methodologies exhibited a notoriously narrow substrate scope, particularly failing to accommodate alkyl hydrazones, thereby restricting the chemical diversity accessible to medicinal chemists. These legacy methods also suffered from low reaction yields and required rigorous anhydrous and oxygen-free environments, which drastically increase manufacturing costs and complicate supply chain logistics for high-purity OLED material and API precursors.
The Novel Approach
In stark contrast, the novel approach detailed in CN111978265B utilizes readily available acyl hydrazides and trifluoroethylimidoyl chlorides as starting materials to construct the triazole ring with exceptional efficiency. This method employs a strategic combination of sodium bicarbonate as a base and ferric chloride as a Lewis acid promoter, enabling the reaction to proceed smoothly in common aprotic solvents like 1,4-dioxane without the need for inert atmosphere protection. The versatility of this new route is demonstrated by its ability to synthesize a wide array of derivatives, including those with diverse alkyl and aryl substituents at the 3 and 4 positions, as shown in the specific examples below.
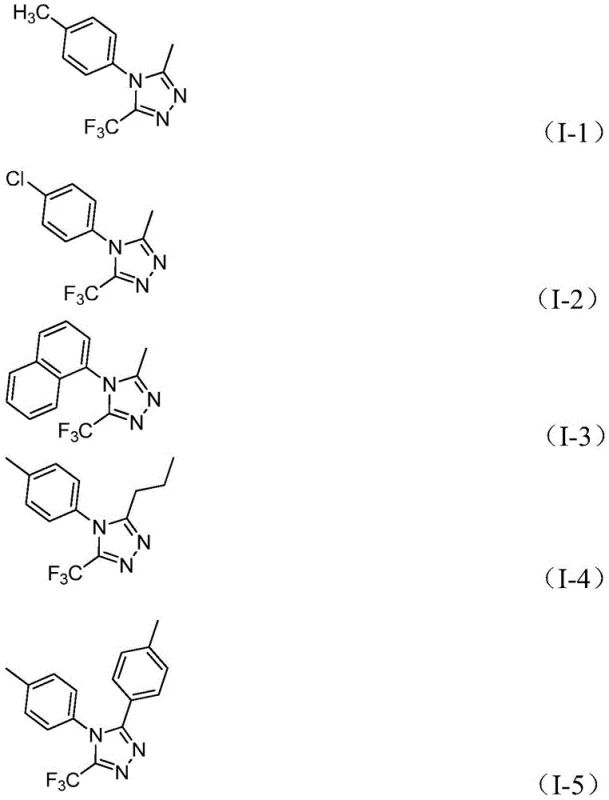
This expanded substrate tolerance allows for the rapid generation of chemical libraries for drug discovery while simultaneously simplifying the manufacturing process for cost reduction in electronic chemical manufacturing and related sectors. The elimination of sensitive reagents and the use of stable, commodity chemicals mark a substantial shift towards more sustainable and economically viable production strategies.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between base-promoted nucleophilic attack and Lewis acid-catalyzed dehydration. Initially, sodium bicarbonate facilitates the intermolecular formation of a carbon-nitrogen bond between the hydrazide and the imidoyl chloride, generating a trifluoroacetamidine intermediate under mild thermal conditions. Subsequently, the addition of ferric chloride acts as a potent Lewis acid to activate the intermediate towards intramolecular dehydration and cyclization, ultimately yielding the stable 5-trifluoromethyl-1,2,4-triazole core. This sequential activation ensures high conversion rates and minimizes the formation of side products that typically arise from uncontrolled reactivity in harsher acidic or basic environments.
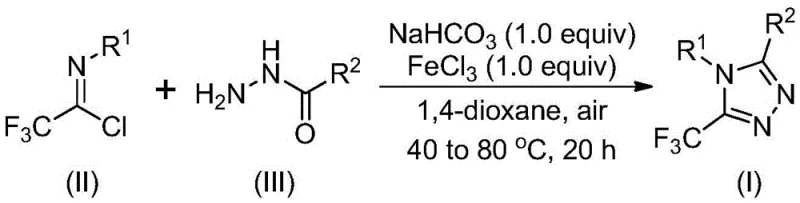
From an impurity control perspective, the use of ferric chloride is particularly advantageous because it promotes the specific cyclization pathway while tolerating various functional groups such as halogens and alkoxy groups on the aromatic rings. The mild nature of the reaction conditions prevents the decomposition of sensitive moieties, ensuring that the final product maintains a high purity profile essential for regulatory compliance in the pharmaceutical industry. This mechanistic understanding allows process chemists to fine-tune reaction parameters to maximize yield and minimize waste, directly contributing to reducing lead time for high-purity pharmaceutical intermediates.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Derivatives Efficiently
The synthesis protocol outlined in the patent provides a straightforward, two-stage heating procedure that is easily adaptable to standard laboratory and pilot plant equipment. The process begins with the mixing of the imidoyl chloride and hydrazide substrates with sodium bicarbonate in an organic solvent, followed by an initial heating phase to form the key intermediate. Detailed standardized synthesis steps see the guide below.
- Mix trifluoroethylimidoyl chloride, hydrazide, and sodium bicarbonate in an aprotic solvent like 1,4-dioxane and stir at 30-50°C.
- After the initial reaction period, add ferric chloride (FeCl3) to the mixture to promote cyclization.
- Heat the reaction system to 70-90°C for 6-10 hours, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this FeCl3-catalyzed methodology presents a compelling value proposition centered on cost efficiency and operational reliability. The shift away from exotic catalysts and sensitive reagents towards commodity chemicals like ferric chloride and sodium bicarbonate fundamentally alters the cost structure of producing these valuable heterocycles. By eliminating the need for specialized anhydrous solvents and inert gas lines, manufacturers can achieve significant cost savings in facility operations and raw material procurement.
- Cost Reduction in Manufacturing: The utilization of inexpensive and widely available catalysts such as ferric chloride removes the financial burden associated with precious metal catalysts often used in cross-coupling reactions. Furthermore, the simplified workup procedure, which involves basic filtration and silica gel treatment, reduces the consumption of purification materials and labor hours. This streamlined process flow translates directly into lower unit costs, allowing companies to maintain competitive pricing margins while adhering to strict quality standards for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Since the starting materials, including various substituted aromatic amines and acyl chlorides, are commercially available from multiple global suppliers, the risk of supply chain disruption is significantly mitigated. The robustness of the reaction against moisture and oxygen means that storage and handling requirements for raw materials are less stringent, reducing the likelihood of batch failures due to reagent degradation. This reliability ensures consistent production schedules and dependable delivery timelines for downstream clients requiring complex polymer additives or agrochemical intermediates.
- Scalability and Environmental Compliance: The reaction conditions described are inherently scalable, having been demonstrated effectively from milligram to gram scales with the potential for multi-kilogram production without fundamental process changes. The use of common solvents like 1,4-dioxane facilitates easier solvent recovery and recycling, aligning with modern green chemistry principles and environmental regulations. This scalability supports the commercial scale-up of complex pharmaceutical intermediates, enabling manufacturers to respond rapidly to market demand fluctuations without compromising on safety or environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route, derived directly from the experimental data and beneficial effects reported in the patent documentation. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What are the primary advantages of this FeCl3-catalyzed method over traditional routes?
A: This method eliminates the need for harsh anhydrous or oxygen-free conditions, utilizes inexpensive and commercially available catalysts like ferric chloride, and offers a broader substrate scope including alkyl hydrazones which were previously unreactive.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is highly scalable due to its operational simplicity, use of common solvents like 1,4-dioxane, and the avoidance of sensitive reagents, making it ideal for commercial manufacturing of pharmaceutical intermediates.
Q: What types of substituents are tolerated on the aromatic rings?
A: The reaction demonstrates excellent functional group tolerance, accommodating various substituents such as methyl, methoxy, halogens, and trifluoromethyl groups at ortho, meta, and para positions without significant yield loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the FeCl3-catalyzed synthesis route for producing high-quality 5-trifluoromethyl-1,2,4-triazole derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent supply regardless of volume requirements. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest international standards for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to leverage this advanced technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you accelerate your development timeline and secure a competitive advantage in the global market.
