Revolutionizing Coenzyme Q10 Production: A Deep Dive into the Grignard Coupling Method for Commercial Scale-Up
Revolutionizing Coenzyme Q10 Production: A Deep Dive into the Grignard Coupling Method for Commercial Scale-Up
The global demand for high-purity Coenzyme Q10 (Ubiquinone-10), a critical component in both pharmaceutical formulations and nutraceutical supplements, necessitates manufacturing processes that balance efficiency with rigorous quality control. Patent CN100351219C introduces a transformative approach to this challenge by leveraging a Grignard reagent coupling strategy that fundamentally alters the synthetic landscape for this valuable molecule. Unlike conventional routes that often rely on complex, multi-step condensations with variable yields, this invention utilizes a derivatized halogenated Q0 quinhydrone as a precise initiator. By reacting this specialized precursor with metallic magnesium to generate a reactive Grignard species, the process enables a direct and highly efficient coupling with decaprenyl bromide. This technical breakthrough not only streamlines the formation of the carbon-carbon bond between the quinone ring and the isoprenoid side chain but also establishes a robust foundation for cost reduction in pharmaceutical intermediates manufacturing. For R&D directors and supply chain leaders, understanding the mechanistic elegance of this patent is key to securing a reliable coenzyme q10 intermediate supplier capable of meeting future market demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for Coenzyme Q10 have historically been plagued by significant inefficiencies that impact both the economic viability and the environmental footprint of production. Classical methods often involve Friedel-Crafts alkylation or condensation reactions that struggle with regioselectivity, frequently resulting in a mixture of ortho- and para-substituted byproducts that are difficult and expensive to separate. Furthermore, these older techniques typically require harsh reaction conditions and stoichiometric amounts of Lewis acids, which generate substantial quantities of hazardous waste and complicate downstream processing. The cumulative effect of these limitations is a process with lower overall yields and higher operational costs, creating bottlenecks for manufacturers attempting to achieve commercial scale-up of complex pharmaceutical intermediates. Additionally, the sensitivity of the quinone moiety to over-reduction or unwanted side reactions during these traditional steps often necessitates extensive purification protocols, further eroding profit margins and extending lead times for high-purity coenzyme q10 intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN100351219C employs a sophisticated organometallic strategy that bypasses many of these inherent drawbacks. The core innovation lies in the use of a specifically designed halogenated starting material, Compound I (2,3-dimethoxy-5-bromo-6-methyl-1,4-dihydroxybenzene ether), which serves as the perfect scaffold for Grignard reagent formation. 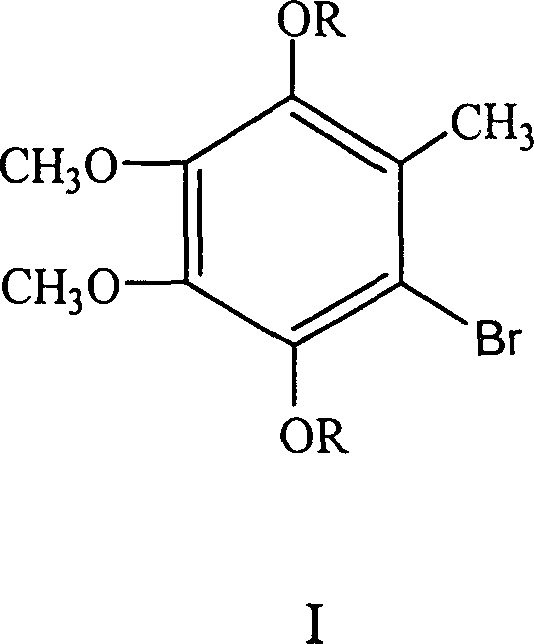 This precursor allows for the controlled generation of a nucleophilic Grignard species that reacts selectively with the electrophilic decaprenyl bromide. The result is the direct formation of the CoQ10 precursor (Compound II) with the side chain attached at the correct position, effectively eliminating the regio-isomer issues common in older syntheses. By reducing the synthesis to essentially two major transformation steps following the Grignard formation—coupling and subsequent oxidation—the process drastically simplifies the workflow. This streamlined approach not only enhances the theoretical yield but also significantly reduces the consumption of solvents and reagents, positioning it as a superior choice for modern, sustainable chemical manufacturing.
This precursor allows for the controlled generation of a nucleophilic Grignard species that reacts selectively with the electrophilic decaprenyl bromide. The result is the direct formation of the CoQ10 precursor (Compound II) with the side chain attached at the correct position, effectively eliminating the regio-isomer issues common in older syntheses. By reducing the synthesis to essentially two major transformation steps following the Grignard formation—coupling and subsequent oxidation—the process drastically simplifies the workflow. This streamlined approach not only enhances the theoretical yield but also significantly reduces the consumption of solvents and reagents, positioning it as a superior choice for modern, sustainable chemical manufacturing.
Mechanistic Insights into Grignard-Copper Catalyzed Coupling
The heart of this synthetic route is the copper-catalyzed cross-coupling reaction, which functions through a mechanism distinct from simple nucleophilic substitution. Initially, the halogenated Compound I undergoes oxidative insertion with metallic magnesium in a solvent system such as tetrahydrofuran (THF) or toluene at temperatures ranging from 40°C to 120°C. This generates the corresponding aryl magnesium bromide, a potent nucleophile. However, direct reaction of aryl Grignard reagents with alkyl halides can often lead to Wurtz-type homocoupling or beta-hydride elimination side reactions. To mitigate this, the patent specifies the inclusion of cuprous salts, such as cuprous iodide (CuI) or cuprous bromide, which act as transmetallation catalysts. The copper species softens the nucleophilicity of the Grignard reagent, facilitating a smoother attack on the decaprenyl bromide while suppressing unwanted side pathways. This catalytic cycle ensures that the coupling proceeds with high fidelity, achieving reported yields of 70% to 80% for the formation of Compound II. 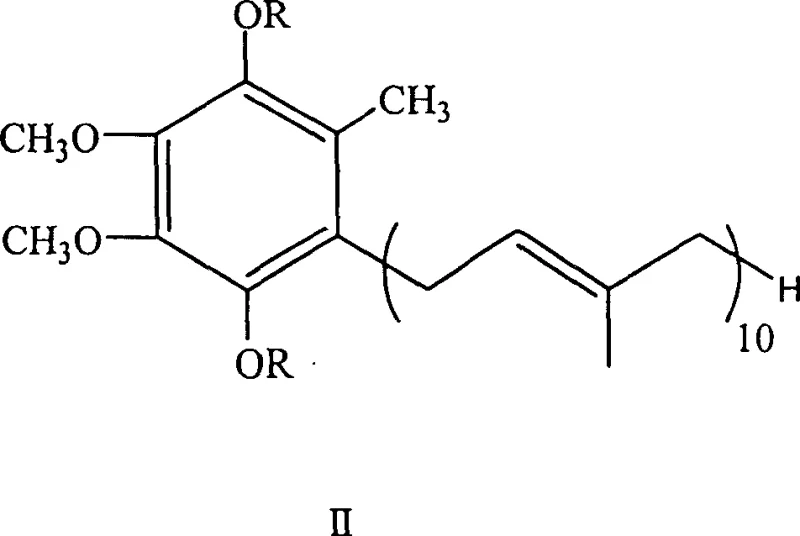 The structural integrity of Compound II, as depicted, confirms the successful attachment of the long isoprenoid chain to the aromatic core without degradation of the sensitive double bonds within the side chain.
The structural integrity of Compound II, as depicted, confirms the successful attachment of the long isoprenoid chain to the aromatic core without degradation of the sensitive double bonds within the side chain.
Following the coupling event, the process moves to a critical deprotection and oxidation phase that defines the final product quality. The coupled intermediate, which retains ether protecting groups (methoxy or benzyloxy) on the phenolic oxygens, is subjected to acidic hydrolysis using concentrated hydrochloric acid. This step cleanly removes the protecting groups to reveal the free hydroquinone structure (Compound III). The final transformation involves the oxidation of this hydroquinone to the corresponding quinone, utilizing mild yet effective oxidants like iron(III) chloride or ceric ammonium nitrate. This specific choice of oxidants is crucial for impurity control; stronger oxidants might degrade the polyunsaturated side chain, whereas FeCl3 and CAN provide the necessary redox potential to form the quinone carbonyls selectively. This meticulous control over the oxidation state ensures that the final Coenzyme Q10 product meets the stringent purity specifications required for pharmaceutical applications, minimizing the presence of colored impurities or over-oxidized byproducts that could compromise safety profiles.
How to Synthesize Coenzyme Q10 Efficiently
Implementing this Grignard-based methodology requires precise control over reaction parameters to maximize efficiency and safety. The process begins with the activation of magnesium metal, often requiring careful drying of the solvent system to prevent premature quenching of the Grignard reagent. Once the Grignard species is formed, the addition of the decaprenyl bromide must be managed to control the exotherm, typically maintaining the reaction temperature between 30°C and 80°C. The subsequent workup involves aqueous quenching and extraction, followed by the acid-mediated deprotection which must be monitored to ensure complete removal of ether groups without damaging the acid-sensitive isoprenoid tail. Finally, the oxidation step is performed in a biphasic or mixed solvent system to facilitate product isolation. For a detailed breakdown of the specific molar ratios, solvent choices, and temperature profiles validated in the patent examples, please refer to the standardized synthesis guide below.
- Preparation of Grignard Reagent: React halogenated Q0 quinhydrone derivative (Compound I) with metallic magnesium in THF or toluene at 40-120°C.
- Coupling Reaction: Treat the Grignard reagent with decaprenyl bromide using cuprous iodide catalyst at 30-80°C to form the coupled intermediate.
- Deprotection and Oxidation: Perform acid hydrolysis to remove protecting groups, followed by oxidation with FeCl3 or ceric ammonium nitrate to yield Coenzyme Q10.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the Grignard coupling process outlined in CN100351219C represents a strategic opportunity to optimize the sourcing of Coenzyme Q10 intermediates. The primary value driver is the substantial simplification of the synthetic route, which directly translates into reduced operational complexity and lower manufacturing overheads. By consolidating the construction of the molecule into fewer high-yielding steps, manufacturers can significantly reduce the volume of solvents and reagents required per kilogram of product. This efficiency gain is not merely theoretical; it results in a tangible decrease in waste disposal costs and energy consumption, aligning with modern green chemistry initiatives. Furthermore, the use of commodity chemicals like magnesium, copper iodide, and standard organic solvents ensures that the supply chain remains resilient against fluctuations in the availability of exotic or proprietary catalysts. This reliability is essential for maintaining continuous production schedules and avoiding costly delays in the delivery of critical active ingredients.
- Cost Reduction in Manufacturing: The elimination of complex multi-step protection and deprotection sequences found in older methodologies leads to a drastic simplification of the production workflow. By avoiding the need for expensive transition metal catalysts often used in palladium-coupling alternatives, and instead utilizing cost-effective copper salts, the raw material bill of materials is optimized. Additionally, the high selectivity of the Grignard coupling minimizes the formation of difficult-to-remove isomers, thereby reducing the burden on purification units such as chromatography columns or crystallization tanks. This streamlined process flow allows for higher throughput in existing reactor infrastructure, effectively lowering the unit cost of production without compromising on the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials, such as halogenated hydroquinones and geranyl/decaprenyl bromides, mitigates the risk of supply disruptions associated with niche reagents. The robustness of the Grignard reaction conditions, which tolerate a range of temperatures and solvent systems (THF, benzene, toluene), provides manufacturing flexibility. This adaptability allows production facilities to switch between solvent grades or sources based on market availability without needing to requalify the entire process. Consequently, suppliers utilizing this technology can offer more consistent lead times and greater volume flexibility, ensuring that downstream pharmaceutical customers receive their orders on schedule regardless of minor upstream logistical variations.
- Scalability and Environmental Compliance: From an environmental perspective, this route offers significant advantages regarding waste management and regulatory compliance. The byproducts generated, primarily magnesium salts and copper residues, are well-understood and can be treated using standard effluent treatment protocols. The avoidance of heavy metals like palladium, which require stringent residual limits in pharmaceutical products, simplifies the quality control burden and reduces the risk of batch rejection. Moreover, the high atom economy of the coupling step means less unreacted material ends up as waste. This makes the process highly scalable from pilot plant batches of 100 kgs to full commercial production of 100 MT annually, satisfying the growing global demand for CoQ10 while adhering to increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Grignard coupling technology for Coenzyme Q10 synthesis. These insights are derived directly from the experimental data and claims presented in patent CN100351219C, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these details is crucial for technical teams assessing the compatibility of this process with existing facility capabilities and quality standards.
Q: What are the key advantages of the Grignard coupling method over traditional Friedel-Crafts alkylation for CoQ10?
A: The Grignard coupling method described in CN100351219C offers superior regioselectivity and yield (70-80% in the coupling step) compared to traditional methods which often suffer from poly-alkylation side reactions and difficult purification processes.
Q: Which oxidants are compatible with the final step of this synthesis route?
A: The patent specifies the use of either iron(III) chloride (FeCl3) or ceric ammonium nitrate (CAN) as effective oxidants to convert the hydroquinone intermediate into the final ubiquinone-10 quinone structure.
Q: Is this process scalable for industrial production of high-purity CoQ10?
A: Yes, the process utilizes standard industrial solvents like THF and toluene and avoids exotic catalysts, making it highly amenable to commercial scale-up from kilogram to metric ton quantities while maintaining stringent purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coenzyme Q10 Supplier
The technological advancements detailed in patent CN100351219C underscore the immense potential for optimizing Coenzyme Q10 production through intelligent process design. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory methodologies into robust, industrial-scale realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of complex intermediates or large-volume supply of finished APIs, our capability to implement advanced coupling chemistries positions us as a strategic partner for your long-term growth.
We invite you to engage with our technical procurement team to discuss how this Grignard-based route can be tailored to your specific cost and quality objectives. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your supply chain configuration. We encourage you to contact us today to obtain specific COA data for our CoQ10 intermediates and to receive comprehensive route feasibility assessments that demonstrate our commitment to excellence in fine chemical manufacturing.
