Advanced FeCl3-Catalyzed Synthesis of 3,3'-Diindole Derivatives for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways for bioactive structural units, and the recent advancements detailed in patent CN101935297B represent a significant leap forward in the preparation of 3,3'-diindole derivatives. These compounds serve as critical scaffolds in natural products with potent anticancer activity and function effectively as chiral catalysts in asymmetric synthesis, making them highly valuable assets for drug discovery pipelines. The disclosed methodology utilizes anhydrous ferric chloride as a catalyst to facilitate the oxidative coupling of 2-arylindoles, offering a greener alternative to legacy processes that often rely on toxic reagents. By leveraging earth-abundant iron catalysts, this technology not only enhances reaction selectivity but also aligns with modern sustainability goals required by global regulatory bodies. As a reliable pharmaceutical intermediates supplier, understanding the nuances of such patented technologies allows us to offer clients superior process options that balance efficiency with environmental compliance. The structural diversity achievable through this method is extensive, accommodating various substituents on the aryl rings without compromising yield or purity standards.
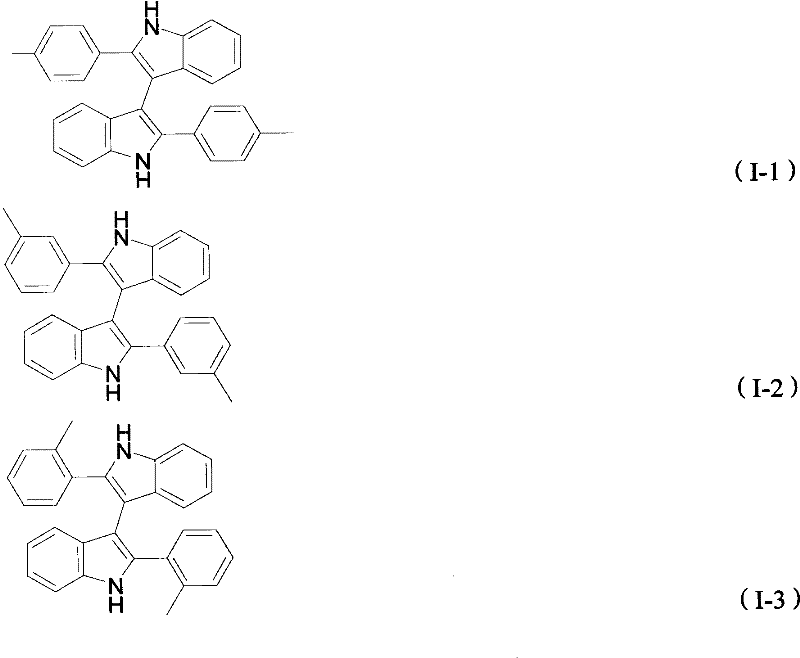
Historically, the construction of the 3,3'-diindole core has been plagued by synthetic inefficiencies and environmental hazards that hindered widespread commercial adoption. Conventional approaches, such as the nucleophilic reaction between indole and isatin followed by reduction, involve multiple steps and generate substantial chemical waste due to the use of stoichiometric reducing agents like sodium borohydride. Another prevalent method involves Suzuki-Miyaura coupling which, while effective, necessitates the use of strong corrosive reagents like liquid bromine for prior halogenation, posing severe safety risks and increasing disposal costs. These traditional pathways often suffer from harsh reaction conditions and poor atom economy, making them less attractive for large-scale manufacturing where cost reduction in pharmaceutical intermediates manufacturing is a primary objective. Furthermore, the reliance on precious metal catalysts in cross-coupling reactions introduces supply chain vulnerabilities and price volatility that procurement teams strive to avoid. The limitations of these older technologies create a pressing need for innovation that can deliver high-purity intermediates without the associated ecological and economic burdens.
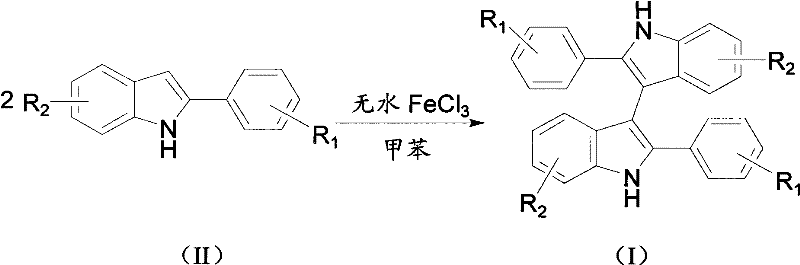
The novel approach presented in the patent data overcomes these historical barriers by introducing a direct oxidative coupling strategy mediated by anhydrous ferric chloride in a toluene solvent system. This method streamlines the synthesis into a single pot operation where 2-arylindole compounds react directly under oxygen protection, eliminating the need for pre-functionalization or hazardous halogenating agents. The reaction conditions are remarkably mild, operating effectively within a temperature range of 90 to 150 degrees Celsius, which reduces energy consumption and equipment stress compared to high-temperature alternatives. By substituting expensive transition metals with inexpensive iron salts, the process achieves substantial cost savings while maintaining high reaction selectivity and yield across a broad substrate scope. This technological shift represents a paradigm change in how complex heterocyclic systems are assembled, prioritizing simplicity and safety without sacrificing chemical performance. For supply chain heads, this translates to a more resilient production model that is less dependent on critical raw materials and more adaptable to fluctuating market demands.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Coupling
The mechanistic pathway of this iron-catalyzed transformation involves the activation of the indole ring through Lewis acid coordination, facilitating the formation of a radical cation intermediate under aerobic conditions. Anhydrous ferric chloride acts as a single-electron oxidant that generates the reactive species necessary for the C-C bond formation at the 3-position of the indole nucleus. The presence of oxygen serves as the terminal oxidant, regenerating the active iron species and ensuring the catalytic cycle continues efficiently without the accumulation of reduced metal by-products. This mechanism avoids the formation of stable organometallic intermediates that often require rigorous quenching procedures, thereby simplifying the downstream workup and purification stages. Understanding this catalytic cycle is crucial for R&D directors who need to assess the feasibility of scaling this chemistry for commercial production of complex pharmaceutical intermediates. The robustness of the iron catalyst against various functional groups ensures that diverse substituents such as alkyl, alkoxy, and halogen atoms can be tolerated without inhibiting the reaction progress.
Impurity control is a paramount concern in the synthesis of pharmaceutical intermediates, and this iron-catalyzed route offers distinct advantages in managing side reactions and by-product profiles. The high selectivity of the ferric chloride catalyst minimizes the formation of polymeric tars or over-oxidized species that are common in uncontrolled oxidative processes. The reaction conditions are tuned to favor the dimerization specifically at the 3-position, preventing unwanted substitution at other reactive sites on the indole ring which could complicate purification. Additionally, the use of toluene as a solvent provides a favorable medium for dissolving both the organic substrates and the inorganic catalyst, ensuring homogeneous reaction kinetics that lead to consistent product quality. For quality assurance teams, this means that the resulting crude material requires less intensive purification, reducing the loss of valuable product during chromatography or crystallization steps. The ability to produce high-purity intermediates with a clean impurity profile is essential for meeting the stringent regulatory requirements of global health authorities.
How to Synthesize 3,3'-Diindole Derivatives Efficiently
Implementing this synthesis route requires careful attention to reagent ratios and atmospheric conditions to maximize yield and reproducibility in a laboratory or pilot plant setting. The standard protocol involves charging a reaction vessel with the 2-arylindole substrate and anhydrous ferric chloride in toluene, followed by heating under an oxygen balloon to maintain the necessary oxidative environment. Detailed standardized synthesis steps are provided below to guide technical teams through the precise execution of this patented methodology. Adhering to the specified molar ratios of substrate to catalyst, typically ranging from 1:0.1 to 1:0.5, is critical for achieving optimal conversion rates without excessive catalyst loading. The reaction time generally spans from 15 to 25 hours depending on the specific substituents present on the starting material, requiring monitoring to determine the exact endpoint for each batch.
- Charge a reaction vessel with 2-arylindole compound and anhydrous ferric chloride in toluene solvent under stirring conditions.
- Maintain the reaction mixture at a temperature between 90°C and 150°C under an oxygen atmosphere for 15 to 25 hours.
- Remove solvent by rotation and purify the crude product via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iron-catalyzed technology offers profound benefits for procurement managers and supply chain leaders focused on optimizing operational expenditures and risk mitigation. The elimination of precious metal catalysts and corrosive halogenating agents directly translates to a significant reduction in raw material costs and hazardous waste disposal fees. This process enhancement allows for cost reduction in pharmaceutical intermediates manufacturing by simplifying the supply chain logistics associated with sourcing specialized reagents. Furthermore, the use of readily available starting materials ensures that production schedules are not disrupted by shortages of exotic chemicals, thereby enhancing supply chain reliability and continuity. The scalability of this method is supported by its simple operational requirements, which do not demand specialized high-pressure equipment or cryogenic conditions, making it accessible for commercial scale-up of complex pharmaceutical intermediates. These factors collectively contribute to a more sustainable and economically viable production model that aligns with the strategic goals of modern chemical enterprises.
- Cost Reduction in Manufacturing: The substitution of expensive palladium or rhodium catalysts with abundant iron salts drastically lowers the direct material costs associated with each production batch. Additionally, the simplified workup procedure reduces solvent consumption and labor hours required for purification, leading to substantial overall cost savings. By avoiding the use of hazardous bromine, the facility also saves on safety infrastructure and waste treatment expenses, further improving the bottom line. This economic efficiency makes the process highly competitive for large-volume manufacturing where margin optimization is critical.
- Enhanced Supply Chain Reliability: Sourcing iron chloride and toluene is far more stable and predictable than relying on supply chains for precious metals or specialized ligands. This stability reduces the risk of production delays caused by vendor shortages or geopolitical issues affecting rare metal availability. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output. Consequently, reducing lead time for high-purity pharmaceutical intermediates becomes achievable through a more resilient and dependable supply network.
- Scalability and Environmental Compliance: The green nature of this synthesis route facilitates easier regulatory approval and permits for expansion, as it generates less toxic waste and emissions. The ability to run the reaction at atmospheric pressure with oxygen simplifies the engineering requirements for scaling from kilograms to tons. This environmental compatibility supports corporate sustainability initiatives and helps maintain a positive relationship with local communities and regulatory agencies. The process is designed to be inherently safer, reducing the potential for accidents and ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide accurate guidance. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing production portfolios. The clarity provided here aims to resolve uncertainties regarding catalyst performance, product quality, and process safety.
Q: What are the advantages of using FeCl3 over traditional catalysts for diindole synthesis?
A: Unlike traditional methods requiring corrosive bromine or expensive palladium, FeCl3 is earth-abundant, cost-effective, and operates under milder conditions with high selectivity.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The reaction demonstrates excellent selectivity with minimal by-product formation, and the final purification via column chromatography ensures stringent purity specifications suitable for API intermediates.
Q: Is this synthetic route environmentally friendly for large-scale production?
A: Yes, the process eliminates hazardous halogenating agents and uses recyclable solvents, significantly reducing environmental impact and waste disposal costs compared to conventional routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-Diindole Derivative Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to full-scale manufacturing. Our commitment to quality is upheld through stringent purity specifications and rigorous QC labs that verify every batch meets the highest industry standards. We understand the critical nature of pharmaceutical intermediates and dedicate our resources to maintaining supply continuity and technical excellence. Our team of experts is ready to collaborate with you to optimize this FeCl3-catalyzed route for your specific application needs. Partnering with us means gaining access to a wealth of chemical knowledge and production capacity that can accelerate your drug development timelines.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements and quality expectations. By engaging with us, you can obtain specific COA data and route feasibility assessments that demonstrate the tangible benefits of this advanced synthesis method. Let us help you secure a stable supply of high-quality 3,3'-diindole derivatives that drive your innovation forward. Reach out today to discuss how we can support your supply chain with reliable and cost-effective solutions.
