Advanced Metal-Free Synthesis of Substituted Dihydrophenanthroline for Commercial Scale
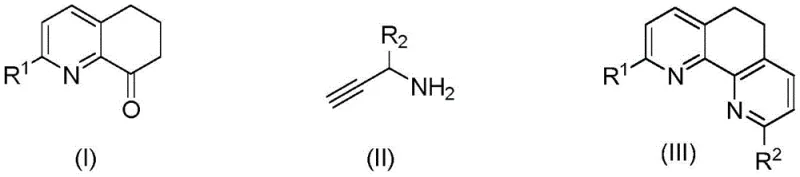
The chemical landscape for synthesizing nitrogen-containing heterocycles is constantly evolving, with a significant shift towards greener, more efficient methodologies. Patent CN112480112B introduces a groundbreaking approach for the synthesis of substituted dihydrophenanthroline compounds, a class of molecules highly valued for their rigidity and electronic properties in coordination chemistry and biomedicine. This technology leverages a metal-free thermal cyclization strategy that utilizes 2-substituted 6,7-dihydroquinoline-8(5H)-one and propargylamine derivatives as key starting materials. By eliminating the reliance on expensive and potentially toxic transition metal catalysts, this method addresses critical pain points in modern pharmaceutical intermediate manufacturing, offering a pathway to high-purity products with simplified downstream processing. The robustness of this protocol allows for wide substrate applicability, accommodating various functional groups including halogens, alkyls, and aryls, which is essential for generating diverse chemical libraries for drug discovery.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the dihydrophenanthroline scaffold has relied on harsh conditions and specialized reagents that pose significant challenges for industrial scale-up. Traditional routes often involve reacting 6,7-dihydroquinoline-8(5H)-one with 3-amino acrolein at extreme temperatures around 180°C for extended periods up to 50 hours, which leads to substantial energy consumption and potential decomposition of sensitive functional groups. Other existing methods utilize transition metal catalysts such as zinc triflate or nickel acetylacetonate, which introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). Furthermore, these metal-catalyzed processes frequently require rigorous purification steps to meet stringent regulatory limits for residual metals, thereby increasing production costs and extending lead times. The use of such catalysts also complicates waste management, as the disposal of metal-laden byproducts requires specialized treatment protocols to ensure environmental compliance.
The Novel Approach
In stark contrast, the methodology disclosed in CN112480112B offers a streamlined, metal-free alternative that operates under significantly milder conditions. By employing simple alcohols like n-butanol as both solvent and reaction medium, the process facilitates the cyclization of quinoline ketones and propargylamines at temperatures ranging from 70°C to 130°C. This thermal activation eliminates the need for external catalysts, inherently reducing the impurity profile associated with metal residues. The reaction demonstrates remarkable efficiency, often reaching completion within 3 to 24 hours, which represents a drastic reduction in processing time compared to legacy methods. This approach not only simplifies the operational workflow but also enhances the overall safety of the manufacturing process by avoiding the handling of sensitive metal complexes. The versatility of this system is further evidenced by its ability to tolerate a broad spectrum of substituents on the aromatic rings, ensuring high yields across a diverse range of target molecules.
Mechanistic Insights into Metal-Free Thermal Cyclization
The core of this innovative synthesis lies in the thermal promotion of a cascade cyclization reaction between the carbonyl group of the dihydroquinoline ketone and the amine functionality of the propargylamine derivative. Under the influence of heat in an alcoholic solvent, the nucleophilic attack of the amine on the ketone initiates the formation of an imine intermediate, which subsequently undergoes intramolecular cyclization involving the alkyne moiety. This sequence effectively constructs the fused phenanthroline ring system without the need for Lewis acid activation typically provided by metal salts. The absence of metal catalysts means that the reaction mechanism is driven purely by thermodynamic and kinetic factors controlled by temperature and solvent polarity. This mechanistic simplicity is a major advantage for process chemists, as it removes variables related to catalyst loading, activation, and deactivation, leading to more predictable and reproducible outcomes. Furthermore, the use of n-butanol as a solvent likely plays a dual role, acting as a proton source/sink to facilitate tautomerization steps essential for aromatization of the final heterocyclic core.
From an impurity control perspective, the metal-free nature of this reaction provides a distinct advantage in managing the quality of the final product. In traditional metal-catalyzed syntheses, side reactions often include metal-mediated oligomerization or incomplete conversion due to catalyst poisoning by substrate impurities. By removing the metal component, the primary impurities are limited to unreacted starting materials or simple condensation byproducts, which are generally easier to separate via standard chromatographic techniques. The patent specifies the use of alkaline alumina for purification, which is particularly effective at removing acidic impurities and polar byproducts that might co-elute with the target dihydrophenanthroline. This targeted purification strategy ensures that the final isolated material meets the high purity standards required for use as a ligand in sensitive catalytic applications or as a scaffold in medicinal chemistry programs, where trace contaminants can significantly alter biological activity or catalytic performance.
How to Synthesize Substituted Dihydrophenanthroline Efficiently
To implement this synthesis effectively, precise control over reaction parameters is essential to maximize yield and minimize byproduct formation. The process begins with the careful selection of the alcohol solvent, with n-butanol being the preferred choice due to its higher boiling point which allows for operation at optimal temperatures without excessive pressure buildup. The stoichiometry of the reactants is also critical, with a slight excess of the propargylamine component often recommended to drive the equilibrium towards product formation. Operators should monitor the reaction progress closely, as the optimal time can vary significantly depending on the electronic nature of the substituents on the quinoline ring; electron-withdrawing groups may accelerate the reaction while bulky steric groups might require extended heating. Following the reaction, the workup procedure involves concentration under reduced pressure followed by chromatographic separation, a step that is crucial for isolating the pure crystalline or liquid product from the reaction matrix.
- Combine 2-substituted 6,7-dihydroquinoline-8(5H)-one and propargylamine compounds in a reaction vessel with n-butanol as the solvent.
- Heat the reaction mixture to a temperature range of 70-130°C, preferably 90°C, and maintain for 3 to 24 hours to facilitate cyclization.
- Cool the solution to room temperature, concentrate under reduced pressure, and purify the crude product using column chromatography with alkaline alumina.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route offers tangible benefits that directly impact the bottom line and operational resilience. The elimination of transition metal catalysts removes a significant cost center associated with the purchase of precious or specialized metal salts, which can be subject to volatile market pricing and supply constraints. Additionally, the simplified purification process reduces the consumption of silica gel or other chromatographic media and minimizes the volume of solvent waste generated, leading to substantial cost savings in waste disposal and environmental compliance fees. The mild reaction conditions also translate to lower energy requirements for heating and cooling, further enhancing the economic viability of large-scale production. By streamlining the manufacturing process, companies can achieve faster turnaround times from raw material sourcing to final product delivery, thereby improving supply chain reliability and responsiveness to market demands.
- Cost Reduction in Manufacturing: The removal of expensive metal catalysts and the associated scavenging agents drastically lowers the raw material costs per kilogram of product. Without the need for rigorous heavy metal testing and removal steps, the analytical and quality control overhead is significantly reduced. This leaner process architecture allows for a more competitive pricing structure for high-purity dihydrophenanthroline intermediates, making it an attractive option for cost-sensitive projects. The use of commodity solvents like n-butanol instead of specialized anhydrous or degassed solvents further contributes to the overall reduction in manufacturing expenses.
- Enhanced Supply Chain Reliability: Relying on readily available organic starting materials rather than specialized catalysts mitigates the risk of supply disruptions caused by geopolitical issues or mining constraints affecting metal availability. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the likelihood of production delays due to failed batches or out-of-specification results. This stability allows for more accurate forecasting and inventory planning, ensuring that critical intermediates are available when needed for downstream synthesis campaigns. The scalability of the process from gram to multi-kilogram scales without significant re-optimization further strengthens supply chain continuity.
- Scalability and Environmental Compliance: The metal-free nature of this synthesis aligns perfectly with green chemistry principles, reducing the environmental footprint of the manufacturing process. The absence of heavy metals simplifies the treatment of effluent streams, making it easier to meet increasingly stringent environmental regulations regarding industrial discharge. The process is inherently safer to operate at scale, as it avoids the potential hazards associated with handling pyrophoric or toxic metal catalysts. This compliance and safety profile facilitates smoother regulatory approvals and audits, accelerating the time to market for new products utilizing these intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these substituted dihydrophenanthroline compounds. Understanding these details is crucial for R&D teams evaluating this technology for integration into their existing workflows. The answers provided are based on the specific experimental data and embodiments disclosed in the patent literature, ensuring accuracy and relevance for technical decision-making. Clients are encouraged to review these points to assess the feasibility of this method for their specific target molecules and production requirements.
Q: Does this synthesis method require transition metal catalysts?
A: No, the patented method described in CN112480112B operates without the need for any metal catalysts, utilizing thermal energy in an alcohol solvent to drive the cyclization reaction.
Q: What are the typical reaction conditions for this cyclization?
A: The reaction typically proceeds in an alcohol solvent such as n-butanol at temperatures between 70°C and 130°C for a duration of 3 to 24 hours, depending on the specific substrate substituents.
Q: How is the final product purified to ensure high quality?
A: The crude reaction mixture is purified using column chromatography with 200-300 mesh alkaline alumina as the stationary phase and a petroleum ether/ethyl acetate mixture as the eluent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydrophenanthroline Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and scalable synthetic routes for complex heterocyclic intermediates like substituted dihydrophenanthrolines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric needs of global pharmaceutical and agrochemical clients. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and assay. Our capability to adapt the metal-free synthesis described in CN112480112B allows us to offer a superior value proposition, combining high quality with cost-effectiveness and supply security.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this metal-free protocol for your supply chain. We are ready to provide specific COA data and route feasibility assessments to demonstrate our commitment to transparency and technical excellence. Let us collaborate to optimize your intermediate sourcing strategy and drive innovation in your drug development pipeline.
