Advanced Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
Advanced Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
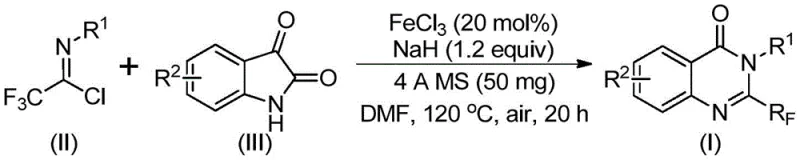
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Quinazolinones, in particular, represent a privileged scaffold found in numerous drugs exhibiting anti-cancer, anticonvulsant, and anti-inflammatory properties. The strategic introduction of a trifluoromethyl group into these scaffolds further enhances their pharmacokinetic profiles by improving lipophilicity and metabolic stability. A groundbreaking preparation method disclosed in patent CN111675662B details a highly efficient synthesis of 2-trifluoromethyl substituted quinazolinone compounds. This innovation leverages an earth-abundant iron catalyst to drive the cyclization of readily available isatins and trifluoroethylimidoyl chlorides. For R&D directors and procurement managers alike, this technology represents a significant leap forward, offering a pathway to high-purity intermediates that bypasses the limitations of traditional precious metal catalysis.
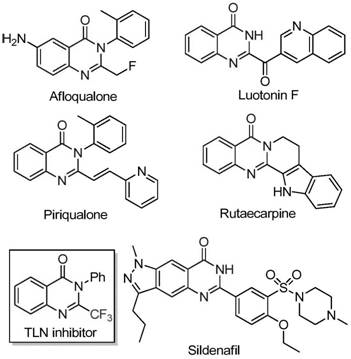
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted quinazolinones has relied heavily on the cyclization of synthons such as anthranilamides or isatoic anhydrides with trifluoroacetic anhydride or ethyl trifluoroacetate. While effective in academic settings, these conventional methodologies suffer from severe drawbacks when translated to industrial scale. The reaction conditions are often harsh, requiring strong bases or high temperatures that can degrade sensitive functional groups. Furthermore, the starting materials, particularly specialized trifluoroacetylating agents, are frequently expensive and subject to supply chain volatility. Perhaps most critically for process chemists, these older routes often exhibit narrow substrate scope and mediocre yields, leading to significant waste generation and difficult purification challenges that inflate the overall cost of goods sold (COGS) for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology described in the referenced patent introduces a paradigm shift by utilizing trifluoroethylimidoyl chloride and isatin as the primary building blocks. This novel approach capitalizes on the high reactivity of the imidoyl chloride moiety, which facilitates a smoother bond formation under milder conditions. By employing ferric chloride as the catalyst, the process avoids the toxicity and cost associated with late-transition metals. The reaction proceeds through a tandem sequence involving initial carbon-nitrogen bond formation followed by an iron-catalyzed decarbonylation and cyclization. This streamlined mechanism not only improves atom economy but also expands the range of compatible substrates, allowing for the facile incorporation of diverse substituents on the aromatic rings, thereby enabling the rapid generation of analog libraries for drug discovery programs.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this synthetic breakthrough lies in the unique catalytic cycle driven by the FeCl3/NaH system. The reaction initiates with the activation of the trifluoroethylimidoyl chloride by the base, generating a reactive nucleophile that attacks the carbonyl carbon of the isatin derivative. This step forms a transient trifluoroacetamidine intermediate. Subsequently, the iron catalyst plays a pivotal role in promoting the decarbonylation of the isatin moiety. This decarbonylation is the key differentiator, as it effectively removes the extra carbonyl oxygen from the isatin ring, allowing the system to rearrange into the thermodynamically stable quinazolinone core. The presence of 4A molecular sieves in the reaction mixture is crucial, as they sequester water generated during the process, driving the equilibrium towards product formation and preventing the hydrolysis of the sensitive imidoyl chloride starting material.
From an impurity control perspective, this mechanism offers distinct advantages. The use of a single metal catalyst reduces the complexity of the metal residue profile compared to dual-catalyst systems. Moreover, the reaction conditions (120°C in DMF) are optimized to favor the desired cyclization over potential side reactions such as polymerization or over-fluorination. The high functional group tolerance observed in the experimental data suggests that the catalytic species is selective for the intended transformation, minimizing the formation of regioisomers or byproducts that would otherwise complicate downstream purification. This selectivity is paramount for meeting the stringent purity specifications required by regulatory bodies for pharmaceutical intermediates, ensuring that the final product possesses a clean impurity profile suitable for subsequent coupling reactions.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The operational simplicity of this protocol makes it highly attractive for process development teams aiming to scale up production. The procedure involves a straightforward one-pot reaction where all reagents are combined in a polar aprotic solvent, typically DMF, which ensures excellent solubility of both the organic substrates and the inorganic catalyst. The reaction profile is biphasic in terms of temperature, starting at a moderate 40°C to allow for the initial coupling, followed by heating to 120°C to drive the cyclization and decarbonylation to completion. This controlled temperature ramp helps manage the exothermicity of the reaction and ensures safety during scale-up. Detailed standardized synthetic steps for implementing this protocol in your facility are provided in the guide below.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C for 18-20 hours under air atmosphere.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the final quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iron-catalyzed methodology offers tangible economic and logistical benefits that directly impact the bottom line. The shift away from precious metals and exotic reagents to commodity chemicals creates a more resilient supply chain less susceptible to geopolitical fluctuations in metal prices. Additionally, the simplified workup procedure, which involves basic filtration and standard chromatography, reduces the consumption of silica gel and solvents compared to more complex multistep syntheses. These factors collectively contribute to a leaner manufacturing process that aligns with modern green chemistry principles while enhancing profit margins.
- Cost Reduction in Manufacturing: The replacement of expensive palladium or rhodium catalysts with ferric chloride results in a drastic reduction in catalyst costs, as iron salts are orders of magnitude cheaper and more abundant. Furthermore, the use of isatin and trifluoroethylimidoyl chloride as starting materials leverages widely available commodity feedstocks, avoiding the premium pricing of specialized trifluoroacetylating agents. This fundamental shift in raw material selection significantly lowers the variable cost per kilogram of the produced intermediate, allowing for more competitive pricing strategies in the global market without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Sourcing critical reagents is a major bottleneck in pharmaceutical manufacturing, but this method utilizes chemicals that are produced on a massive industrial scale globally. Isatin and simple aromatic amines used to prepare the imidoyl chloride are standard catalog items with multiple qualified suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, ensuring consistent batch-to-batch performance and reliable delivery schedules even during periods of raw material scarcity.
- Scalability and Environmental Compliance: The protocol is inherently scalable, having been demonstrated to work efficiently from milligram to gram scales with consistent yields. The absence of toxic heavy metals simplifies the environmental compliance landscape, as wastewater treatment does not require specialized heavy metal scavenging steps. This ease of waste management translates to lower disposal costs and a smaller environmental footprint, facilitating faster regulatory approvals for new manufacturing sites and supporting the company's sustainability goals through the adoption of greener catalytic technologies.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is critical for successful implementation. The following questions address common concerns regarding the practical application of this technology in a commercial setting. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable foundation for your process evaluation.
Q: What are the primary advantages of using FeCl3 over precious metal catalysts?
A: Using ferric chloride (FeCl3) eliminates the need for expensive precious metals like palladium or rhodium, drastically reducing raw material costs and simplifying heavy metal removal processes required for pharmaceutical compliance.
Q: Can this synthesis method tolerate diverse functional groups?
A: Yes, the method demonstrates excellent functional group tolerance, successfully accommodating substituents such as halogens (F, Cl, Br), alkyl groups (Me), and electron-withdrawing groups (NO2) on the aromatic rings without significant yield loss.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The protocol uses cheap, commercially available starting materials like isatin and operates under relatively mild conditions (up to 120°C), making it highly scalable from gram-level laboratory synthesis to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this cost-effective synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you optimize your supply chain for the future.
