Advanced Metal-Free Synthesis of Substituted Dihydrophenanthroline for Commercial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more sustainable and efficient synthetic routes for complex heterocyclic scaffolds. A significant advancement in this domain is documented in patent CN112480112A, which details a novel method for synthesizing substituted dihydrophenanthroline compounds. These compounds serve as critical bidentate nitrogen ligands, widely utilized in stabilizing transition metal complexes for homogeneous catalysis, as well as in the development of photoelectric materials and sensors. The traditional reliance on harsh conditions or expensive catalysts has long been a bottleneck for R&D teams seeking reliable pharmaceutical intermediates supplier partners. This new approach addresses those challenges by introducing a metal-free protocol that operates under remarkably mild conditions, offering a pathway to high-purity products with simplified downstream processing. By leveraging this technology, manufacturers can achieve substantial cost savings and improved environmental compliance without compromising on the structural integrity or purity of the final dihydrophenanthroline derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the dihydrophenanthroline scaffold has been fraught with significant technical and economic hurdles that impact the overall viability of commercial scale-up of complex polymer additives and related fine chemicals. Prior art methods often necessitate extreme reaction temperatures, such as 180°C, maintained for prolonged periods extending up to 50 hours, which drastically increases energy consumption and operational costs. Furthermore, many existing protocols rely heavily on transition metal catalysts like zinc triflate or cobalt salts, which introduce severe complications regarding product purification and heavy metal residue limits. The presence of these metals often requires additional, costly scavenging steps to meet the stringent purity specifications demanded by the pharmaceutical and electronic material sectors. Additionally, some conventional routes utilize solvents like ethylene glycol which can be difficult to remove completely, leading to potential impurity profiles that complicate regulatory approval processes for active pharmaceutical ingredients. These factors collectively contribute to extended lead times and reduced overall process efficiency, making traditional methods less attractive for modern supply chain requirements.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN112480112A presents a streamlined, metal-free synthesis strategy that fundamentally alters the economic equation for producing these valuable intermediates. By utilizing 2-substituted 6,7-dihydroquinoline-8(5H)-one and propargylamine compounds as starting materials in an alcohol solvent, the reaction proceeds efficiently at temperatures ranging from 70°C to 130°C. The preferred embodiment operates at a moderate 90°C for just 3 hours, representing a drastic reduction in thermal energy input compared to prior art. This method eliminates the need for any metal catalyst, thereby removing the associated costs of catalyst procurement and the subsequent purification burden of metal removal. The broad substrate applicability allows for the introduction of various functional groups, including halogens, alkyls, and aryls, without significant loss in yield, ensuring versatility for diverse R&D applications. This robustness makes it an ideal candidate for cost reduction in electronic chemical manufacturing and other high-value sectors requiring consistent quality.
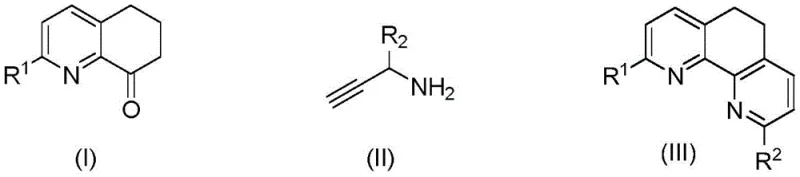
Mechanistic Insights into Metal-Free Thermal Cyclization
The mechanistic pathway of this transformation is a testament to the power of thermal activation in driving cyclization reactions without external catalytic assistance. The reaction likely proceeds through a nucleophilic attack of the amine nitrogen on the carbonyl carbon or a conjugate addition sequence facilitated by the inherent electronic properties of the quinoline ketone system. The absence of a metal center suggests that the reaction relies on the precise alignment of molecular orbitals and the thermal energy provided by the solvent system to overcome the activation barrier. This metal-free nature is particularly advantageous for R&D directors focused on purity, as it inherently prevents the formation of metal-organic impurities that are notoriously difficult to separate. The use of n-butanol as a solvent not only provides the necessary thermal medium but may also participate in hydrogen bonding networks that stabilize transition states, further promoting the formation of the rigid dihydrophenanthroline framework. Understanding these subtle electronic and steric interactions is crucial for optimizing the process for specific substrates, ensuring that the final product meets the rigorous standards required for high-purity OLED material or pharmaceutical applications.
Controlling the impurity profile in this synthesis is achieved through the careful regulation of reaction time and temperature, which minimizes side reactions such as polymerization or over-alkylation. The patent data indicates that extending the reaction time or adjusting the temperature allows for the optimization of yield across a wide range of substituents, from electron-donating methyl groups to electron-withdrawing trifluoromethyl groups. This flexibility ensures that the impurity spectrum remains predictable and manageable, a key factor for supply chain heads concerned with batch-to-batch consistency. The purification process, utilizing alkaline alumina column chromatography, is specifically tailored to remove unreacted starting materials and minor byproducts, resulting in a product of exceptional chemical purity. By avoiding metal catalysts, the risk of generating metal-complexed byproducts is entirely negated, simplifying the analytical validation process. This level of control over the chemical outcome translates directly into enhanced supply chain reliability, as it reduces the likelihood of batch failures due to out-of-specification impurity levels.
How to Synthesize Substituted Dihydrophenanthroline Efficiently
To implement this synthesis effectively, one must adhere to the specific stoichiometric ratios and solvent volumes outlined in the patent to ensure reproducibility and optimal yield. The process begins with the precise weighing of the 2-substituted 6,7-dihydroquinoline-8(5H)-one and the propargylamine derivative, maintaining a mass ratio between 1:1.5 and 1:4 to drive the equilibrium towards product formation. The reaction is conducted in a standard glass vessel equipped with a condenser to prevent solvent loss during the heating phase, which is critical for maintaining the concentration of reactants. Detailed standardized synthesis steps see the guide below for the exact procedural workflow that guarantees success in a laboratory or pilot plant setting.
- Mix 2-substituted 6,7-dihydroquinoline-8(5H)-one and propargylamine compounds in a mass ratio of 1: 1.5 to 4 with n-butanol solvent.
- Heat the reaction mixture to 70-130°C, preferably 90°C, and maintain for 3 to 24 hours depending on substrate reactivity.
- Cool to room temperature, concentrate under reduced pressure, and purify via column chromatography using alkaline alumina.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the elimination of transition metal catalysts represents a significant strategic advantage that directly impacts the bottom line of chemical manufacturing operations. The removal of expensive metals such as palladium, platinum, or even cheaper alternatives like zinc or cobalt from the bill of materials results in immediate raw material cost reductions. Furthermore, the absence of these metals obviates the need for specialized scavenger resins or complex extraction protocols designed to lower residual metal content to ppm levels, which are often required for pharmaceutical grade intermediates. This simplification of the downstream processing workflow leads to substantial cost savings in terms of both consumables and labor hours, allowing procurement managers to negotiate more competitive pricing structures. The use of common alcohol solvents like n-butanol, which are readily available and inexpensive compared to specialized anhydrous solvents, further contributes to the overall economic efficiency of the process. These factors combine to create a highly cost-effective manufacturing route that enhances the competitiveness of the final product in the global market.
- Cost Reduction in Manufacturing: The metal-free nature of this synthesis eliminates the procurement costs associated with transition metal catalysts, which can be volatile in price and subject to supply constraints. Additionally, the removal of metal scavenging steps reduces the consumption of auxiliary chemicals and filtration media, leading to a leaner and more economical production process. The simplified workup procedure also decreases the volume of waste generated, lowering disposal costs and aligning with green chemistry principles. By streamlining the synthesis to fewer steps with higher atom economy, manufacturers can achieve significant operational efficiencies that translate into better margins. This approach ensures that the production of high-purity pharmaceutical intermediates remains financially viable even in fluctuating market conditions.
- Enhanced Supply Chain Reliability: Relying on readily available starting materials and common solvents mitigates the risk of supply chain disruptions caused by the scarcity of specialized reagents. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even when sourcing from different vendors. This stability is crucial for supply chain heads who need to guarantee continuous production schedules to meet customer demand without interruption. The reduced complexity of the process also shortens the manufacturing cycle time, allowing for faster turnaround on orders and improved responsiveness to market needs. Consequently, partners can rely on a steady flow of materials without the delays often associated with complex catalytic processes.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of toxic heavy metals make this process inherently safer and easier to scale from kilogram to tonne quantities. Regulatory compliance is significantly simplified as there is no need to monitor and report on heavy metal discharge or residues, reducing the administrative burden on environmental health and safety teams. The use of alcohol solvents facilitates easier solvent recovery and recycling, further minimizing the environmental footprint of the operation. This alignment with sustainability goals not only meets current regulatory standards but also future-proofs the manufacturing process against tightening environmental legislation. Such attributes are increasingly important for companies aiming to maintain a strong corporate social responsibility profile while expanding their production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent's background and beneficial effects sections. These insights are designed to clarify the operational advantages and technical feasibility for potential partners evaluating this technology for their own production lines. Understanding these details is essential for making informed decisions about integrating this route into existing manufacturing portfolios. The answers provided reflect the specific data points and claims made within the intellectual property documentation to ensure accuracy and relevance.
Q: Does this synthesis method require transition metal catalysts?
A: No, the method described in patent CN112480112A operates without any metal catalyst, eliminating the need for expensive metal removal steps and reducing heavy metal contamination risks.
Q: What are the optimal reaction conditions for high yield?
A: The optimal conditions involve using n-butanol as the solvent at a temperature of 90°C for approximately 3 hours, which balances reaction rate and energy consumption effectively.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the mild reaction conditions, absence of sensitive metal catalysts, and use of common alcohol solvents make this process highly scalable and safe for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Dihydrophenanthroline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain a competitive edge in the global fine chemical market. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like the metal-free dihydrophenanthroline synthesis can be successfully translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify every batch. Our capability to handle complex heterocyclic chemistry allows us to offer customized solutions that address the specific needs of R&D directors and procurement managers alike. By partnering with us, you gain access to a supply chain that prioritizes quality, consistency, and technological innovation.
We invite you to engage with our technical procurement team to discuss how this metal-free synthesis can be adapted to your specific project requirements. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to this greener, more efficient route for your supply chain. Our specialists are ready to provide specific COA data and route feasibility assessments to support your decision-making process. Whether you require small quantities for research or large volumes for commercial manufacturing, NINGBO INNO PHARMCHEM is positioned to be your trusted partner in delivering high-quality chemical intermediates. Contact us today to explore the possibilities of optimizing your production with our advanced synthesis capabilities.
