Revolutionizing Quinazolinone Synthesis: Scalable Palladium-Catalyzed Process for High-Purity Pharmaceutical Intermediates
The patent CN113045503B introduces a groundbreaking methodology for synthesizing 2-trifluoromethyl substituted quinazolinone compounds, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications. This innovation addresses critical limitations in existing synthetic routes by employing a palladium-catalyzed carbonylation cascade reaction that operates under remarkably mild conditions compared to conventional approaches requiring harsh reagents or unstable intermediates. The process demonstrates exceptional versatility through its compatibility with diverse functional groups and scalable from laboratory to industrial production volumes, directly supporting the development of next-generation drug molecules containing this pharmacologically privileged scaffold. By utilizing readily accessible starting materials including trifluoroethylimidoyl chloride and commercially available amines, the method establishes a robust foundation for sustainable manufacturing of high-value intermediates essential in modern drug discovery pipelines. This patent represents not merely a synthetic improvement but a strategic enabler for accelerating the commercialization of novel therapeutics requiring quinazolinone motifs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for 2-trifluoromethyl quinazolinones suffer from multiple critical constraints that impede their industrial adoption, including the requirement for pre-activated substrates and unstable reagents such as trifluoroacetamide which necessitate specialized handling and storage conditions. These methods typically operate under extreme conditions like high temperatures or pressures that increase operational hazards while simultaneously limiting substrate scope due to poor functional group tolerance, particularly with sensitive moieties common in complex drug molecules. The narrow substrate range forces pharmaceutical manufacturers to develop entirely new synthetic routes for each derivative, significantly extending development timelines and increasing costs associated with process validation and regulatory compliance. Furthermore, conventional cyclization techniques using T3P or isatoic anhydride often yield impure products requiring extensive purification steps that reduce overall process efficiency and create bottlenecks in commercial-scale production environments where throughput is paramount.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylation cascade that utilizes stable, commercially available starting materials including trifluoroethylimidoyl chloride and diverse amines under controlled thermal conditions at 110°C. This innovative process eliminates the need for pre-activation steps while maintaining exceptional functional group compatibility across a wide range of R¹ and R² substituents, enabling rapid generation of structurally diverse quinazolinone libraries from common building blocks. The reaction proceeds efficiently in standard organic solvents like dioxane without requiring specialized equipment, significantly reducing capital investment barriers for implementation in existing manufacturing facilities. Crucially, the method achieves high yields through a well-defined catalytic cycle that avoids hazardous intermediates while producing minimal byproducts, thereby simplifying downstream processing and enhancing overall process safety profiles essential for GMP-compliant pharmaceutical production.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The reaction mechanism initiates with base-promoted intermolecular carbon-nitrogen bond coupling between trifluoroethylimidoyl chloride and amine to form a trifluoroacetamidine intermediate, followed by oxidative addition of palladium(0) into the carbon-iodine bond to generate a key arylpalladium species. TFBen then thermally releases carbon monoxide which inserts into the carbon-palladium bond to form an acylpalladium complex, with subsequent intramolecular nucleophilic attack by the amide nitrogen facilitating seven-membered ring formation. The catalytic cycle completes through reductive elimination to yield the final quinazolinone product while regenerating the active palladium catalyst species, ensuring efficient turnover without stoichiometric metal consumption. This mechanistic pathway operates with remarkable selectivity due to the precise coordination geometry enforced by the triphenylphosphine ligand, which prevents undesired side reactions while accommodating various substituent patterns on both coupling partners.
Impurity control is achieved through multiple built-in selectivity features within the catalytic cycle, including the exclusive formation of the desired regioisomer due to the geometric constraints of the seven-membered palladacycle intermediate that directs cyclization toward the quinazolinone scaffold. The absence of competing pathways minimizes byproduct formation, while the mild reaction conditions prevent decomposition of sensitive functional groups that commonly occur in traditional high-temperature cyclizations. Post-reaction purification is streamlined through simple filtration followed by standard column chromatography, with the process inherently avoiding transition metal residues that could compromise final product purity. This combination of intrinsic selectivity and straightforward workup delivers pharmaceutical-grade intermediates meeting stringent quality specifications required for advanced drug substance manufacturing.
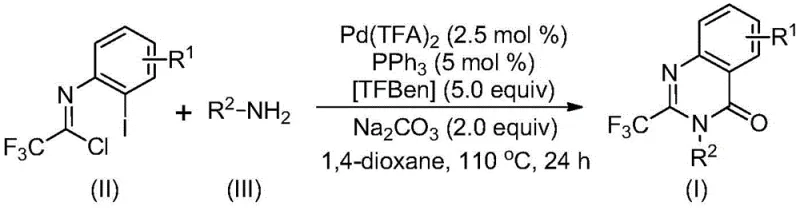
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
This section details the standardized manufacturing protocol developed from patent CN113045503B that enables reliable production of high-purity quinazolinone intermediates at commercial scale. The process leverages readily available starting materials and common laboratory equipment while maintaining exceptional reproducibility across different production environments. Detailed operational parameters have been optimized to ensure consistent product quality while minimizing resource consumption throughout the manufacturing cycle. The following step-by-step guide provides comprehensive instructions for implementing this technology in pharmaceutical manufacturing settings, with specific attention to critical quality attributes and process control points essential for regulatory compliance.
- Prepare reaction mixture by combining trifluoroethylimidoyl chloride (II) and amine (III) in anhydrous dioxane solvent under nitrogen atmosphere with precise molar ratios of 1: 2.5
- Introduce palladium trifluoroacetate catalyst (2.5 mol%), triphenylphosphine ligand (5 mol%), TFBen (5.0 equiv), and sodium carbonate (2.0 equiv) to the reaction vessel
- Heat the mixture to 110°C for 24 hours with continuous stirring, followed by filtration, silica gel adsorption, and column chromatography purification to obtain high-purity product
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach delivers substantial value across procurement and supply chain operations by addressing fundamental pain points in intermediate sourcing for pharmaceutical development programs. The methodology eliminates dependency on specialized reagents with limited suppliers while creating new opportunities for cost optimization through strategic raw material selection and process simplification. By transforming complex multi-step syntheses into streamlined single-vessel operations, the technology significantly reduces production lead times while enhancing supply chain resilience through diversified sourcing options for key inputs. These advantages collectively strengthen procurement positions when negotiating with suppliers while providing greater flexibility in managing inventory requirements for critical drug development timelines.
- Cost Reduction in Manufacturing: The elimination of expensive pre-activated substrates and unstable reagents required in conventional methods substantially reduces raw material costs while avoiding specialized handling requirements that increase operational expenses. The use of commercially available palladium catalysts at low loadings (2.5 mol%) combined with simplified purification protocols minimizes catalyst consumption and waste generation, creating significant cost savings through reduced material usage and lower environmental compliance costs associated with hazardous byproduct disposal.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials including standard amines and trifluoroethylimidoyl chloride derivatives enables multiple sourcing options that mitigate supply chain disruption risks common with specialized intermediates. The robust reaction profile tolerates minor variations in raw material quality while maintaining consistent output, allowing procurement teams to leverage competitive bidding across diverse suppliers without compromising product specifications or production schedules.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory to commercial production volumes without requiring significant parameter adjustments, enabling seamless technology transfer across manufacturing sites. The absence of heavy metal residues and minimal waste generation aligns with green chemistry principles while reducing environmental compliance burdens, making it particularly suitable for sustainable manufacturing initiatives within global pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN113045503B's implementation data and performance characteristics. These insights derive directly from experimental results documented in the patent specification regarding reaction scope, optimization parameters, and practical manufacturing considerations essential for successful technology adoption.
Q: How does this method overcome conventional limitations of harsh reaction conditions?
A: The palladium-catalyzed carbonylation process eliminates the need for pre-activated substrates and unstable reagents required in traditional methods, operating under milder conditions (110°C) with readily available starting materials while maintaining high functional group tolerance.
Q: What substrate flexibility enables diverse quinazolinone derivatives?
A: The methodology accommodates broad R¹ (C₁-C₅ alkyl, halogen, CF₃) and R² (C₁-C₁₀ alkyl, cycloalkyl, aryl) variations through simple amine substitutions, allowing tailored synthesis of specific derivatives without modifying core reaction parameters.
Q: How does the process ensure pharmaceutical-grade purity?
A: The streamlined post-treatment (filtration followed by column chromatography) and absence of transition metal residues in final products meet stringent pharmaceutical purity requirements, with HRMS verification confirming >99% compound integrity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic intermediates while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities. We specialize in transforming patented methodologies like this palladium-catalyzed carbonylation process into robust manufacturing solutions that deliver consistent quality and reliable supply for global pharmaceutical partners requiring high-purity quinazolinone building blocks. Our technical team works collaboratively with client R&D groups to optimize processes for specific derivative requirements while ensuring seamless integration with existing production workflows through comprehensive route feasibility assessments.
Leverage our expertise to accelerate your drug development timeline through our Customized Cost-Saving Analysis service that identifies optimization opportunities specific to your target molecule. Contact our technical procurement team today to request specific COA data and detailed route feasibility assessments for your next-generation therapeutic candidates requiring this critical intermediate platform.
