Scalable Enaminone Production via Sodium Enolate Intermediates for Global Pharma Supply
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more efficient and cost-effective routes to critical building blocks. A pivotal development in this arena is detailed in Chinese Patent CN101781222A, which discloses a robust method for preparing enaminone compounds through an enolate intermediate. Enaminones are versatile multifunctional intermediates possessing both electrophilic carbonyl centers and nucleophilic amine centers, making them indispensable for synthesizing nitrogen-containing heterocycles found in numerous active pharmaceutical ingredients (APIs). This patent introduces a streamlined approach that bypasses the limitations of traditional condensation reactions, utilizing readily available ketones with alpha-methyl or alpha-methylene groups. By leveraging a sodium-mediated formylation followed by dehydration condensation with dimethylamine hydrochloride, this technology offers a compelling alternative for the reliable enaminone supplier seeking to optimize their manufacturing portfolio. The significance of this innovation lies not just in the chemical transformation itself, but in its potential to redefine cost structures and supply chain reliability for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of enaminone compounds has been plagued by significant operational and economic hurdles that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Prior art, such as U.S. Patent USP 4579675, relies on the condensation of beta-diketones with substituted amines, a process that necessitates the use of toluene or benzene for azeotropic water removal. This requirement not only extends reaction times considerably but also introduces safety and environmental concerns associated with handling large volumes of aromatic solvents. Furthermore, other established methods described in literature, such as those utilizing terminal alkynes and palladium catalysts (e.g., Pd(PPh3)2Cl2), involve prohibitively expensive raw materials and harsh reaction conditions that are ill-suited for ton-scale production. The reliance on sensitive organolithium reagents like n-butyllithium or isopropyllithium, as seen in Org. Lett. 2007, further exacerbates the issue by demanding strict anhydrous conditions and specialized equipment, thereby inflating the cost reduction in pharmaceutical intermediates manufacturing efforts. These conventional pathways often suffer from variable yields dependent on substituent effects and generate complex waste streams that complicate downstream processing.
The Novel Approach
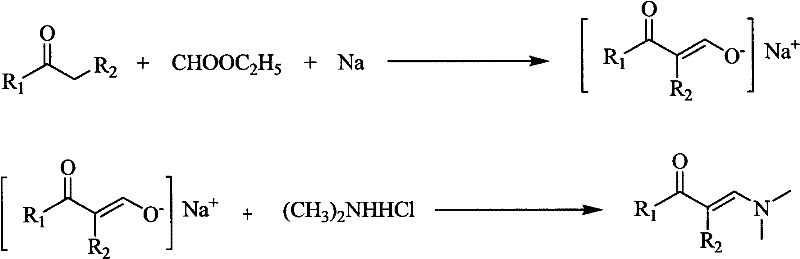
In stark contrast to these cumbersome legacy processes, the methodology outlined in CN101781222A presents a paradigm shift towards simplicity and efficiency. This novel approach utilizes a two-step sequence starting with the formylation of ketones using ethyl formate and an alkali metal, typically sodium, to generate a stable enolate intermediate. This intermediate is then subjected to a dehydration condensation reaction with dimethylamine hydrochloride under mild conditions. The brilliance of this route lies in its use of commodity chemicals; ethyl formate and sodium are inexpensive and widely available, eliminating the supply chain bottlenecks associated with specialty catalysts. The reaction proceeds smoothly at temperatures ranging from 0°C to 50°C, removing the need for energy-intensive heating or cryogenic cooling systems. Moreover, the process avoids the use of toxic heavy metals or moisture-sensitive organometallics, resulting in a cleaner reaction profile that facilitates easier product isolation. This strategic simplification directly addresses the pain points of reducing lead time for high-purity enaminones, allowing manufacturers to respond more agilely to market demands while maintaining rigorous quality standards.
Mechanistic Insights into Sodium-Mediated Enolate Formylation
To fully appreciate the technical superiority of this synthesis, one must delve into the mechanistic nuances of the sodium-mediated enolate formation. The process initiates with the suspension of an alkali metal, such as metallic sodium, in an organic solvent compatible with ethyl formate, such as diethyl ether or toluene. Upon the dropwise addition of the alpha-methyl or alpha-methylene ketone substrate mixed with ethyl formate, the alkali metal acts as a strong base and reducing agent, facilitating the deprotonation of the alpha-carbon. This generates a resonance-stabilized alkali metal enolate, a highly nucleophilic species that is crucial for the subsequent carbon-carbon bond formation. The molar ratio of the ketone to ethyl formate is carefully controlled, typically between 1:1 and 1:10, with a preferred ratio of 1:3 to drive the equilibrium towards the enolate. The presence of the organic solvent plays a dual role: it disperses the solid alkali metal to ensure uniform reaction kinetics and dissolves the resulting enolate salt to prevent passivation of the metal surface. This careful orchestration of reagents ensures a high conversion rate without the formation of significant by-products, laying the foundation for the high selectivity observed in the final enaminone product.
Following the formation of the enolate, the second stage involves the introduction of dimethylamine hydrochloride, usually as an aqueous solution, into the enolate suspension. This step triggers a nucleophilic attack and subsequent dehydration to form the carbon-nitrogen double bond characteristic of the enaminone structure. The use of an aqueous amine salt solution in a biphasic system with the organic solvent creates a mild reaction environment that controls the exotherm and prevents degradation of the sensitive intermediate. The reaction temperature is maintained between 0°C and 60°C, preferably 20-30°C, ensuring that the dehydration proceeds efficiently without promoting side reactions such as polymerization or hydrolysis. The resulting enaminone exists in the organic phase, allowing for straightforward separation via liquid-liquid extraction. This mechanistic pathway effectively bypasses the need for external dehydrating agents or Lewis acids, as the thermodynamics of the enolate formation and the subsequent condensation drive the reaction to completion. Understanding this mechanism is vital for a reliable enaminone supplier to optimize parameters for different substrates, ensuring consistent quality across diverse batches.
How to Synthesize Enaminone Compounds Efficiently
Implementing this synthesis route in a production setting requires adherence to specific operational parameters to maximize yield and safety. The process is designed to be robust, accommodating a variety of ketone substrates ranging from aliphatic ketones like acetone and butanone to aromatic ketones like acetophenone. The initial step involves creating a homogeneous suspension of the alkali metal, where the molar concentration of sodium is optimized between 0.5 mol/L and 5 mol/L to balance reaction rate and heat dissipation. Following the formylation, which typically takes 4 to 24 hours, the resulting enolate slurry is ready for the amination step. The addition of the dimethylamine hydrochloride solution must be controlled to manage gas evolution and maintain the pH balance necessary for the condensation. After the reaction is complete, usually within 1 to 5 hours, the workup involves standard unit operations: phase separation, washing of the organic layer to remove inorganic salts, drying over agents like anhydrous magnesium sulfate, and solvent removal. The crude product can then be purified via vacuum distillation or recrystallization using a petroleum ether and acetone system, yielding high-purity enaminones suitable for downstream pharmaceutical applications. For detailed standard operating procedures and specific batch records, please refer to the technical guide below.
- Suspend alkali metal (e.g., Sodium) in an organic solvent and add a mixture of alpha-methyl ketone and ethyl formate at 0-50°C to form the alkali metal enolate.
- Prepare a suspension of the obtained enolate in organic solvent and add an aqueous solution of dimethylamine hydrochloride dropwise at 0-60°C.
- Separate the organic layer, dry, concentrate, and purify the crude enaminone product via recrystallization or vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this enolate-based synthesis strategy offers profound benefits for procurement managers and supply chain directors focused on cost reduction in pharmaceutical intermediates manufacturing. The most immediate impact is the drastic reduction in raw material costs. By replacing expensive palladium catalysts, organolithium reagents, and specialized Lewis acids with commodity sodium metal and ethyl formate, the direct material cost of goods sold (COGS) is significantly lowered. Furthermore, the elimination of azeotropic distillation setups reduces energy consumption and capital expenditure on specialized glassware or reactors, contributing to substantial overall cost savings. The simplicity of the process also translates to reduced operator training requirements and lower maintenance costs for production equipment, as the mild reaction conditions place less stress on reactor linings and seals. This economic efficiency allows suppliers to offer more competitive pricing without compromising on margin, a critical factor in the highly price-sensitive generic pharmaceutical market.
- Cost Reduction in Manufacturing: The economic model of this synthesis is built on the substitution of high-cost inputs with low-cost alternatives. Traditional methods often require stoichiometric amounts of expensive dehydrating agents or catalytic quantities of precious metals that are difficult to recover. In this novel process, the reagents are inexpensive bulk chemicals, and the alkali metal byproduct (sodium salt) is water-soluble and easily removed during the aqueous wash, eliminating the need for costly metal scavenging resins. Additionally, the high selectivity of the reaction minimizes the formation of impurities, which reduces the load on purification units and increases the overall mass balance efficiency. This lean manufacturing approach ensures that resources are utilized effectively, driving down the cost per kilogram of the final API intermediate and enhancing the profitability of the entire value chain.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability of raw materials. The reagents specified in this patent—sodium, ethyl formate, dimethylamine hydrochloride, and common solvents like ether or toluene—are produced globally in massive quantities for various industries. This ubiquity means that the risk of supply disruption due to vendor-specific issues or geopolitical constraints is minimal. Unlike specialized catalysts that may have long lead times or single-source dependencies, these commodity chemicals can be sourced from multiple qualified vendors, ensuring continuity of supply. This diversification of the supply base is crucial for maintaining production schedules and meeting the just-in-time delivery expectations of multinational pharmaceutical clients. It effectively decouples the production of high-value enaminones from the volatility of the specialty chemical market.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden complexities, but this method is inherently scalable. The reaction does not generate hazardous gases or require extreme pressures, making it safer to operate at the 100 MT scale. From an environmental standpoint, the absence of heavy metals and chlorinated solvents (in many embodiments) simplifies waste treatment and disposal. The aqueous waste streams primarily contain inorganic salts and amines, which are easier to treat biologically or chemically compared to heavy metal-laden effluents. This alignment with green chemistry principles not only reduces environmental compliance costs but also enhances the corporate social responsibility profile of the manufacturer, a growing requirement for inclusion in the supply chains of top-tier global pharma companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enaminone synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure. The answers reflect the practical realities of scaling this chemistry, from reagent handling to final product specification.
Q: What are the primary advantages of this enolate-based synthesis over traditional Lewis acid methods?
A: This method eliminates the need for expensive Lewis acid catalysts and complex azeotropic water removal systems. It utilizes common alkali metals and mild temperatures (0-50°C), significantly simplifying the equipment requirements and reducing raw material costs compared to prior art like USP 4579675.
Q: Is this process suitable for large-scale industrial manufacturing of enaminones?
A: Yes, the patent explicitly states the process is adapted for large-scale industrial production. The reaction conditions are mild, avoiding harsh reagents like n-butyllithium, and the workup involves standard separation and distillation techniques that are easily scalable in a CDMO environment.
Q: What is the expected purity and yield profile for these enaminone intermediates?
A: The described method achieves high reaction selectivity and yields, with examples demonstrating isolation yields ranging from approximately 46% to over 93% depending on the substrate. The products are easily purified via recrystallization or vacuum distillation to meet stringent pharmaceutical specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Enaminone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising chemistry described in CN101781222A can be realized at the volumes your business demands. Our facilities are equipped with state-of-the-art reactors capable of handling alkali metal reactions safely, along with rigorous QC labs that enforce stringent purity specifications for every batch of enaminone intermediates we produce. We understand that consistency is key in pharmaceutical manufacturing, and our quality management systems are designed to detect and eliminate even trace impurities that could affect downstream synthesis. By partnering with us, you gain access to a supply chain that is not only cost-effective but also technically superior and compliant with international regulatory standards.
We invite you to leverage our technical capabilities to optimize your supply chain for enaminone compounds. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to contact our technical procurement team today to request specific COA data from our pilot runs and comprehensive route feasibility assessments. Whether you are developing a new generic API or optimizing an existing process, NINGBO INNO PHARMCHEM is committed to delivering high-purity enaminones that empower your R&D and production goals. Let us collaborate to turn this innovative synthetic method into a competitive advantage for your organization.
